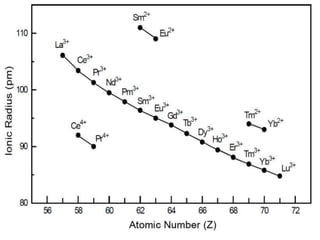
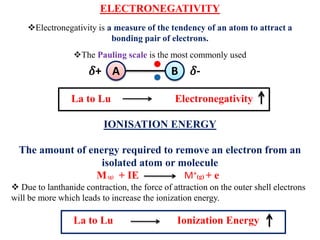
The lanthanide contraction is the result of poor shielding of the 4f electrons by inner orbitals. As atomic number increases from La to Lu, the nuclear charge increases and 4f electrons penetrate closer to the nucleus. This causes the atomic and ionic radii to steadily decrease, resulting in smaller sizes down the lanthanide series. Due to the decreasing size, properties like ionization energy and electronegativity increase while chemical reactivity and basicity of hydroxides decrease from La to Lu.



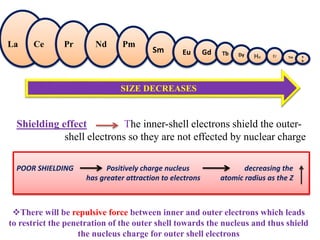
![5d
6s
4f
nucleus
1
2
3
1
Nucleus charge on f-orbitals is
more
[Attractive force > Repulsive
force]
2
Due to unsymmetrical shape of
f-orbital , it cannot shield the
outer orbitals so, the outer
orbitals also feels nuclear
charge
3
Nucleus force of attraction on
outer electron increases
and therefore it inward towards
the nucleus and cause reduce in
size
Outer shell
Inner shell](https://image.slidesharecdn.com/lanthanidecontraction-210131035623/85/Lanthanide-contraction-6-320.jpg)

![SEPERATION OF LANTHANIDES
Since all the elements exhibit the +3 oxidation state as common,
they have similar properties
Thus, the separation of elements in its pure state is difficult.
we use Repeated Fractional Crystallization
[ Ion Exchange method]
Similarities in atomic size of second and third transition series
elements :
Due to lanthanide contraction, the atomic and ionic radius of the
next and before on the lanthanide elements with same group, are
about same.
For examples, the atomic radius of Zr [ group — 4 , period —
5] and Hf [ group — 4 , period — 6] are 160 pm and 159
pm respectively .](https://image.slidesharecdn.com/lanthanidecontraction-210131035623/85/Lanthanide-contraction-9-320.jpg)
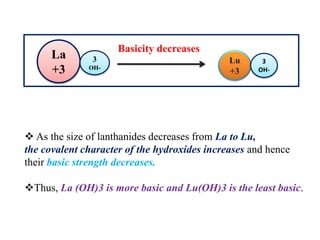
![BASIC STRENGTH OF HYDROXIDES
As the size decreases, charge to size ratio increase, the ionic
character decreases or covalent character increases making
hydroxides less and less basic [removal of OH-]
More the charge to size ratio, the electron cloud of anion is
more polarized, more covalent character [ fajan’s rule]
CHARGE
COVALENT CHARACTER
SIZE](https://image.slidesharecdn.com/lanthanidecontraction-210131035623/85/Lanthanide-contraction-11-320.jpg)