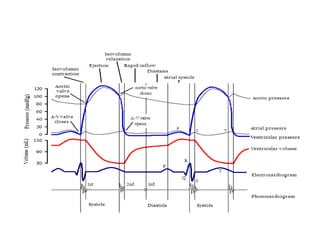
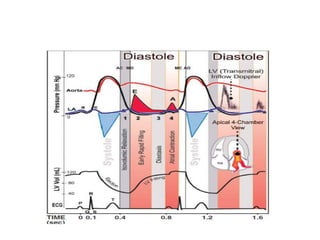
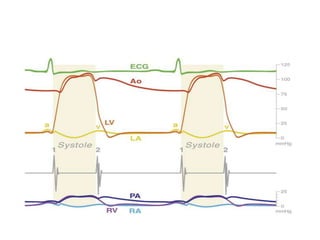
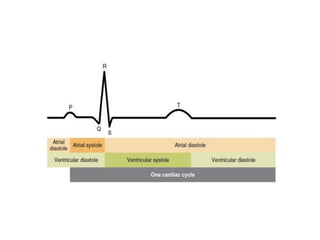
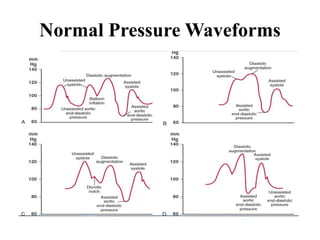
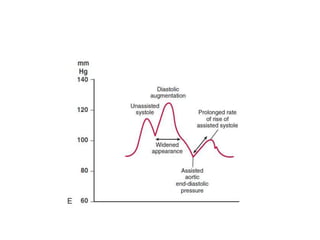
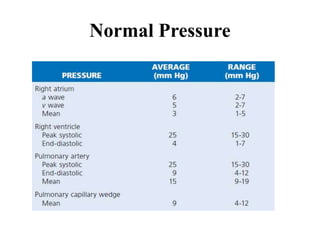
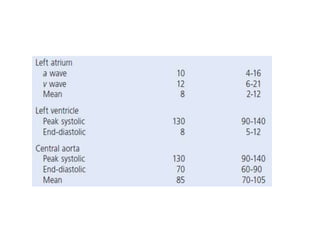
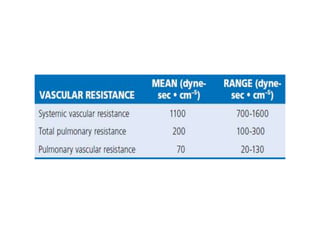
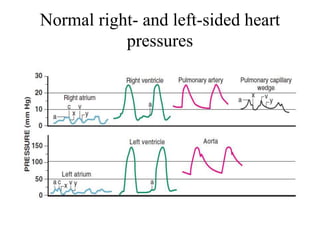
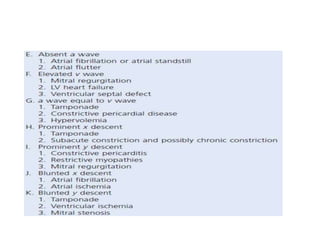
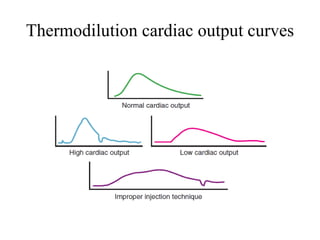
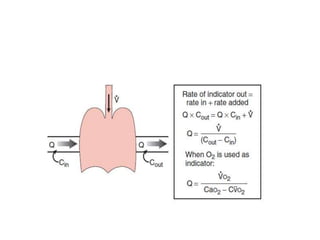
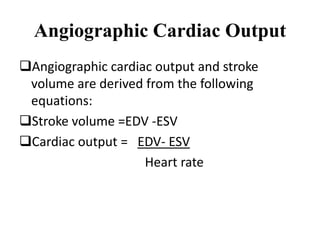
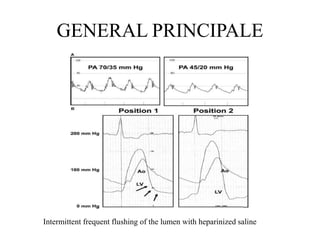
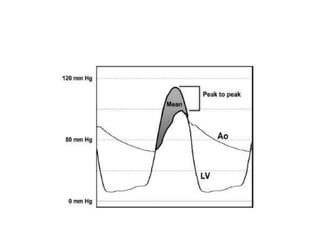
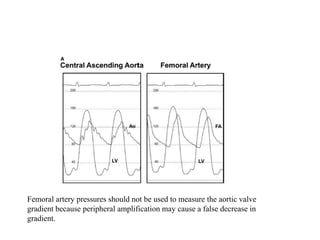
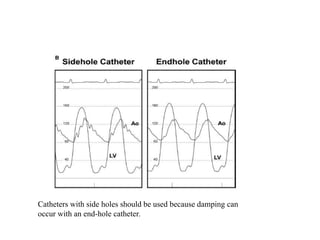
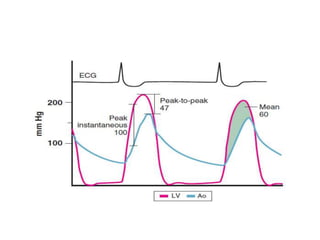
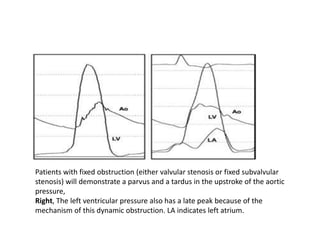
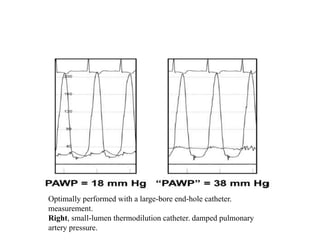
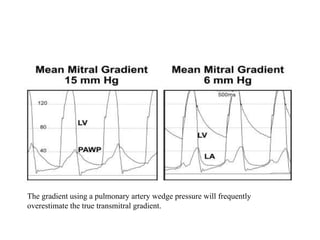
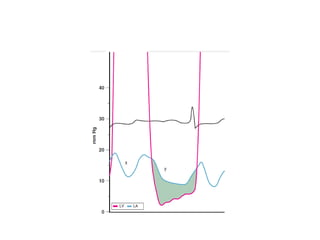
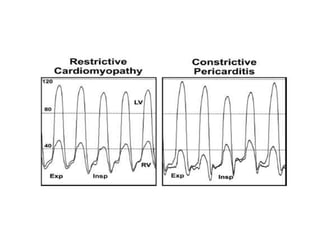
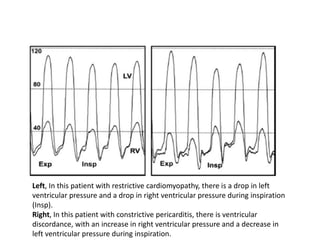
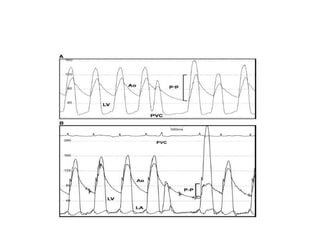
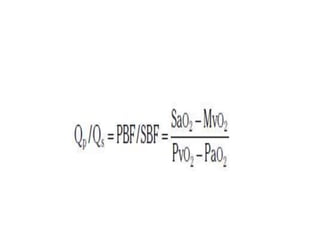
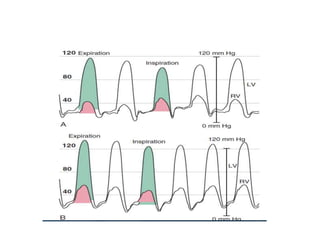
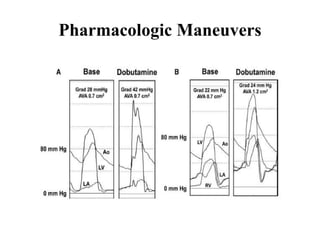
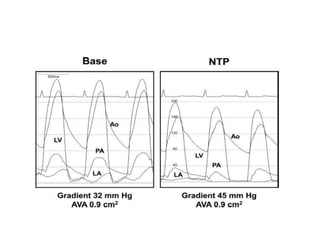
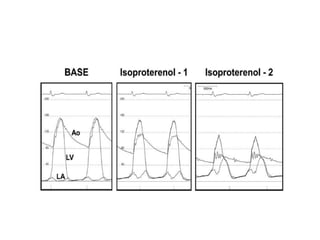
The document summarizes key aspects of cardiac catheterization and hemodynamic data collection. It describes the normal cardiac cycle, pressure measurement systems, normal pressure waveforms, methods to measure cardiac output like thermodilution and Fick, how to evaluate valvular stenosis and regurgitation, determine vascular resistance and shunts. Specific details are provided on assessing aortic stenosis, mitral stenosis, right-sided valves and quantifying regurgitant fractions. Oxygen saturation analysis and Fick principles are outlined for shunt determinations.