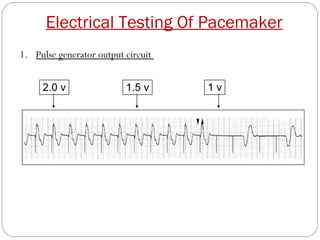
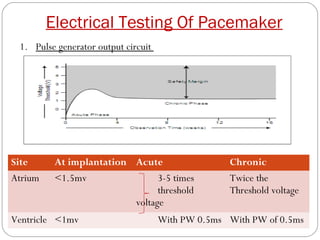
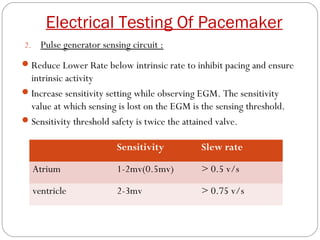
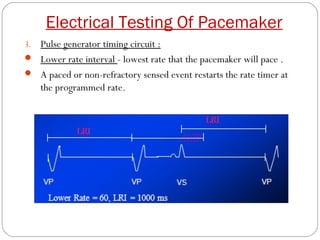
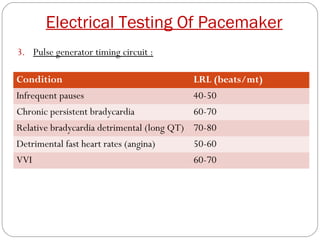
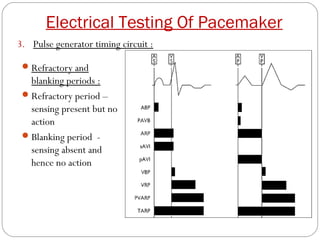
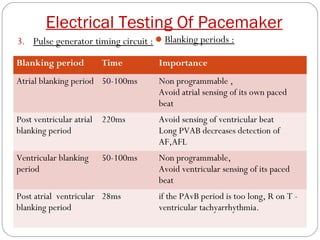
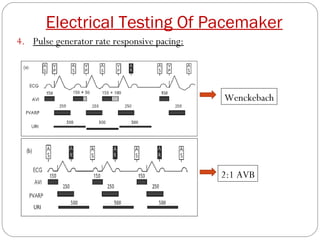
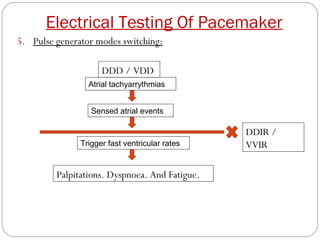
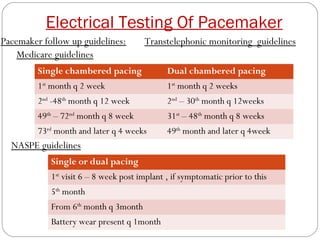
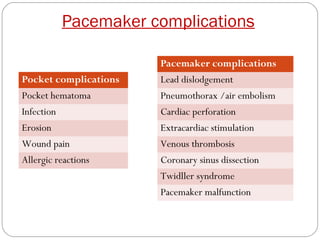
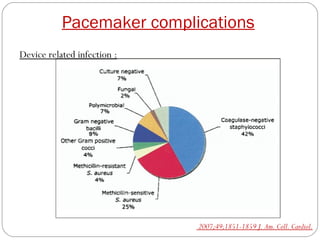
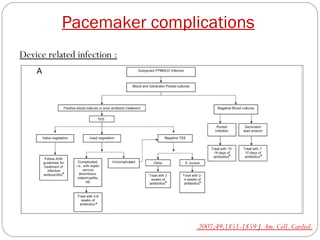
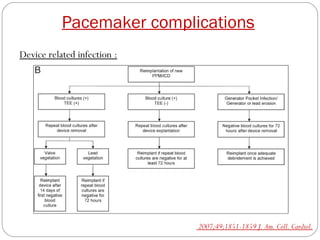
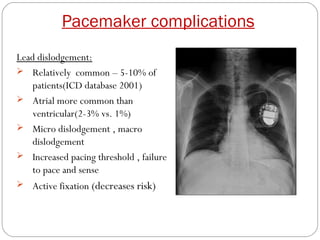
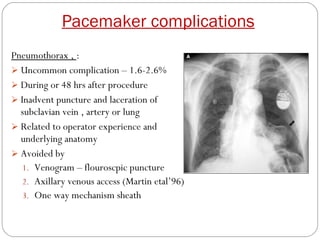
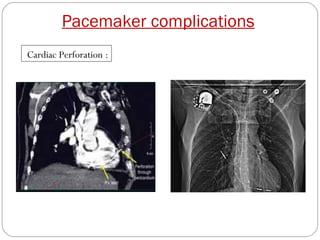
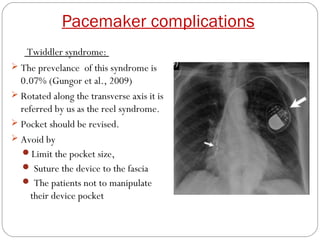
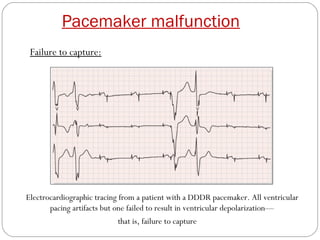
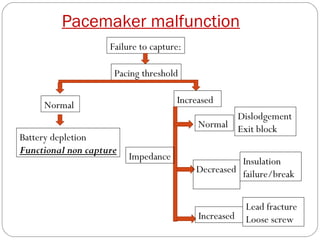
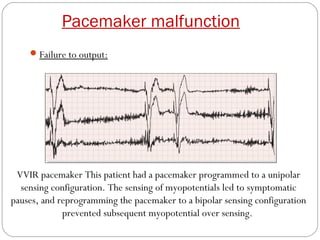
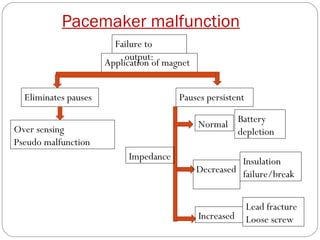
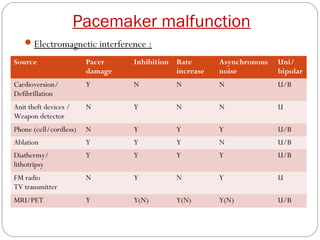
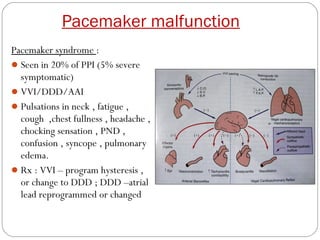
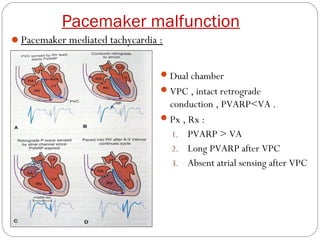
This document discusses electrical testing of pacemakers and pacemaker complications. It describes the components of a pacemaker including the battery, pacing impedance, pulse generator, and modes and mode switching. It then discusses testing various aspects of the pulse generator including output circuit, sensing circuit, timing circuit, and rate responsive pacing. Finally, it briefly outlines some common pacemaker complications such as pocket complications, lead issues, infections, and device malfunctions.