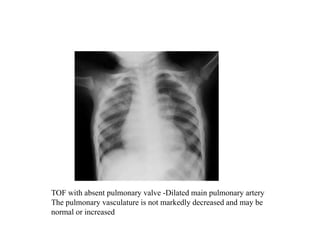
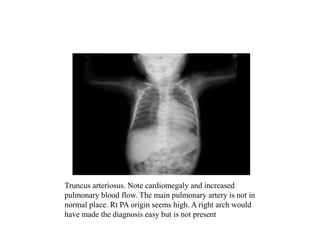
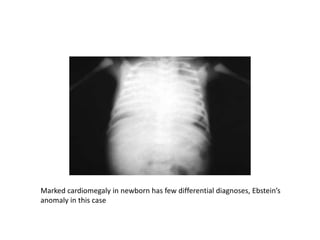
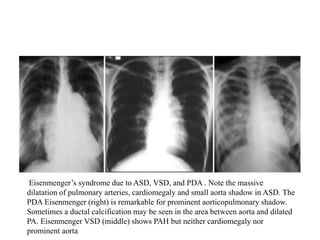
This document provides guidance on evaluating chest x-rays for congenital heart disease. It outlines key aspects to examine such as cardiac size and position, pulmonary vasculature, and abnormalities in specific heart structures. Common errors made in interpretation are also discussed, such as misdiagnosing over- or under-penetration of images. Example x-rays of various congenital heart conditions are provided to demonstrate typical features, including tetralogy of Fallot, transposition of the great vessels, and total anomalous pulmonary venous return.