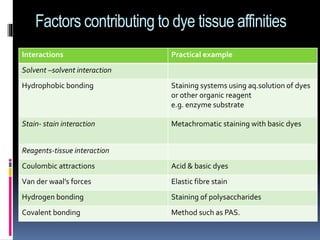
The document discusses the principles and methods of hematoxylin and eosin (H&E) staining, a common histopathological technique used to outline tissue and cellular components to identify diseases. It covers the types of stains, classifications of stains, staining reactions, and the interaction between stains and tissue components, including historical developments of staining techniques. Various methods and factors influencing staining reactions, as well as practical tips for effective staining procedures, are also detailed.