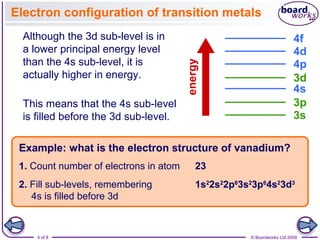
This document discusses electron configurations of atoms, ions, and transition metals. It provides examples of writing the electron configuration of vanadium and nickel ions. For atoms, electrons are filled into sublevels starting with the lowest energy level. For transition metals, the 4s sublevel is filled before the 3d sublevel even though 3d has a higher energy. When writing configurations for ions, electrons are added or removed according to the ion's charge, starting with the outermost sublevels first except for transition metal ions which remove electrons from the 4s sublevel before the 3d sublevel.