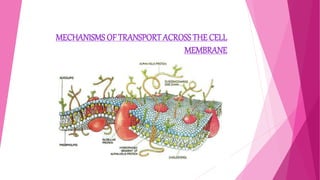
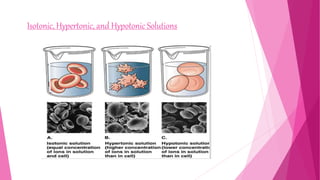
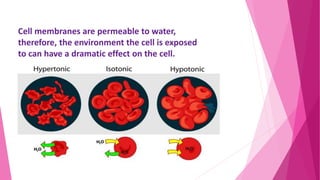
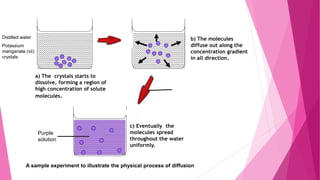
The document summarizes the main mechanisms of transport across the cell membrane: passive transport and active transport. Passive transport includes osmosis, simple diffusion, and facilitated diffusion, and does not require energy. Active transport requires the cell to use energy to move molecules against a concentration gradient. The document also describes types of solutions (hypotonic, hypertonic, isotonic) and how they affect cell volume. Diffusion is defined as the net movement of substances from an area of high concentration to low concentration down a concentration gradient.