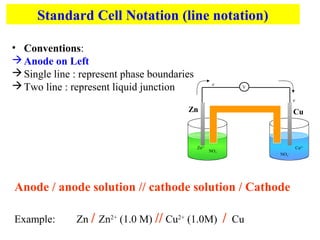
This document introduces electrochemistry and electrochemical cells. It discusses oxidation, reduction, galvanic cells which produce electrical current from spontaneous reactions, and electrolytic cells which consume current for non-spontaneous reactions. It also describes the parts of electrochemical cells including the anode, cathode, salt bridge, and electron flow. Standard cell notation is introduced to represent electrochemical cells using line notation.