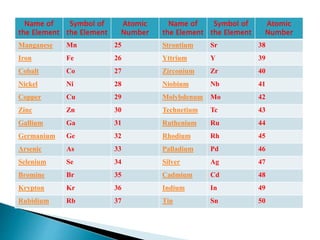
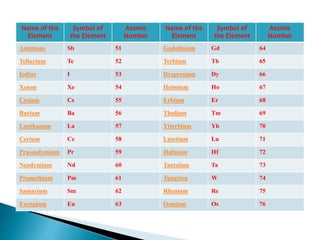
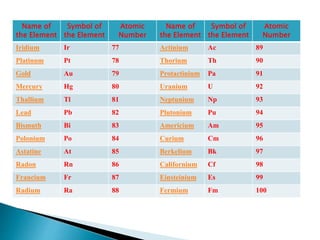
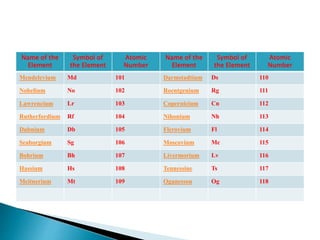
The document discusses the historical development of the periodic table, detailing contributions from various scientists including Dobereiner, Newlands, Meyer, Mendeleev, and Moseley. It highlights the classification systems based on atomic mass and the eventual shift to atomic number as the organizing principle for the modern periodic table. Key concepts such as periodicity, grouping of elements, and the prediction of undiscovered elements are emphasized throughout.








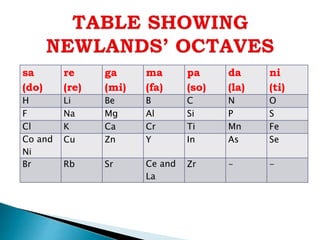
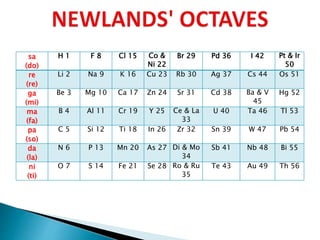
![ It contained the elements from hydrogen to thorium.
Properties of every eighth element were similar to that of the first element.
Seeing the word octave applied to this table may lead one to think that Newlands
recognised periods of eight elements with repeating properties, as we do with the modern
periodic table, for example: Li Be B C N O F Ne.
However, each sequence of Newlands' octaves contain only seven elements. Count the
elements in the columns! In Newlands' day the group 8 (18) rare gas elements, He, Ne, Ar,
Kr & Xe, had not yet been discovered.
To Newlands, H to F & F to Cl are octaves of eight elements, the eighth element repeating
the properties of the first.
There are seven notes in a musical octave: A B C D E F G, after which you start again with
A'; similarly for Newlands, seven elements H Li G Bo C N O, then the 8th is F and you start
again. [Note that Newlands treated H as a halogen.]](https://image.slidesharecdn.com/historicaldevelopmentofperiodictable-200503140526/85/Historical-development-of-periodic-table-10-320.jpg)