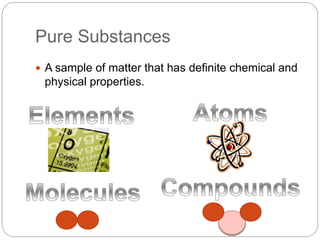
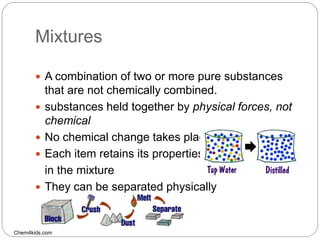
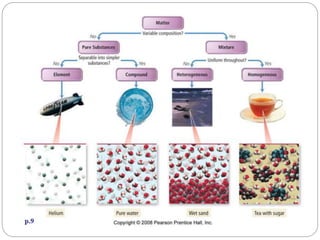
The document discusses the classification of matter into pure substances, elements, compounds, and mixtures. Elements are pure substances made of only one type of atom that cannot be broken down further. Compounds are pure substances composed of two or more elements chemically bonded together in a fixed ratio. Mixtures are combinations of substances that are not chemically bonded and can be separated physically. The document provides examples of elements, compounds, and mixtures and asks the reader to identify which class each example belongs to.