Embed presentation
Downloaded 113 times

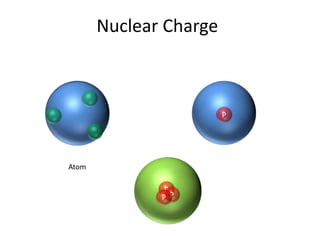


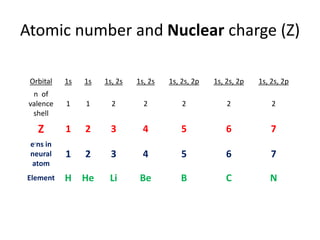




1) Effective nuclear charge (Zeff) is the nuclear charge actually experienced by an electron of an atom, which depends on the orbital and is less than the actual nuclear charge due to shielding by inner shell electrons. 2) Zeff is calculated using the formula Zeff = Z - σ, where Z is the actual nuclear charge and σ is the shielding constant which accounts for screening of the nuclear charge by inner electrons. 3) Factors that affect Zeff include the size of the atom, total nuclear charge, shielding by inner electrons, and penetration of the outer electron into the charge cloud.








