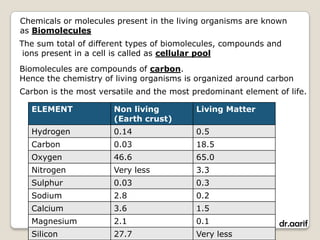
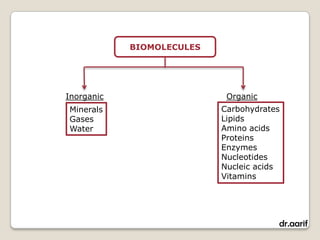
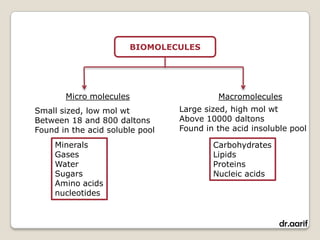
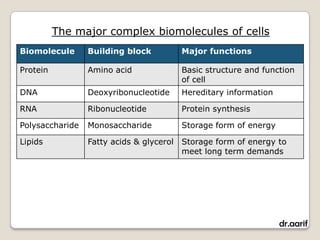
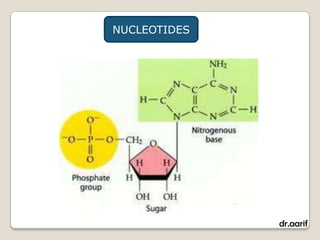
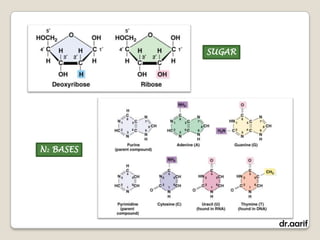
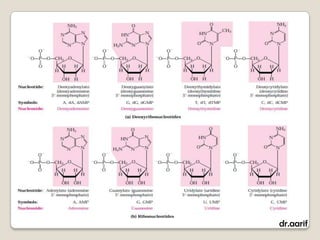
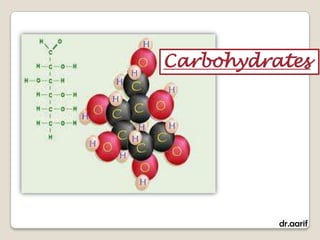
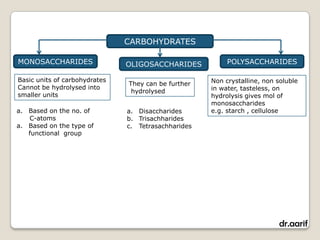
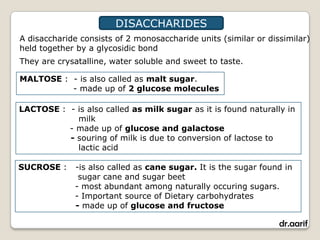
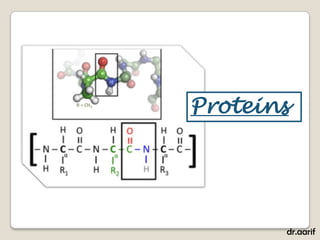
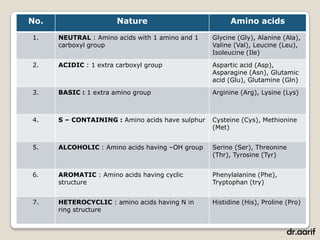
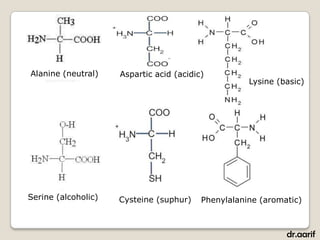
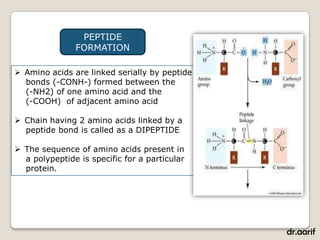
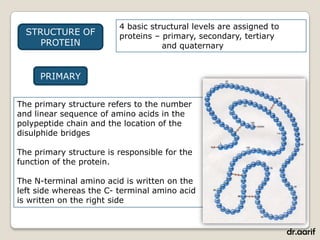
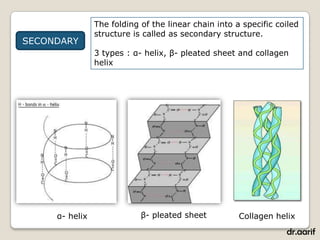
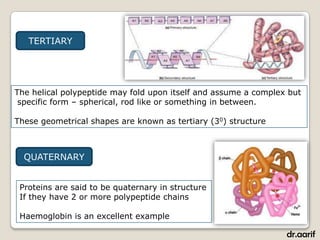
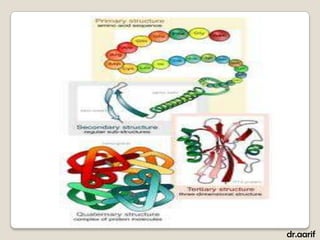
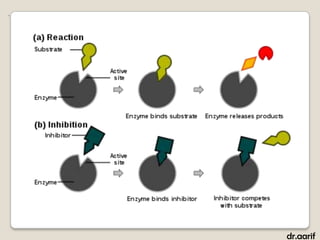
Here are the key points about lipids:
- Lipids are organic compounds that are relatively insoluble in water but soluble in organic solvents. They serve important structural and energy storage functions.
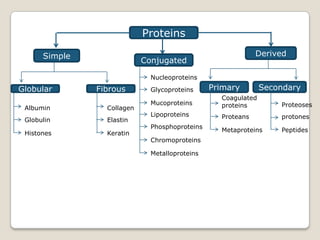
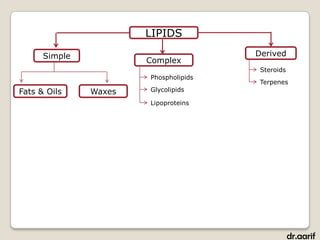
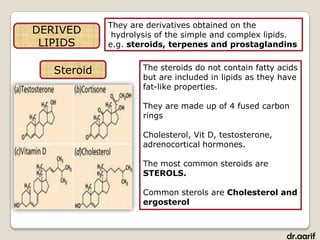
- The main types of lipids include fatty acids, triglycerides, phospholipids, and steroids.
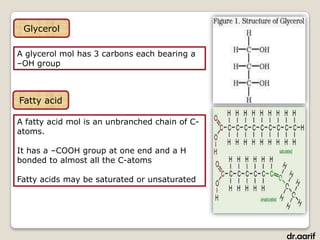
- Fatty acids are the building blocks of fats and oils. They are long hydrocarbon chains that can be saturated or unsaturated.
- Triglycerides are composed of fatty acids attached to a glycerol backbone. They are the main form of energy storage in animals.
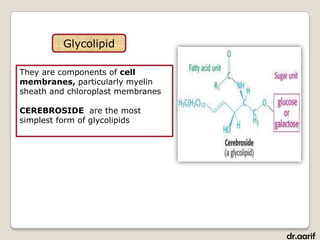
- Phospholipids are a major component of cell membranes. They have a glycerol backbone with two fatty acids and