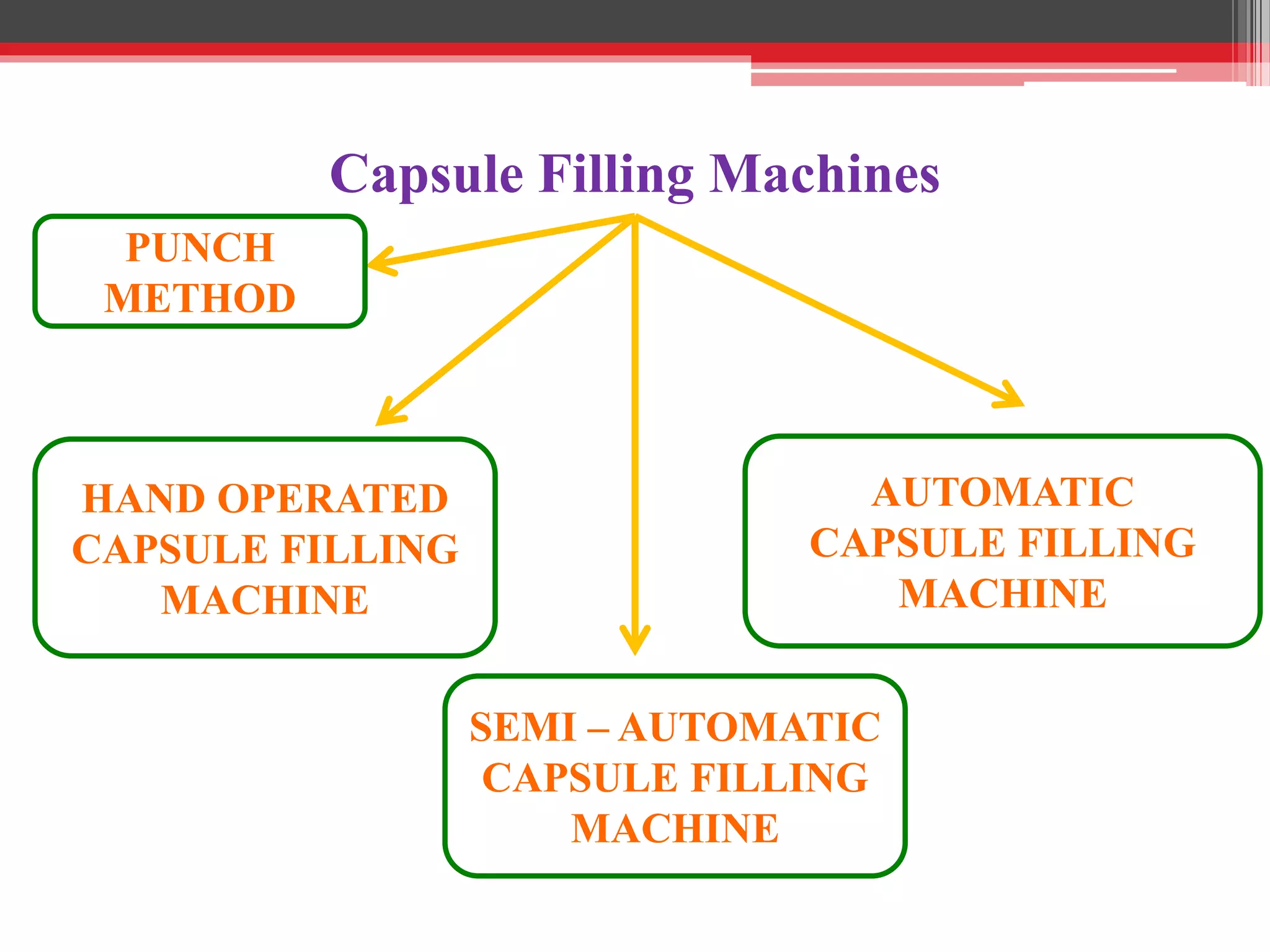
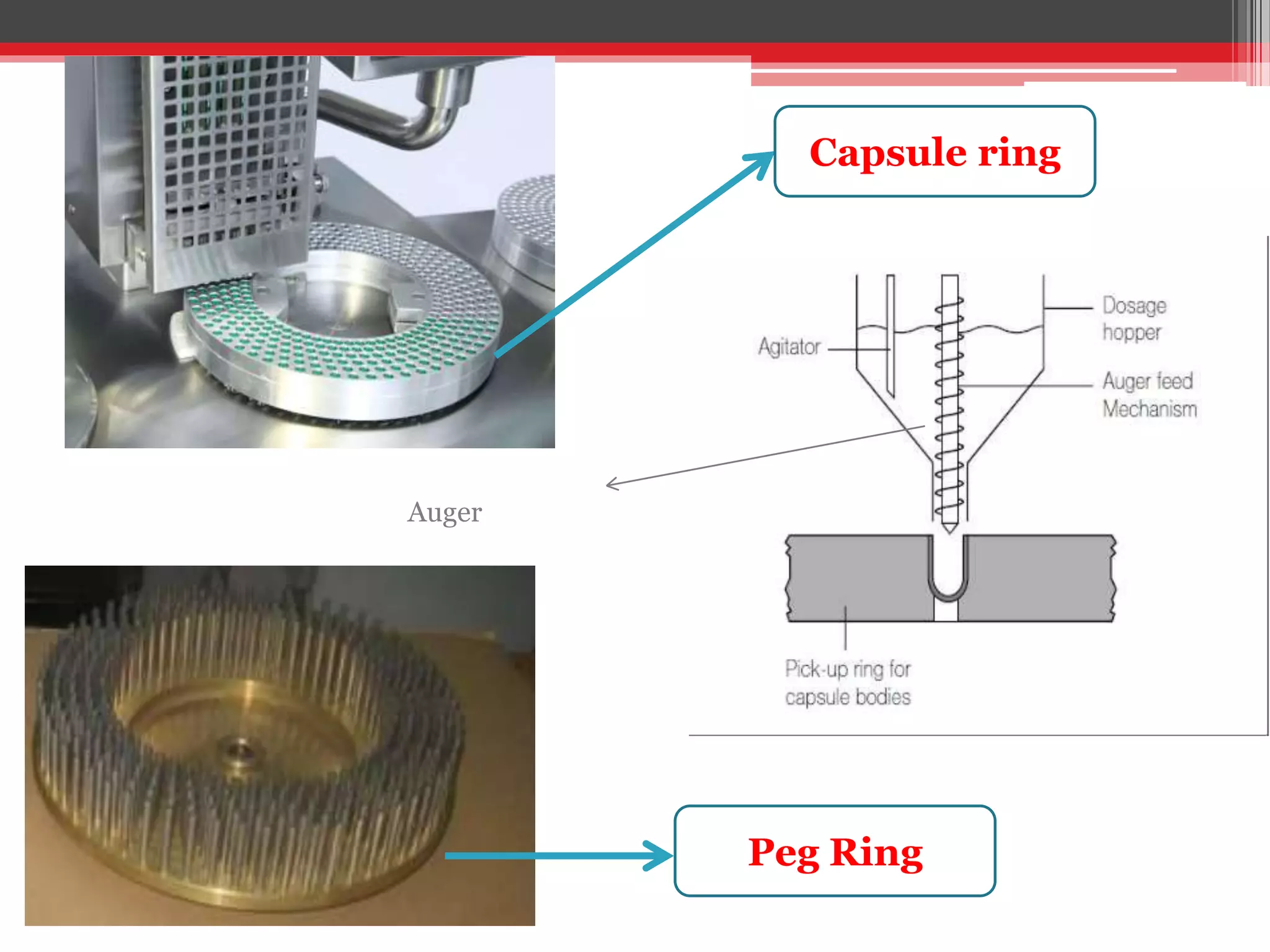
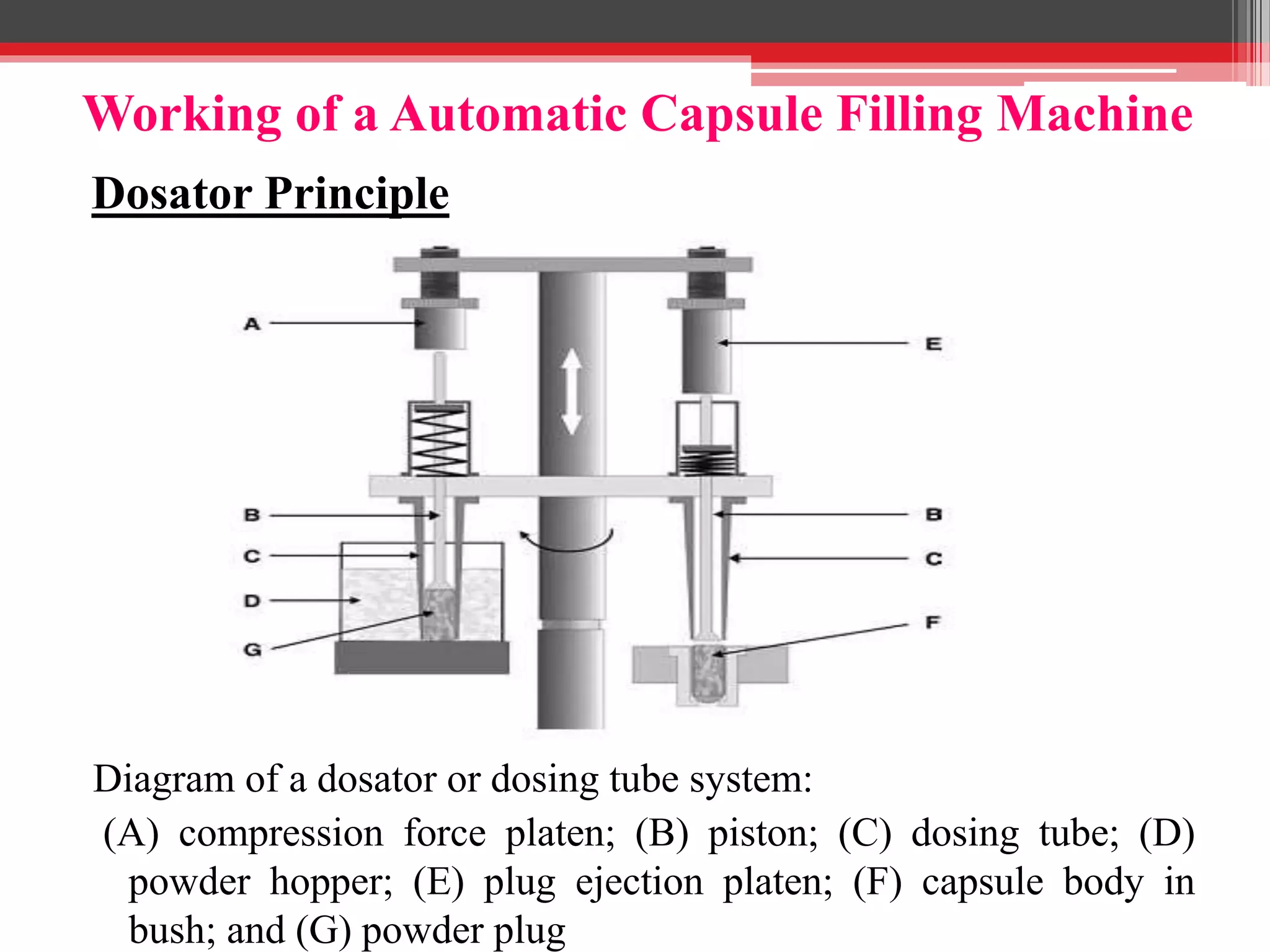
The document provides a comprehensive overview of hard gelatin capsules, detailing their production process, filling techniques, and quality control tests. It discusses capsule components, manufacturing methods, equipment, and special formulation techniques, as well as the various sizes and types of capsules available. Essential quality control measures, including weight variation and dissolution tests, are also highlighted to ensure product consistency and efficacy.