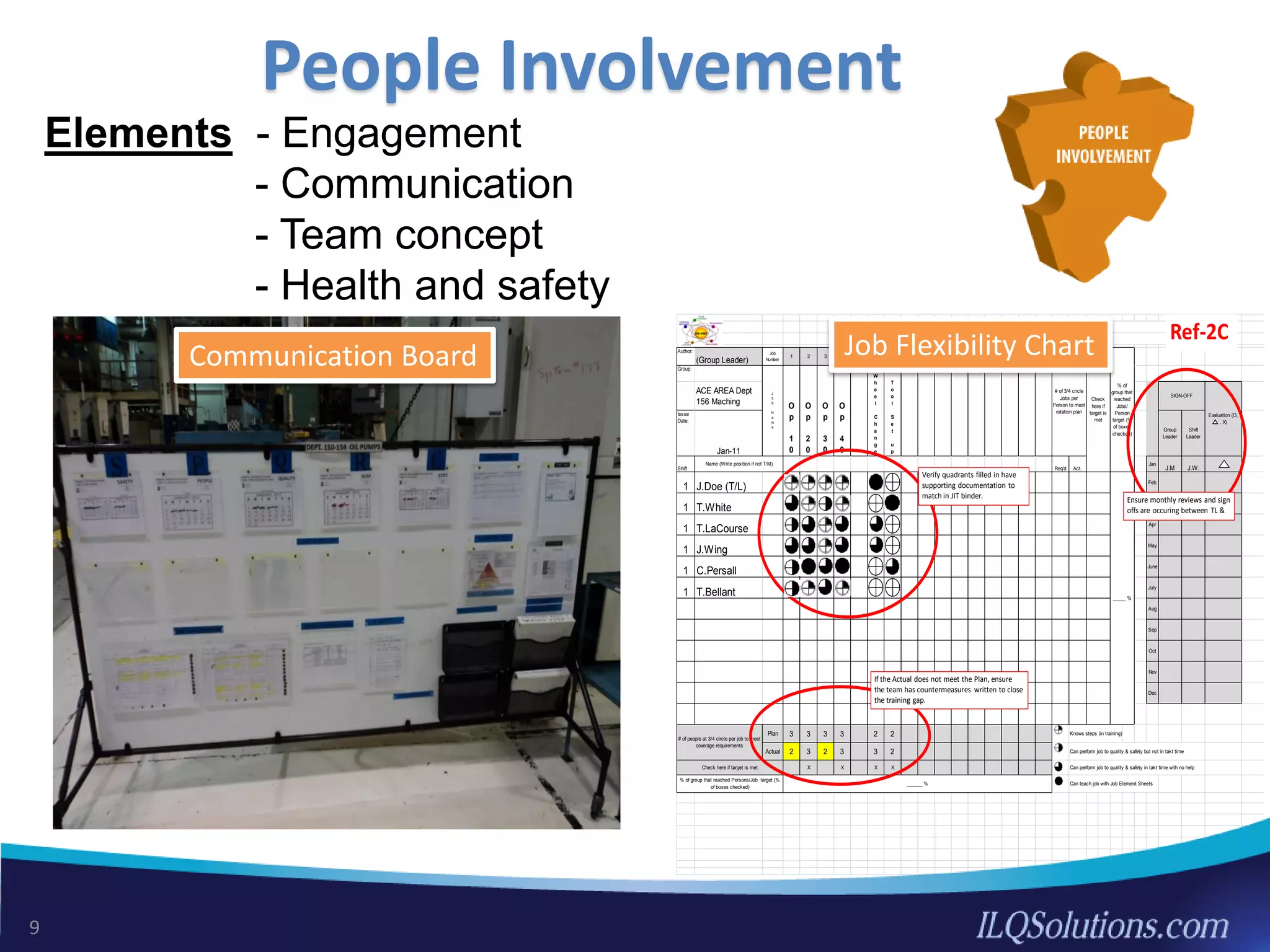
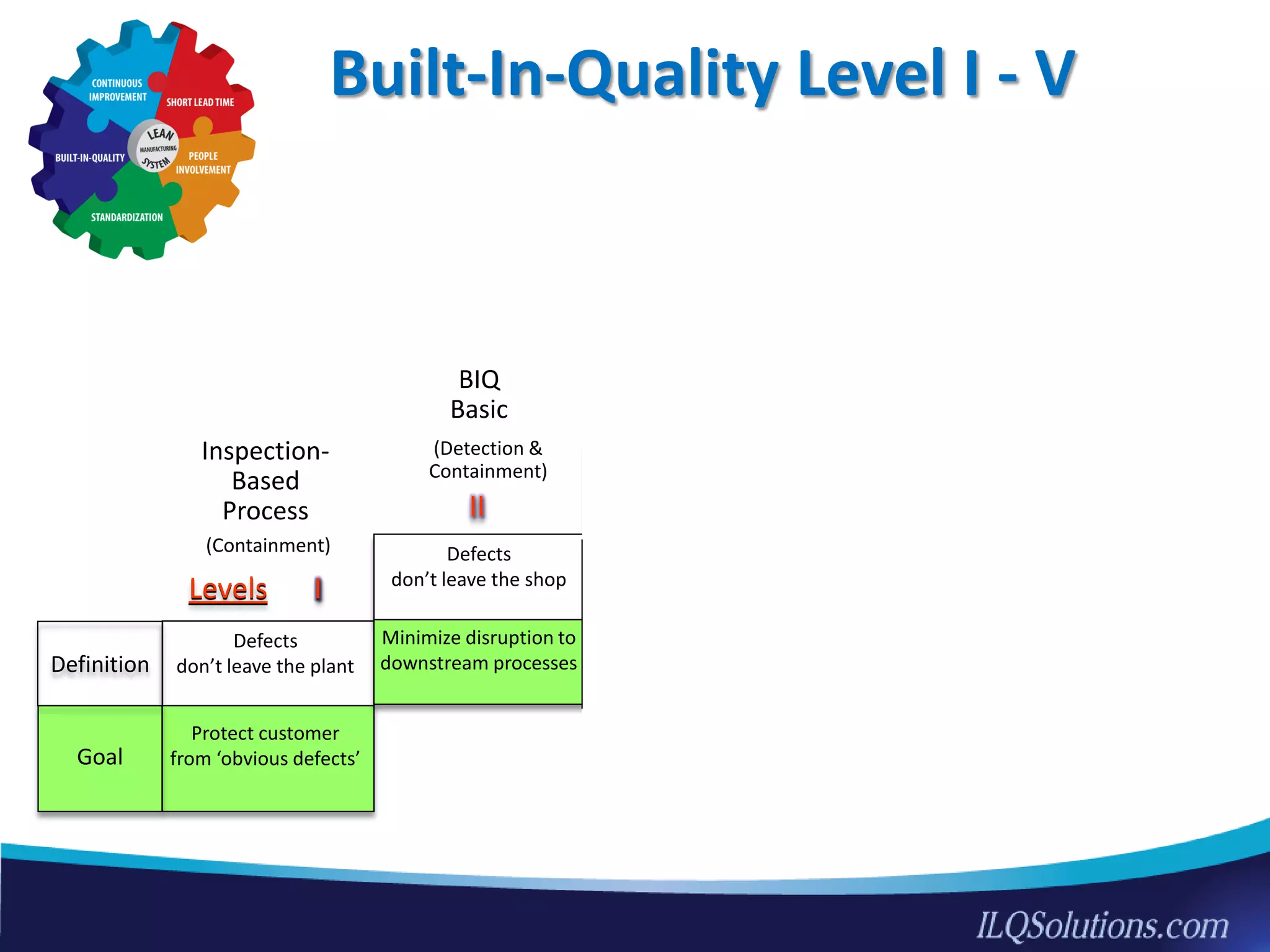
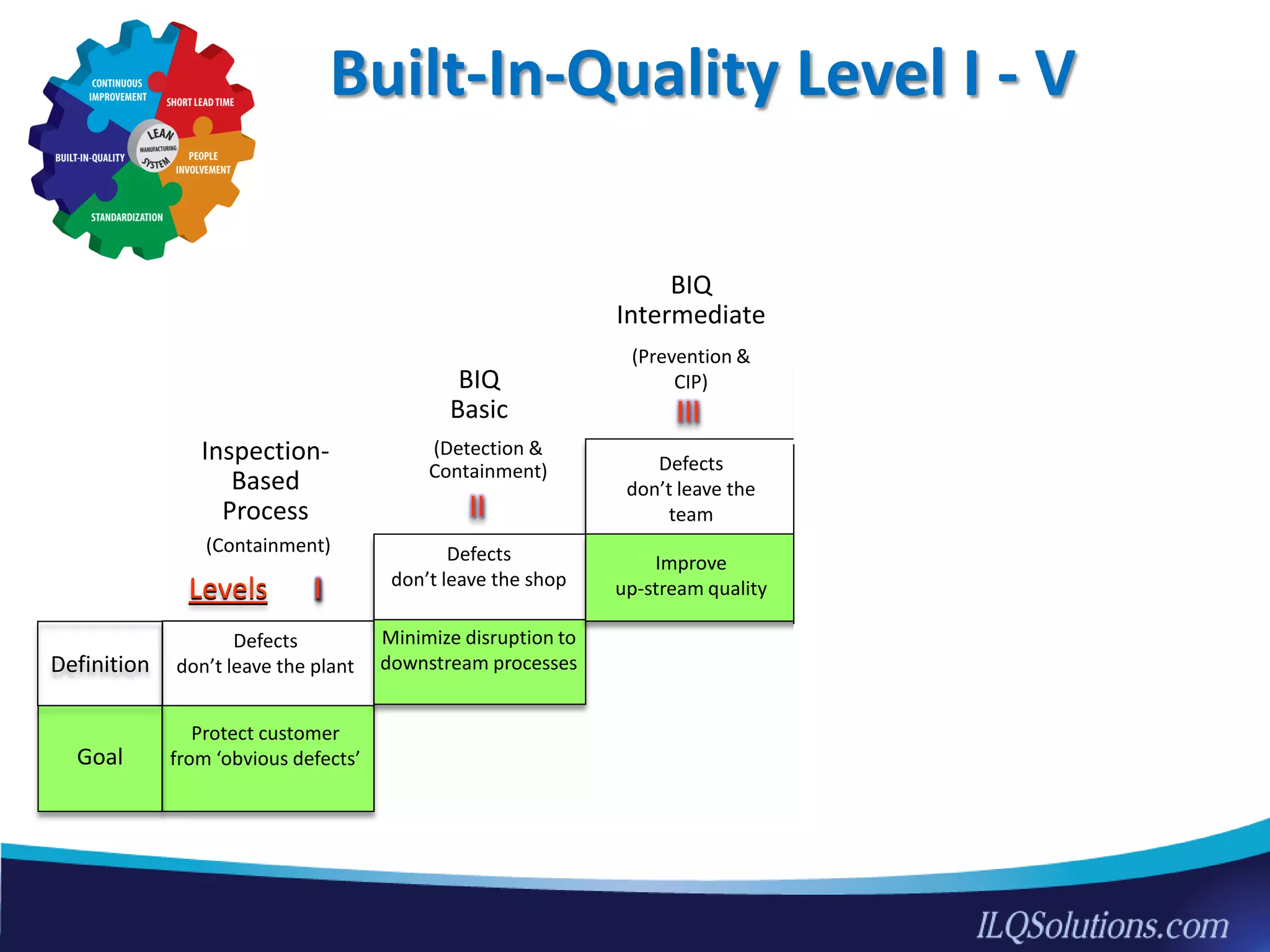
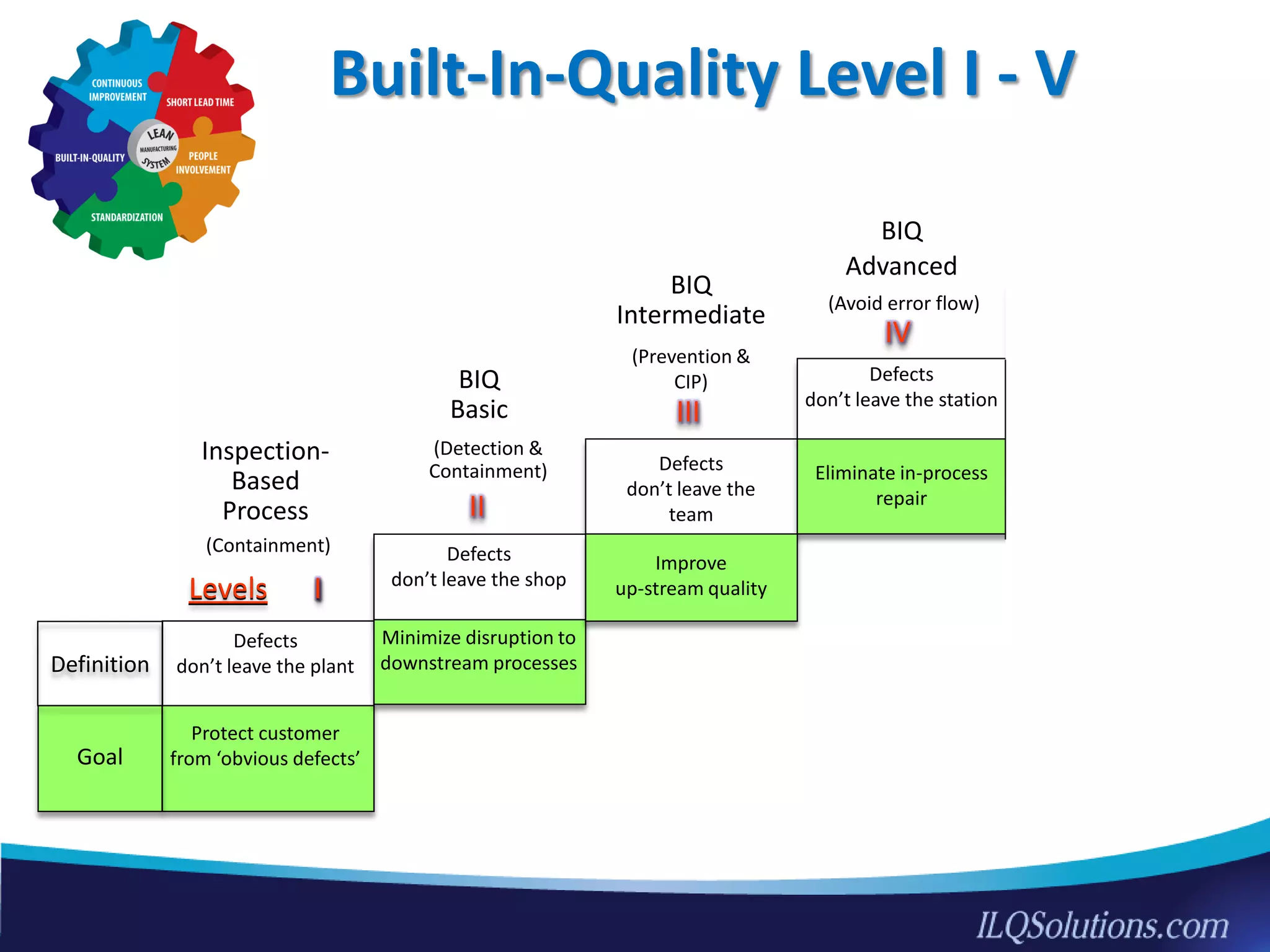
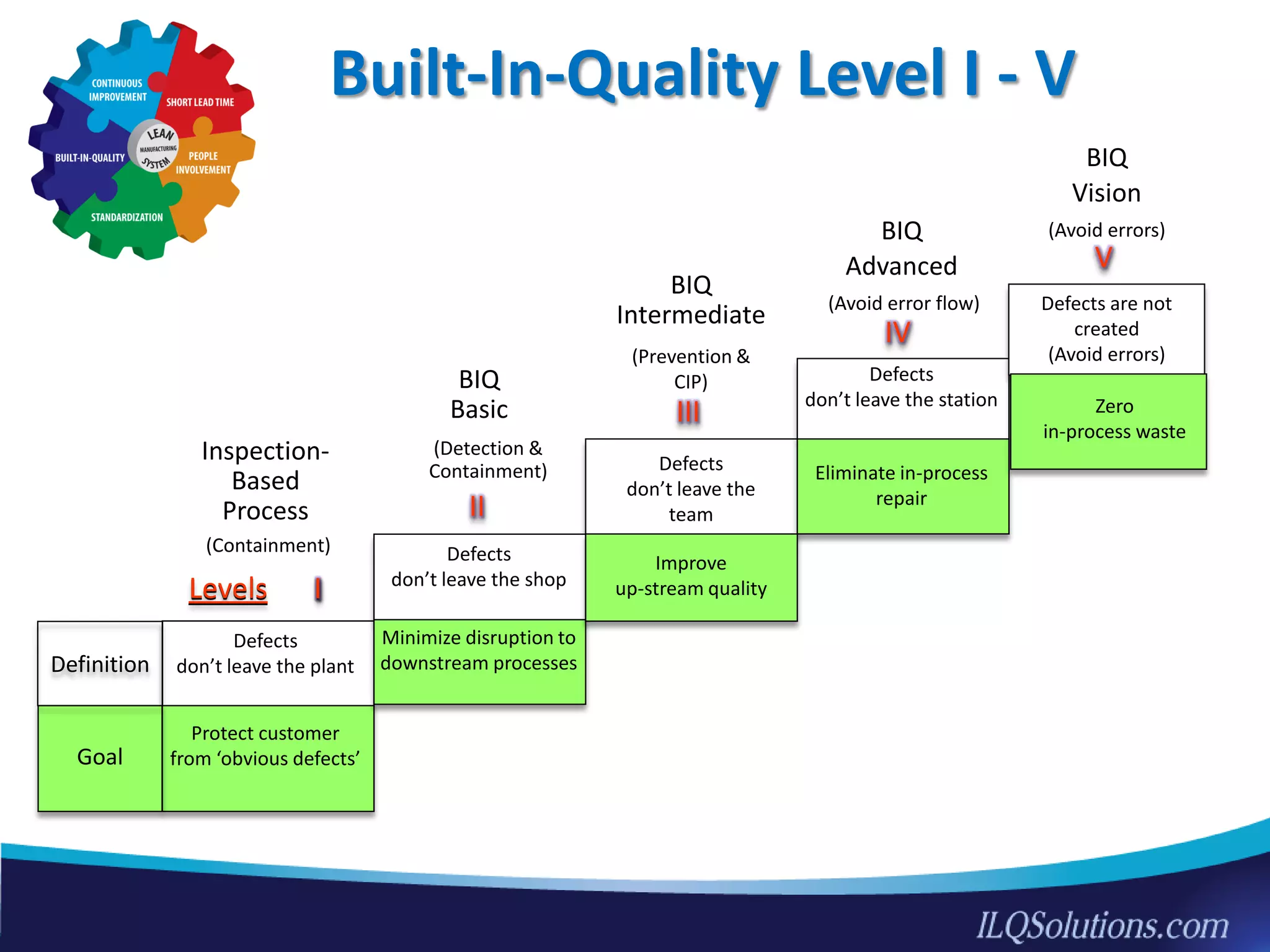
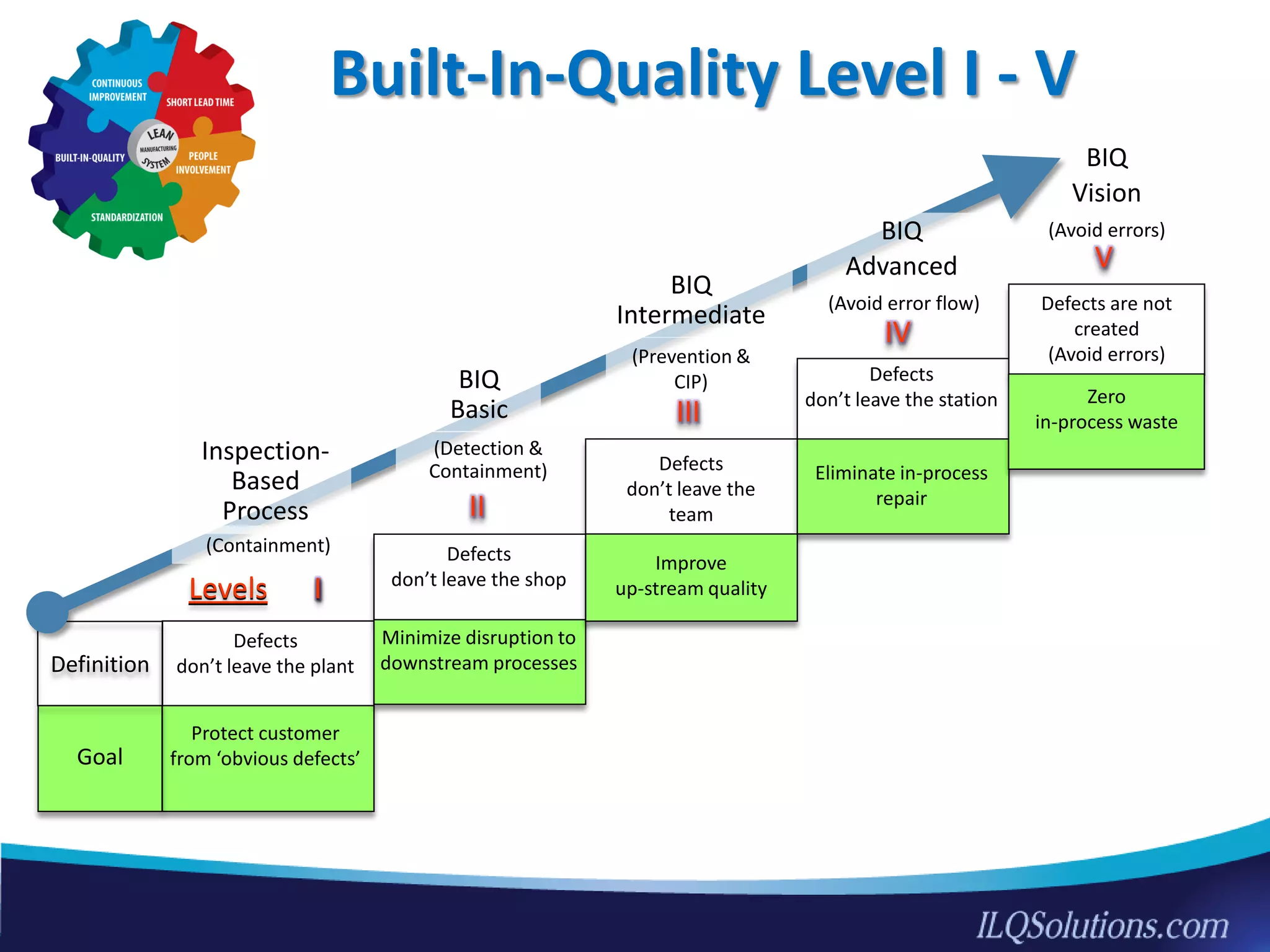
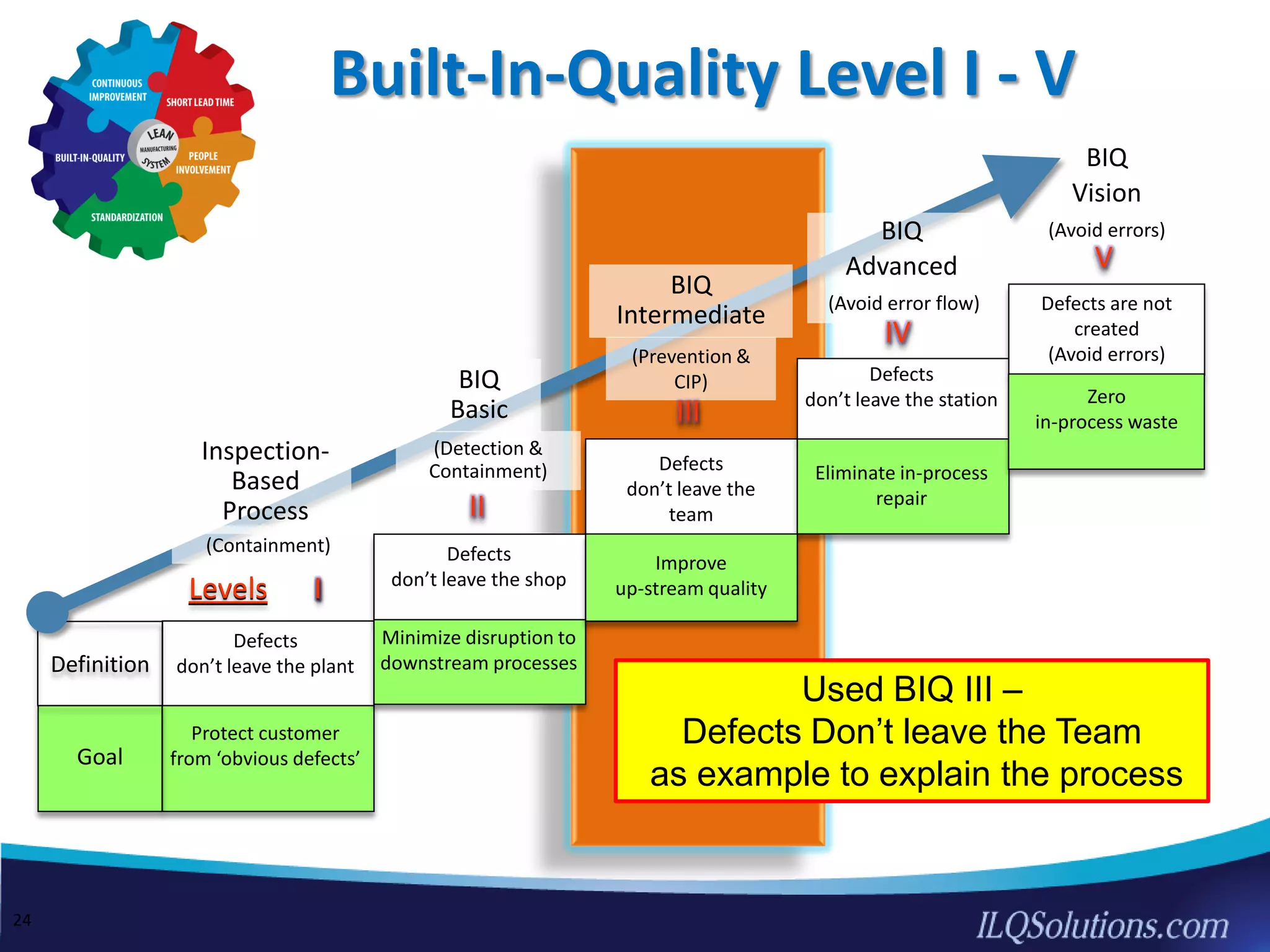
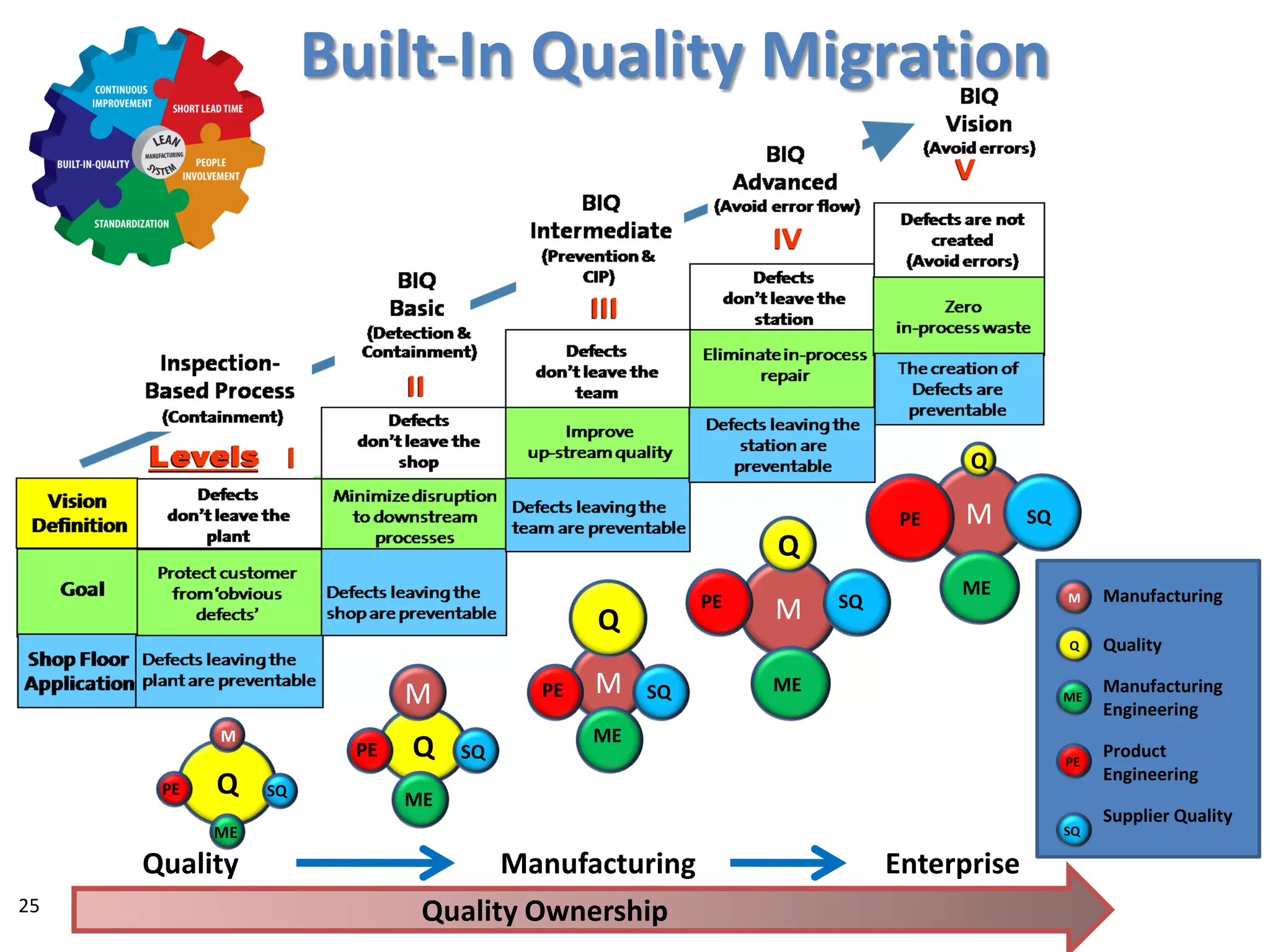
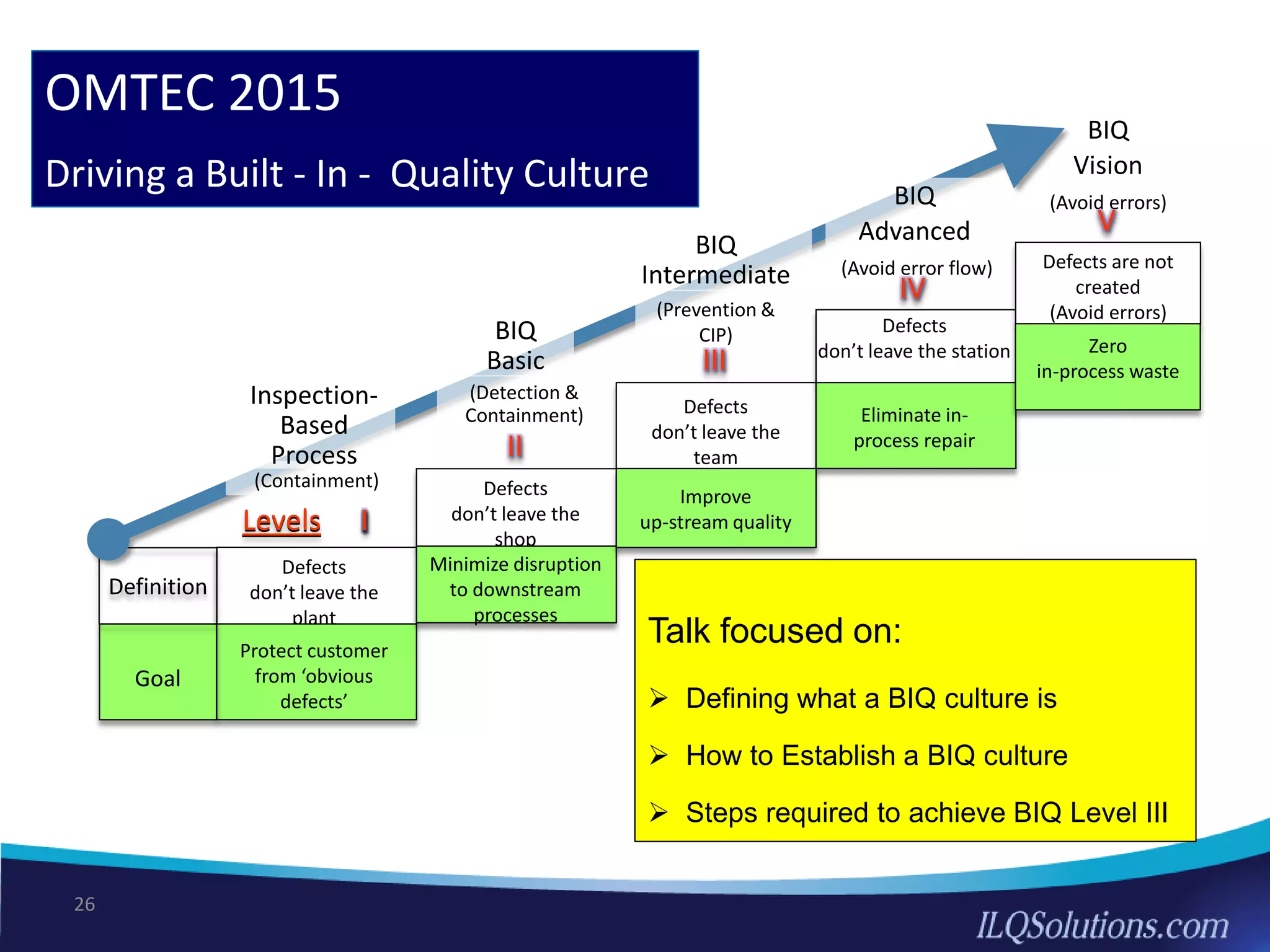
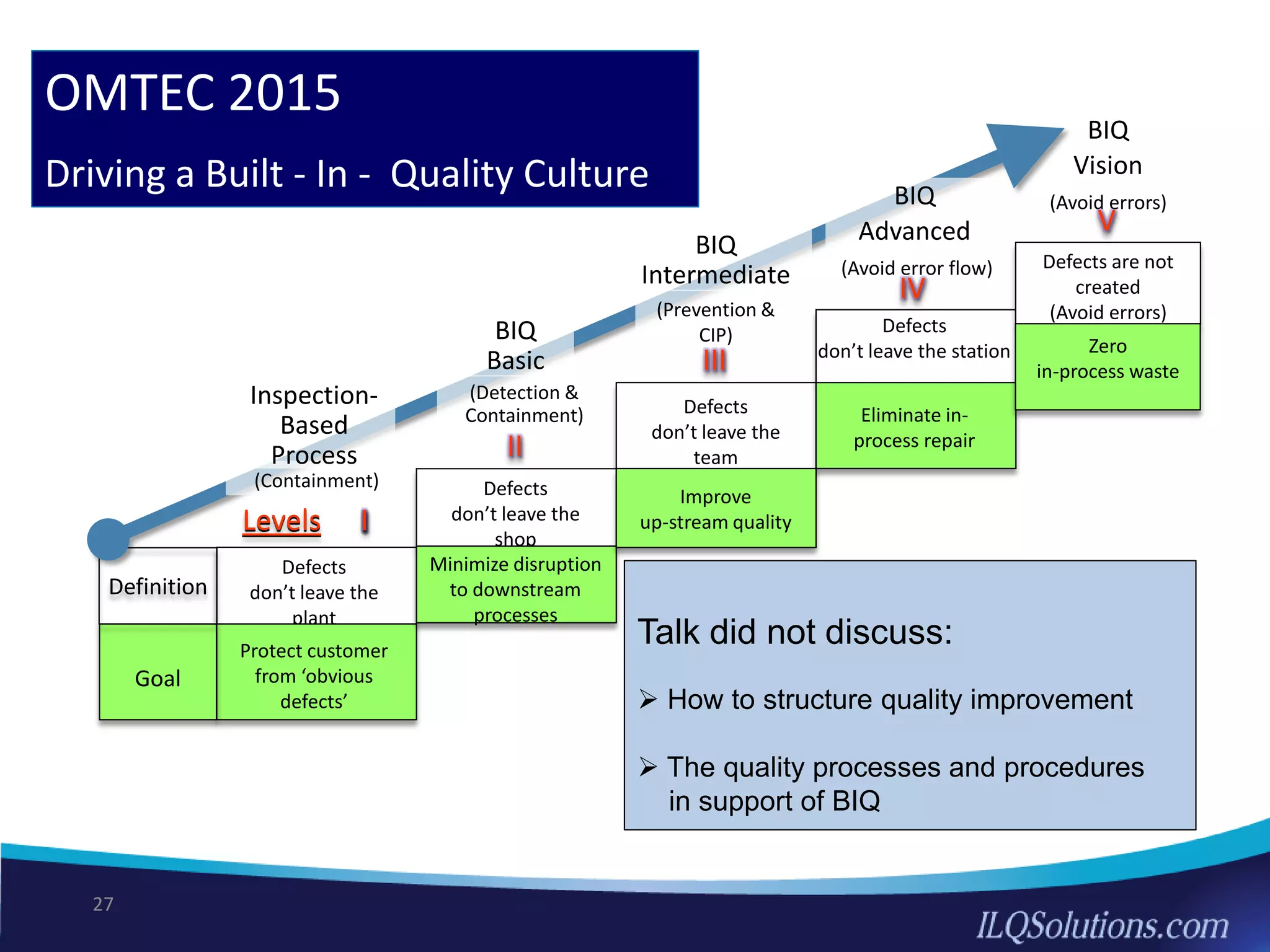
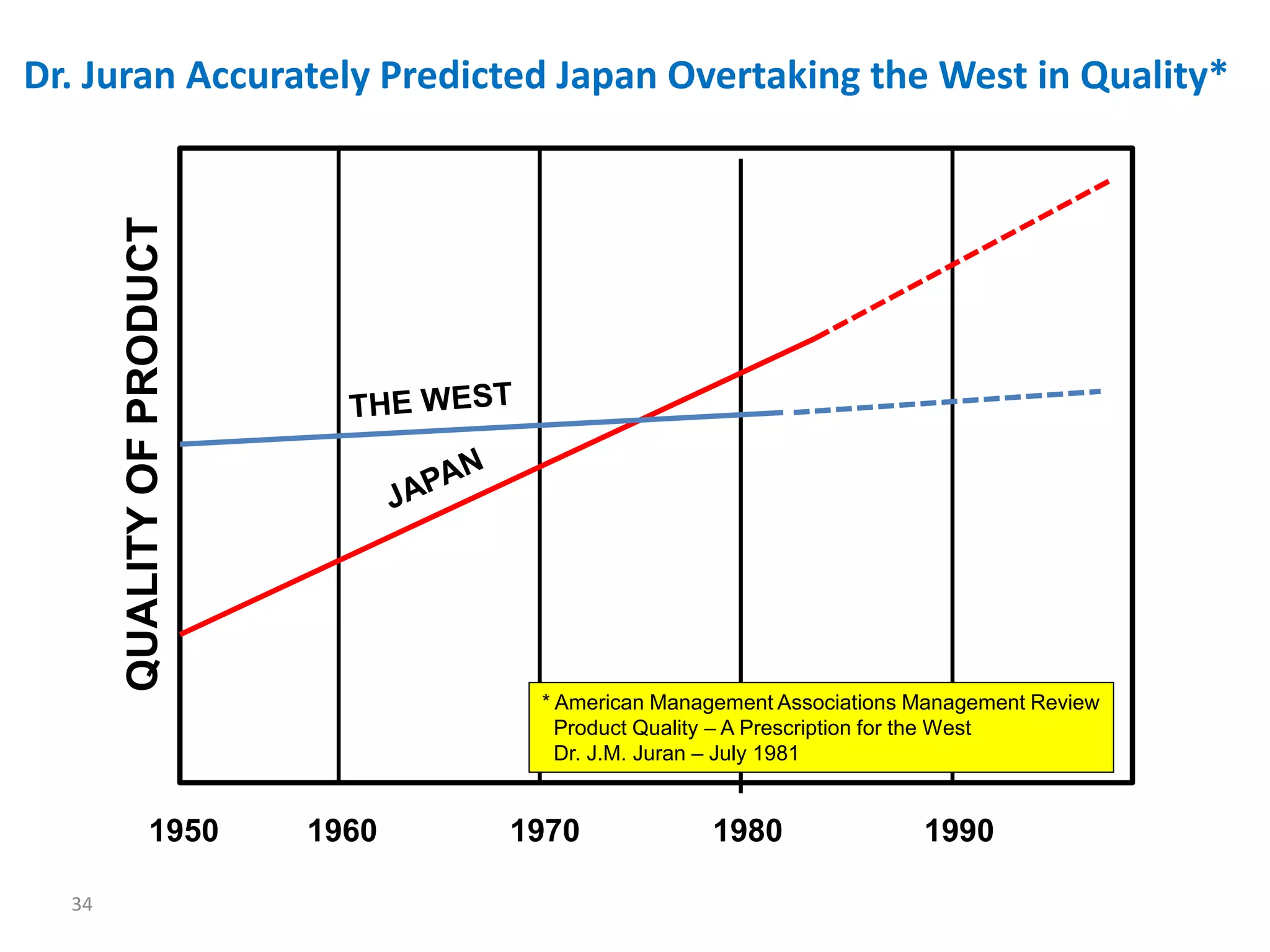
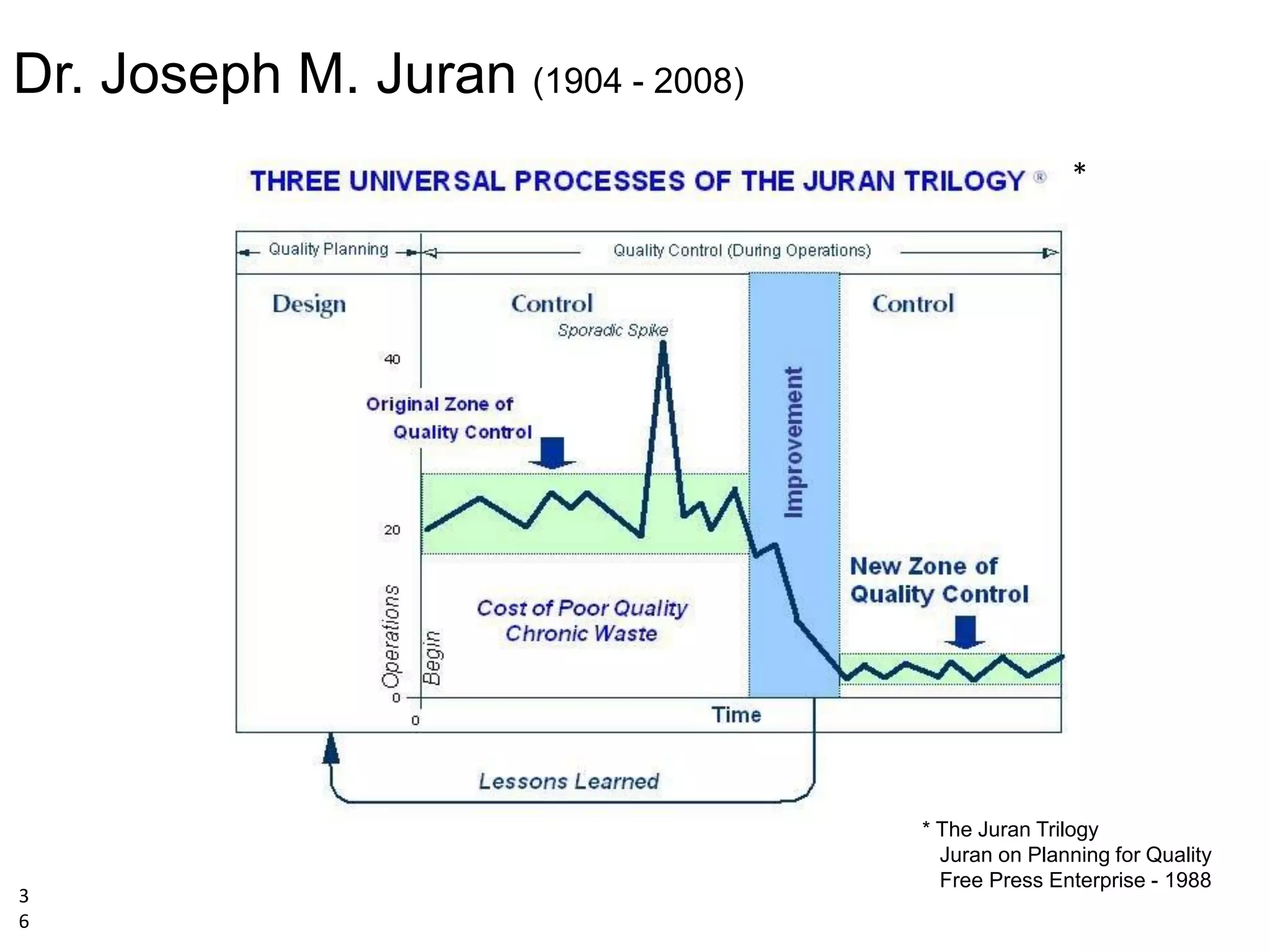
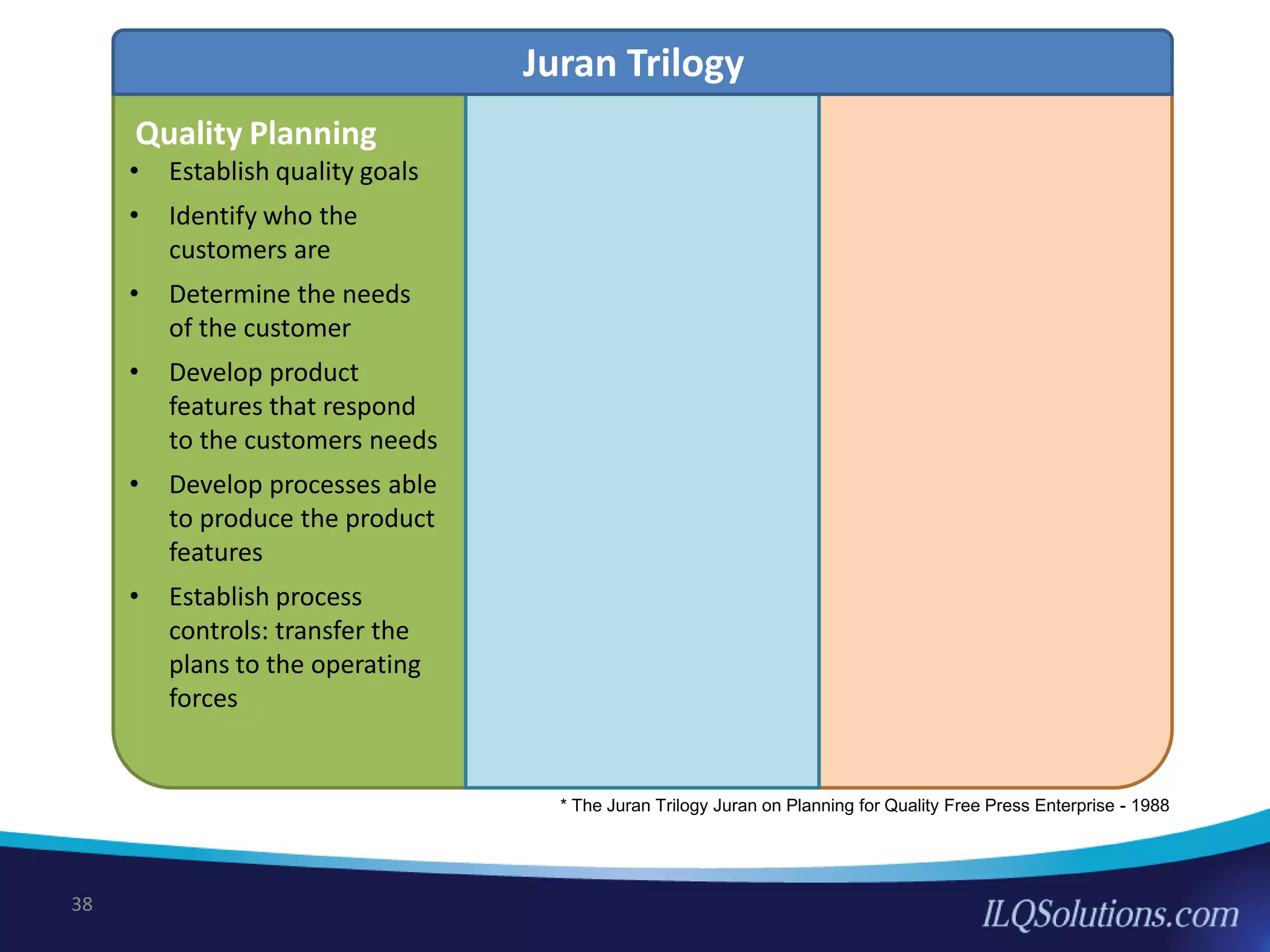
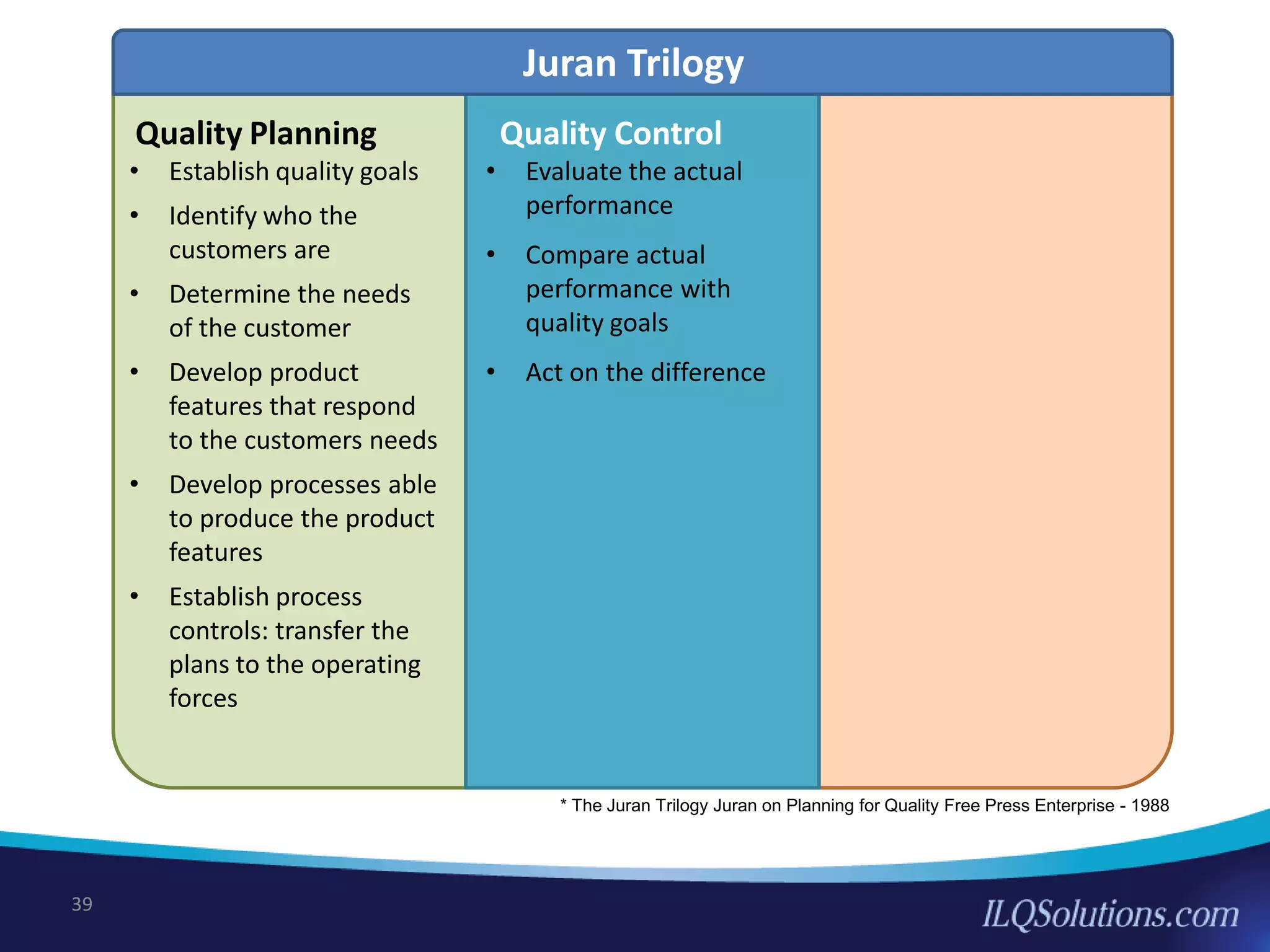
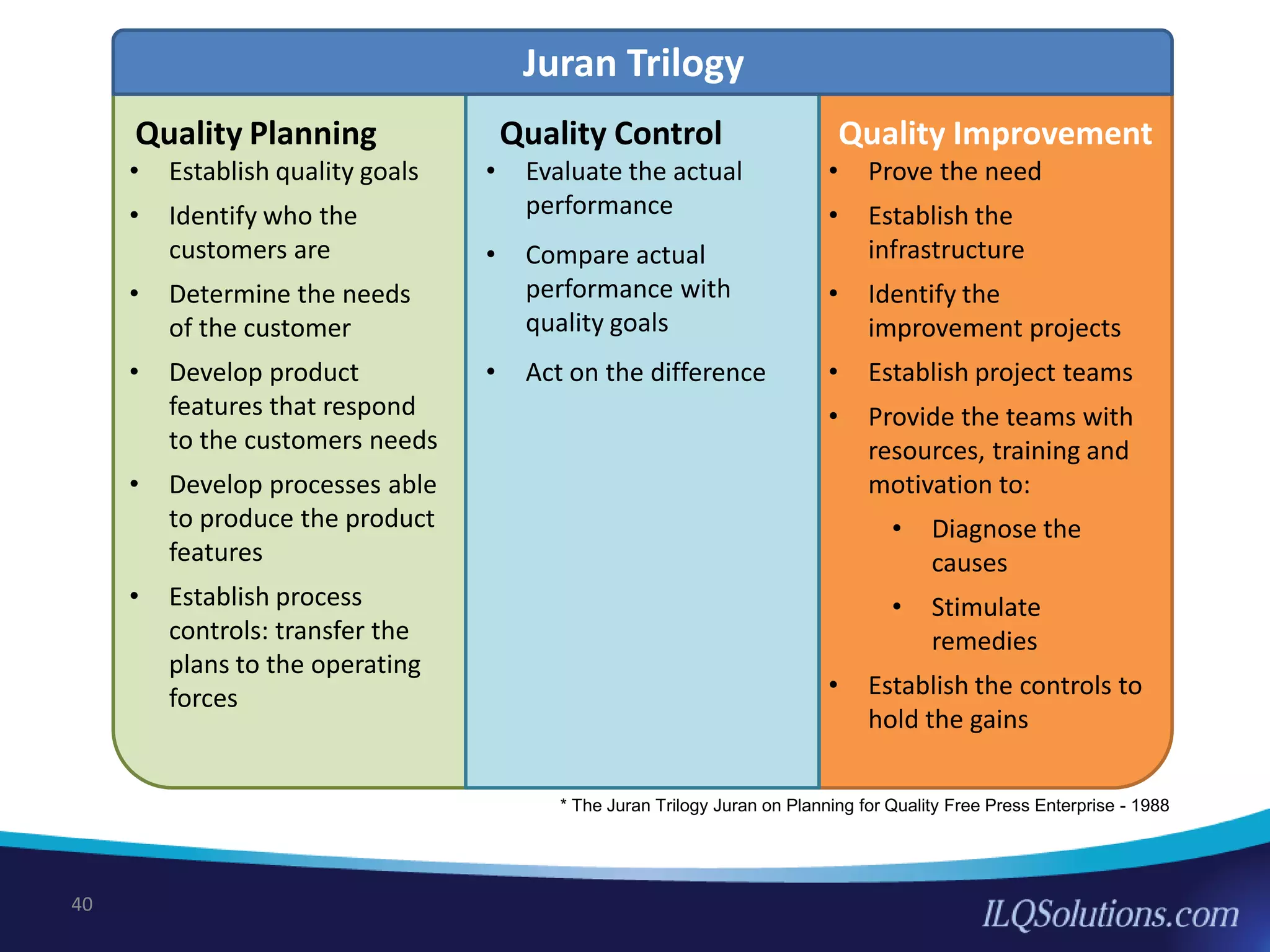
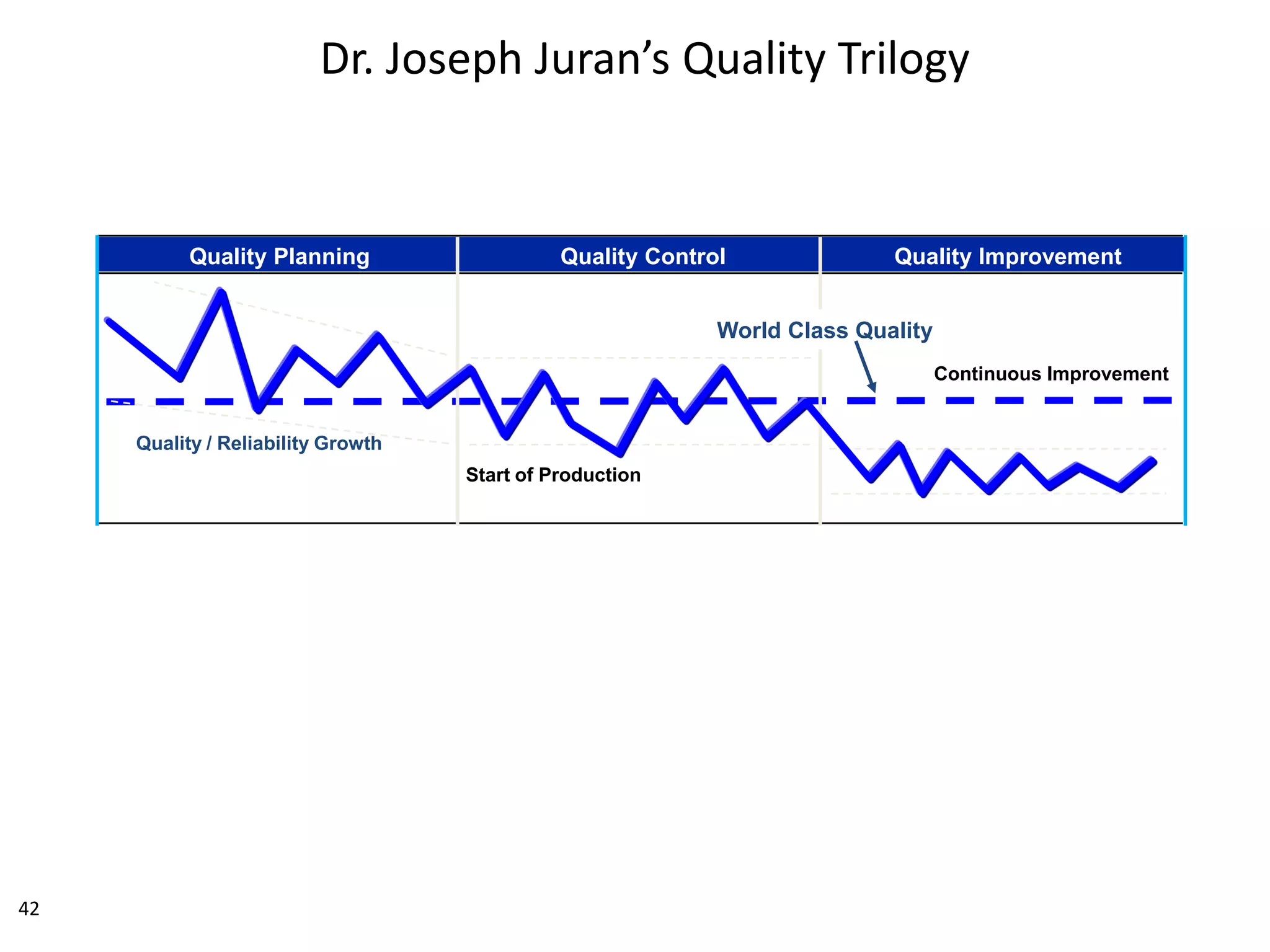
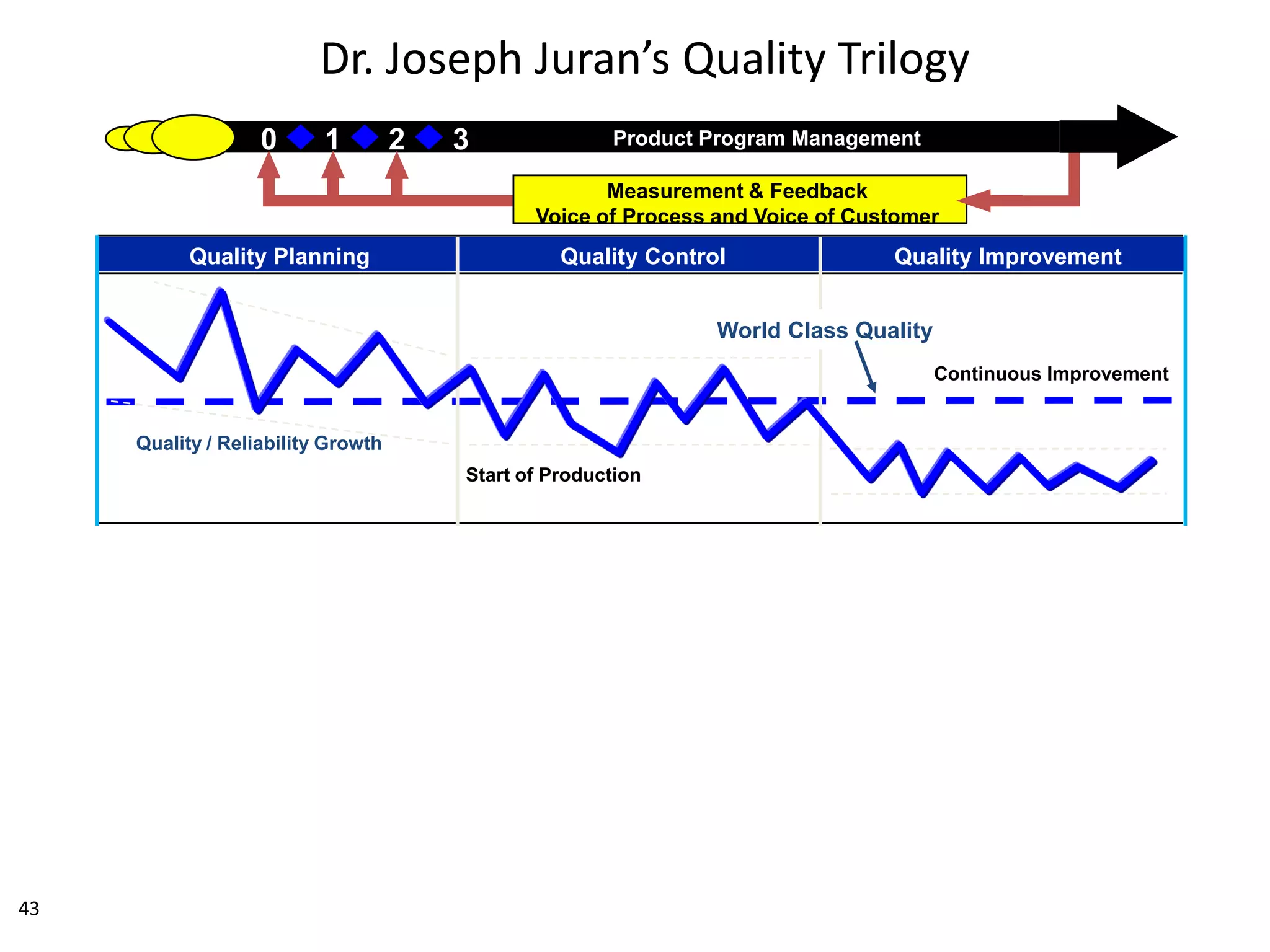
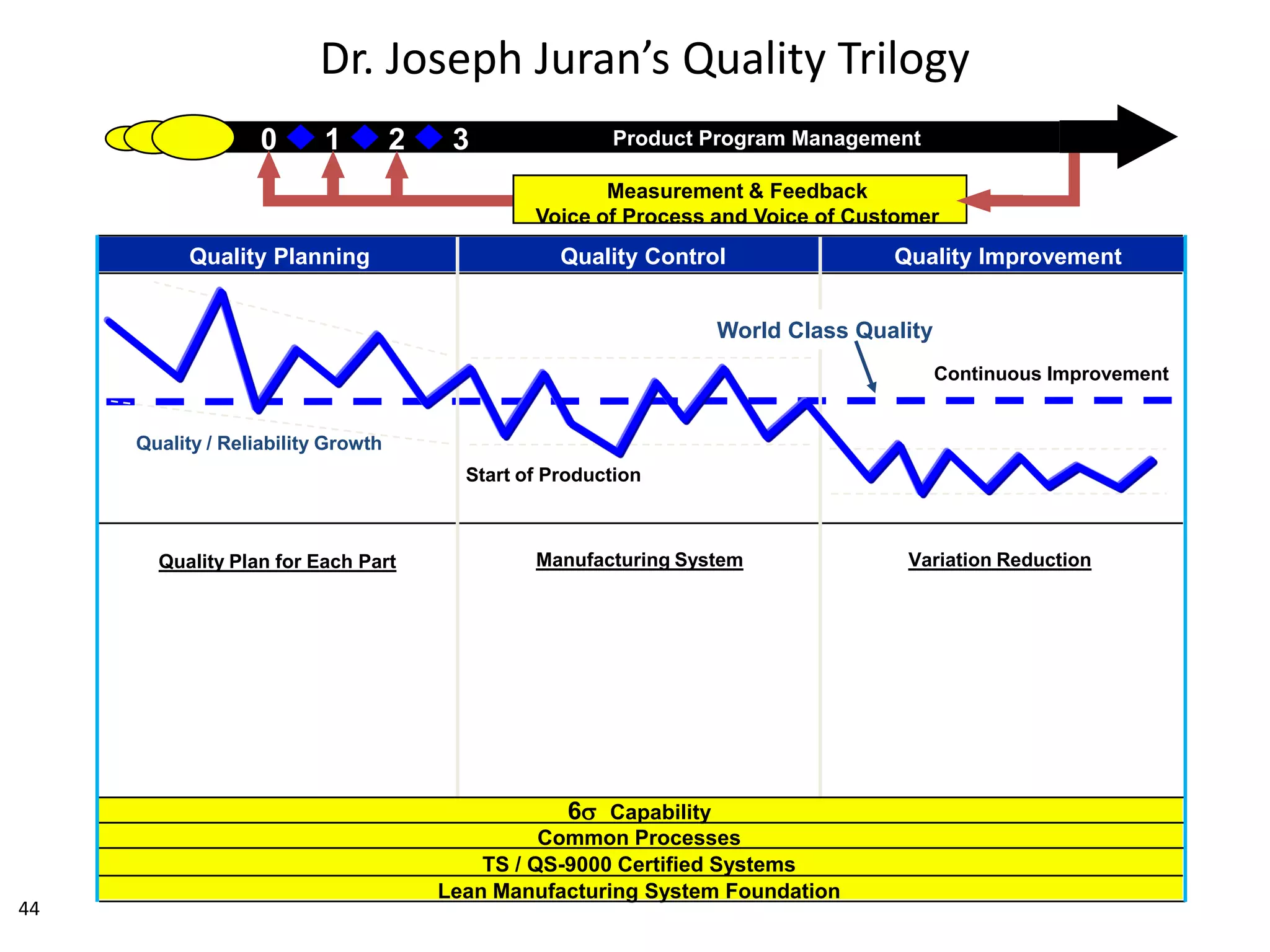
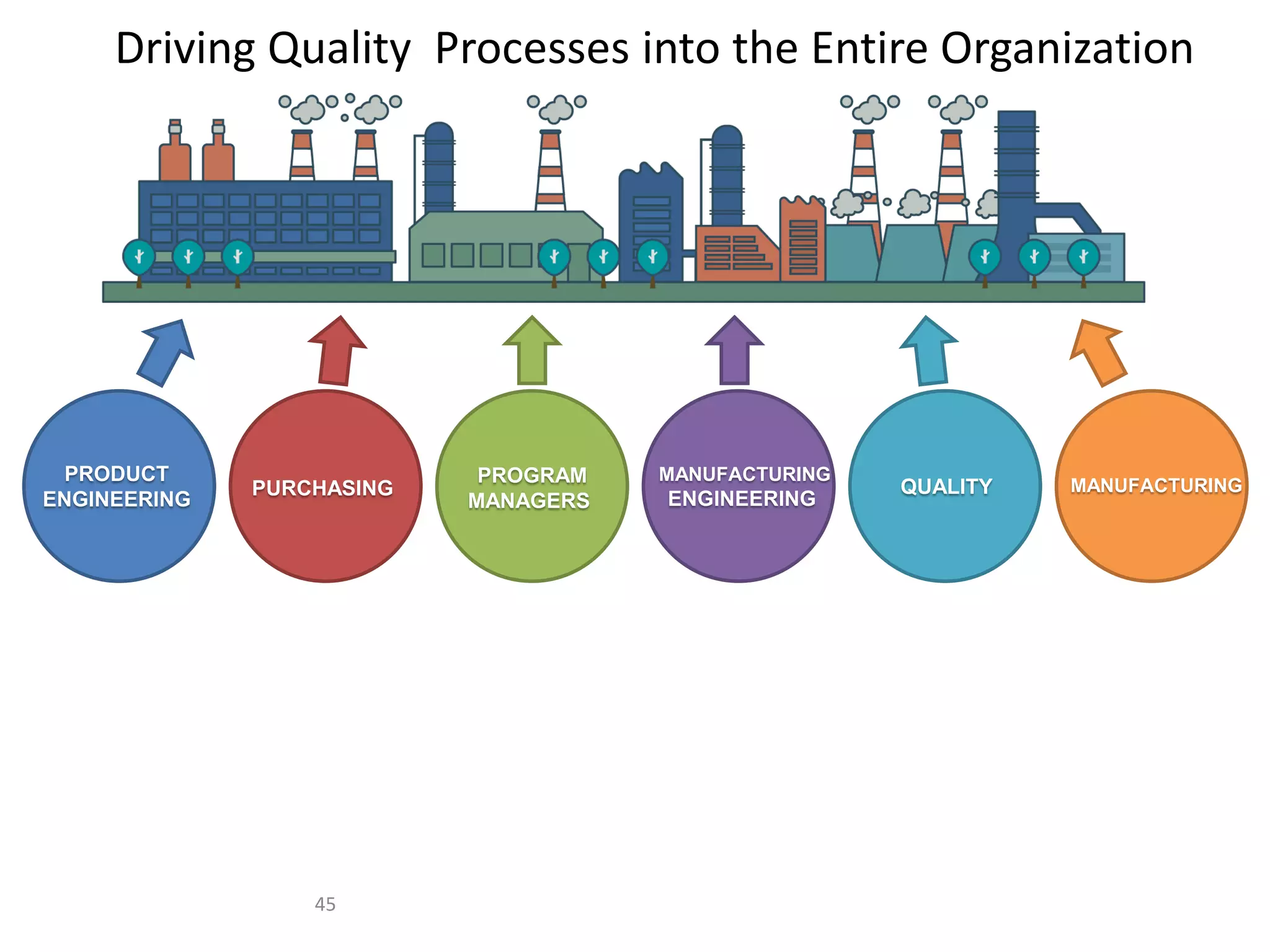
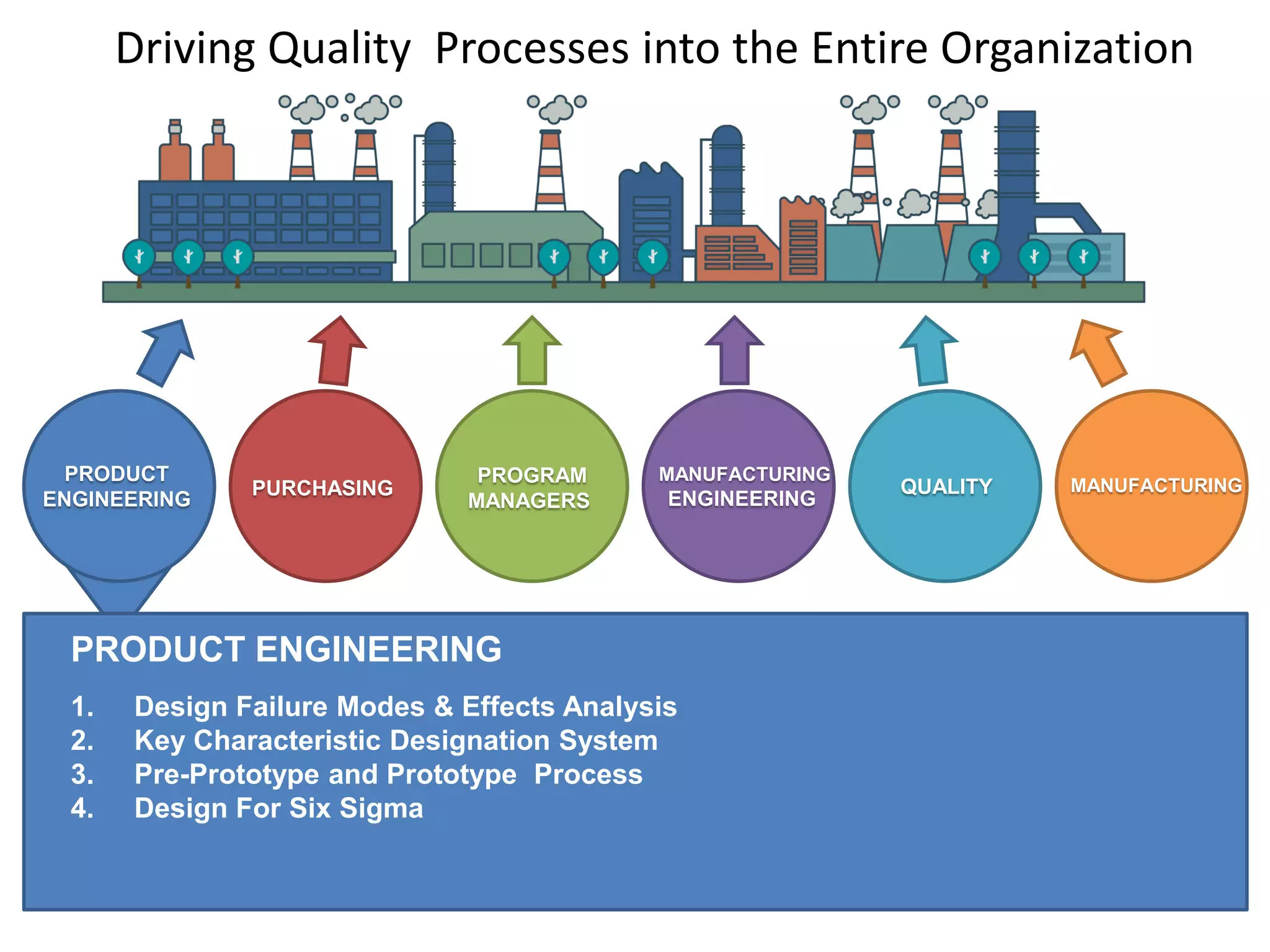
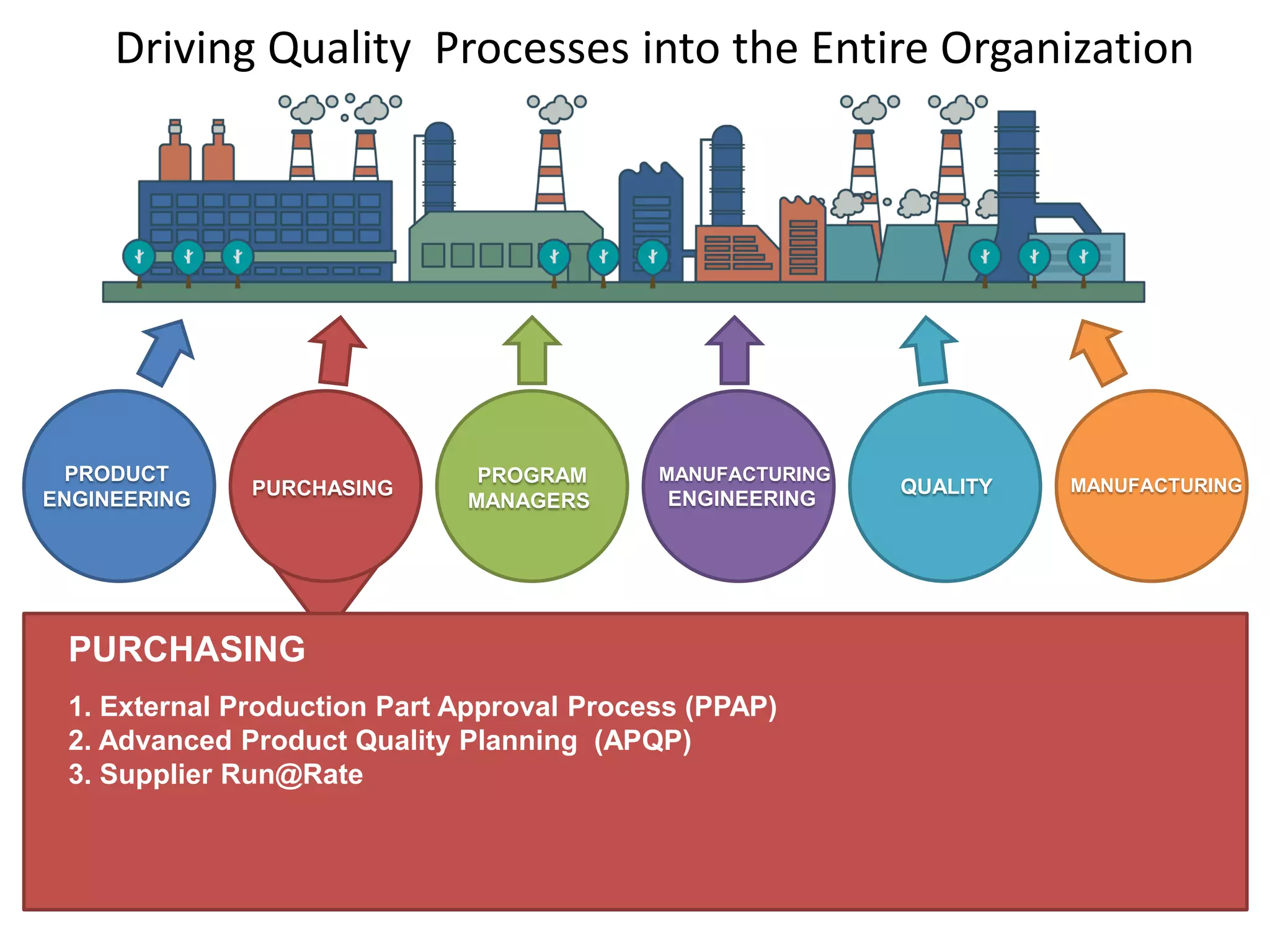
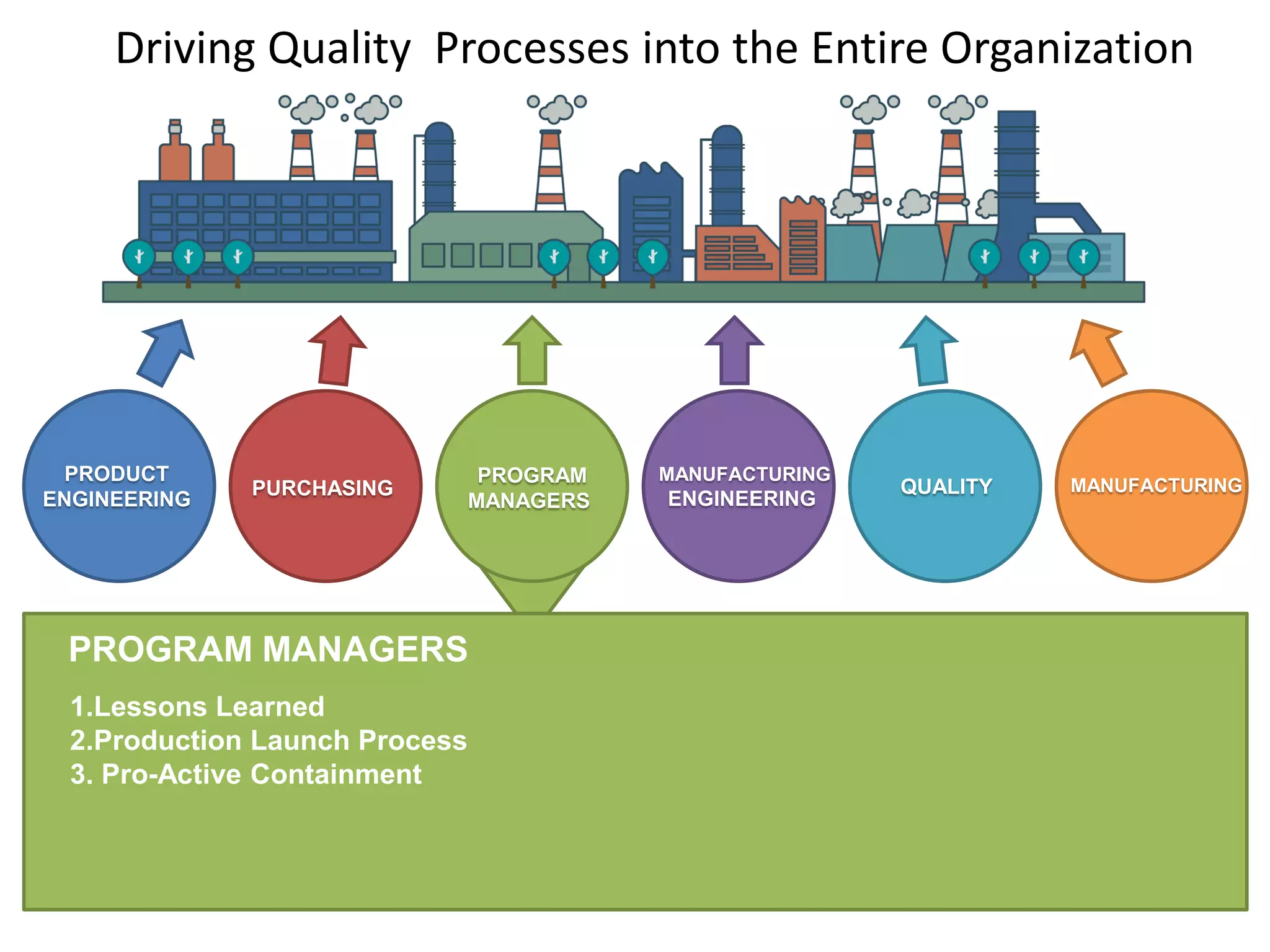
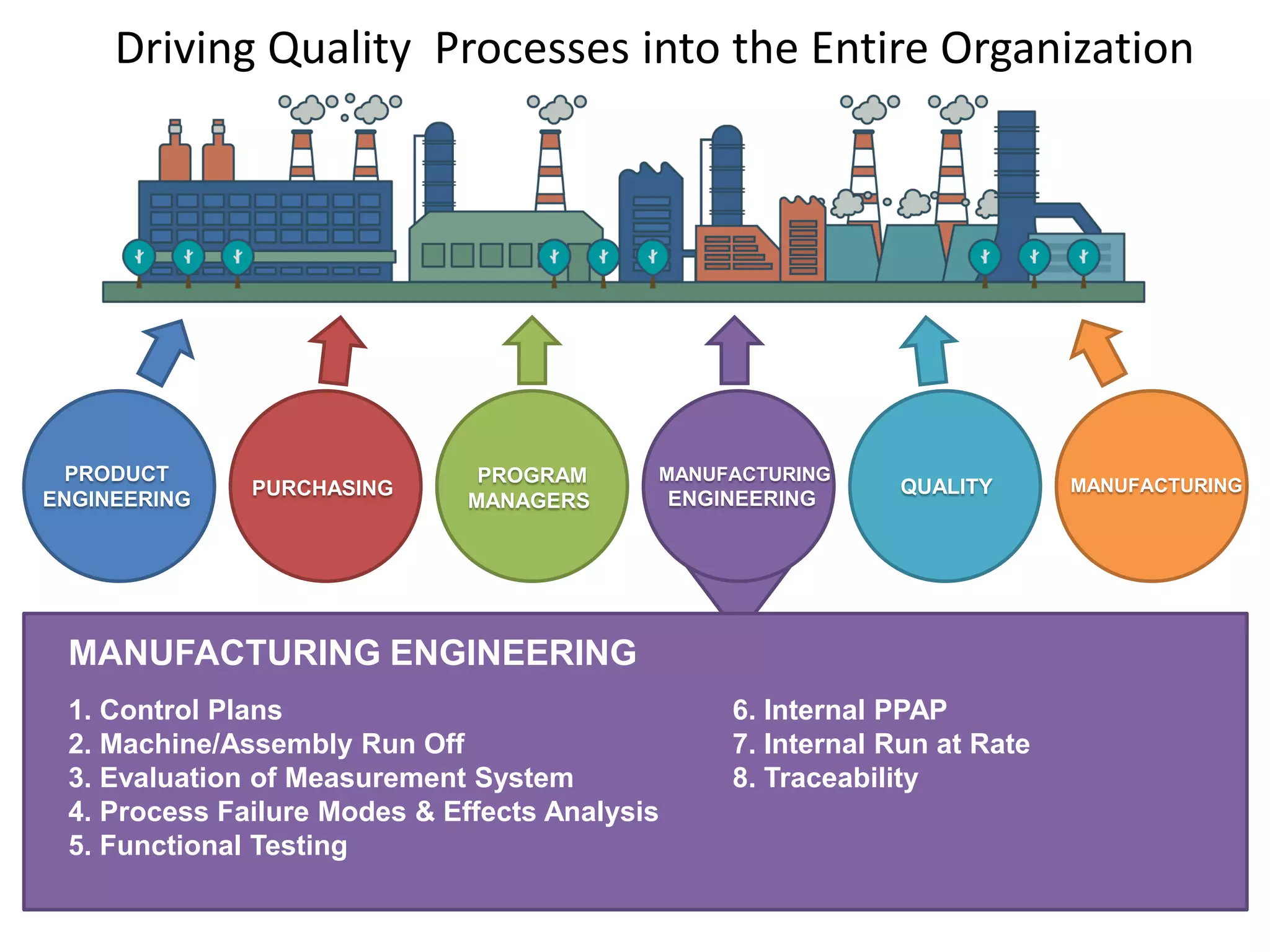
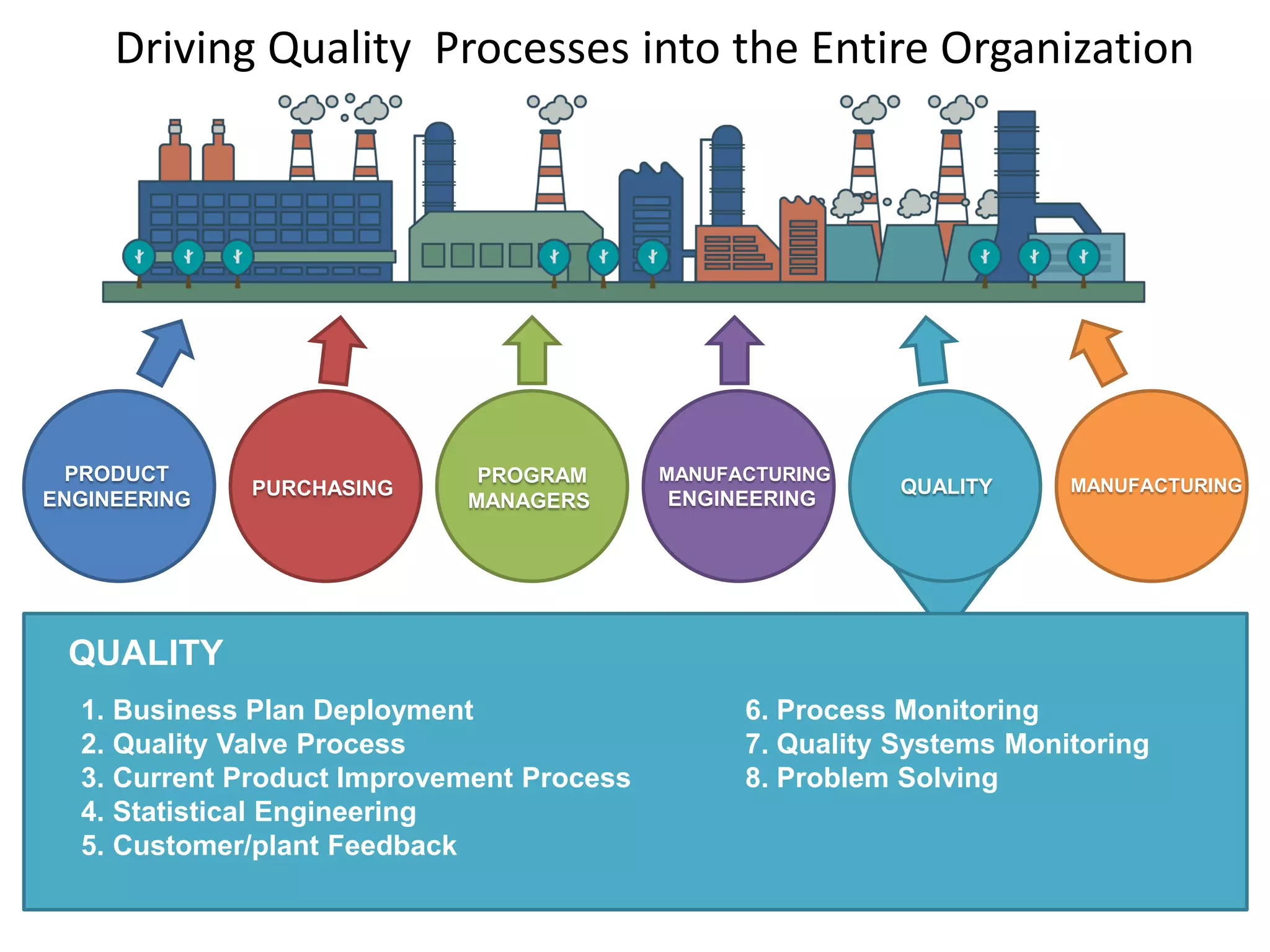
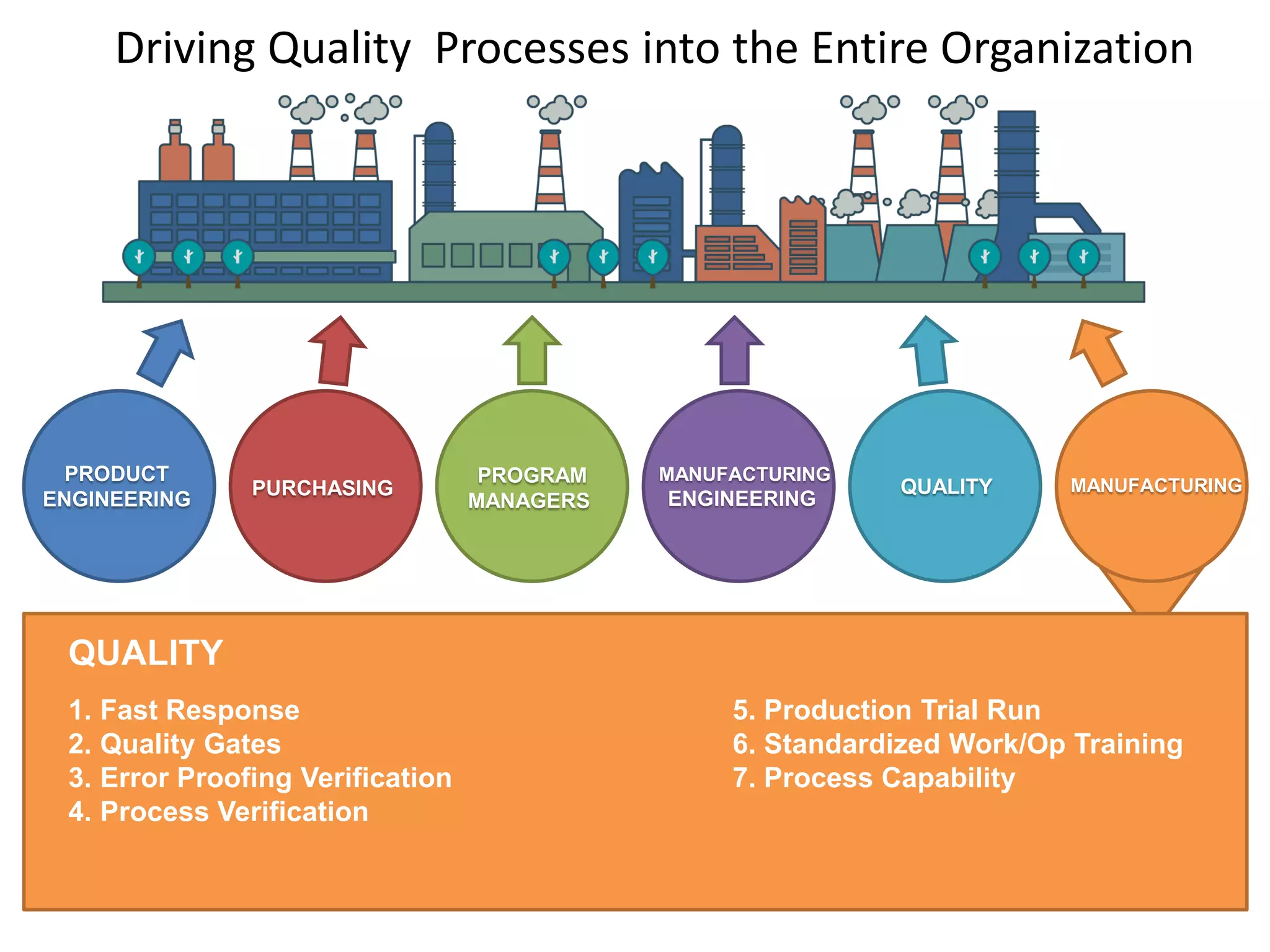
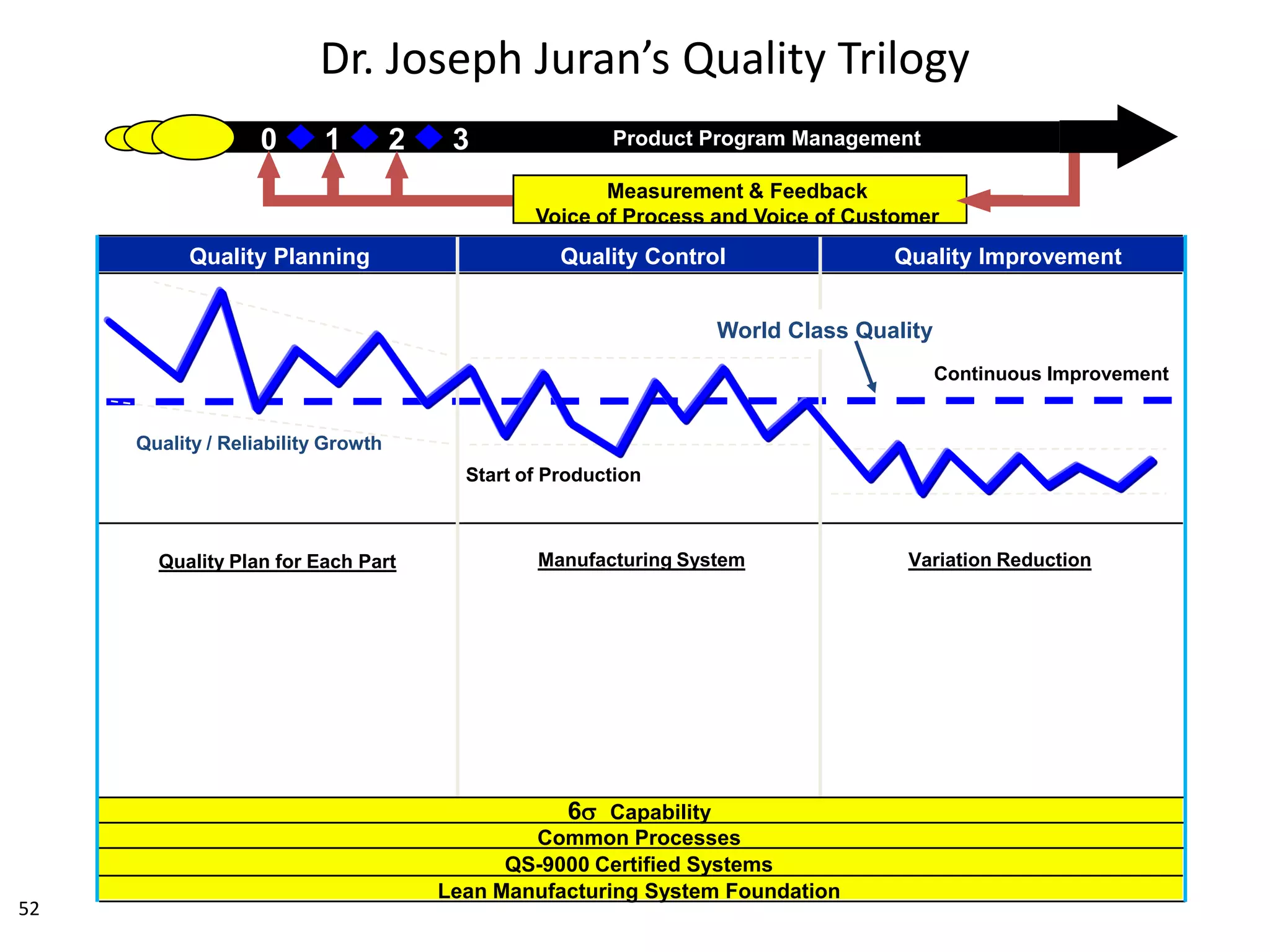
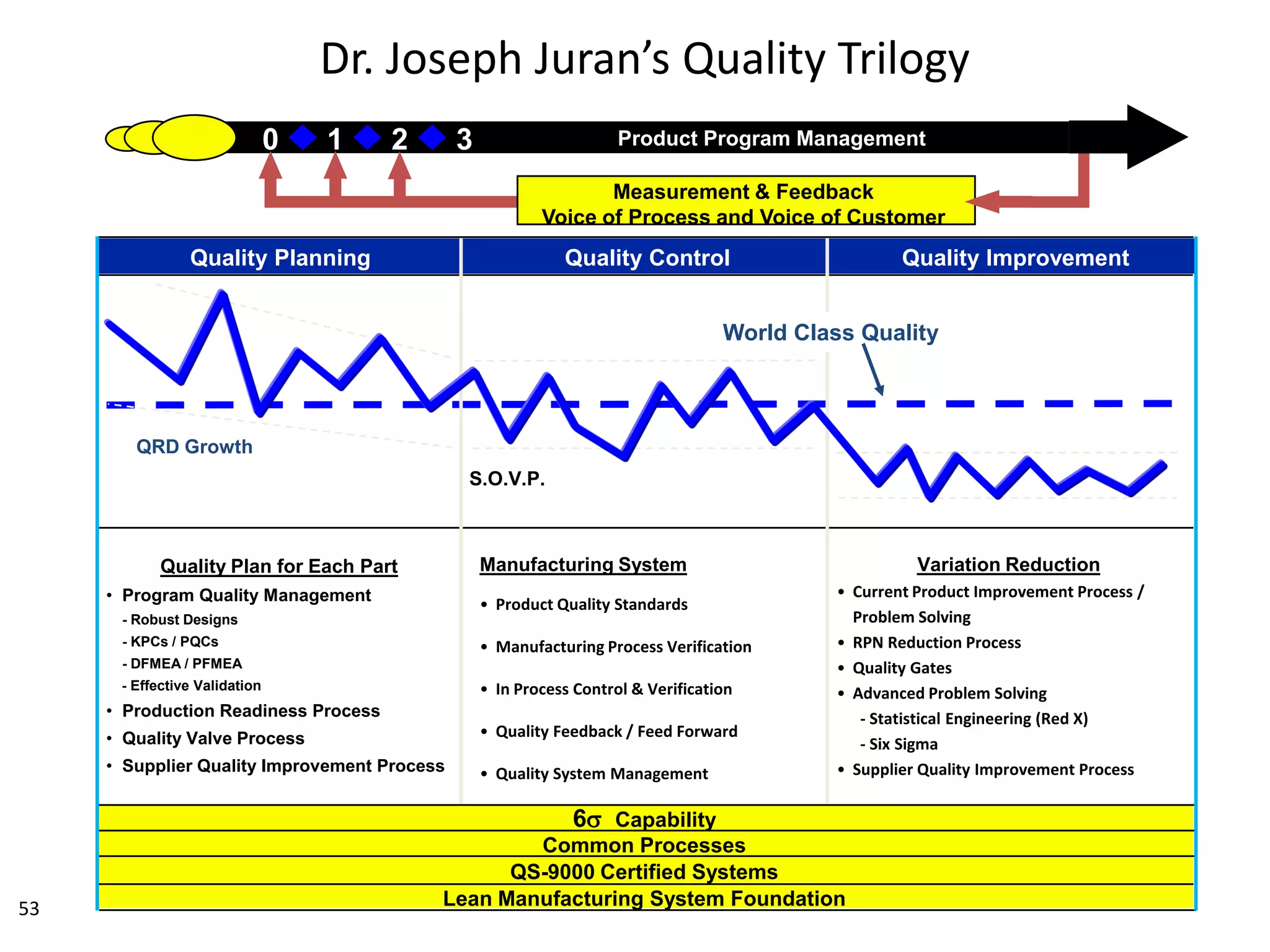
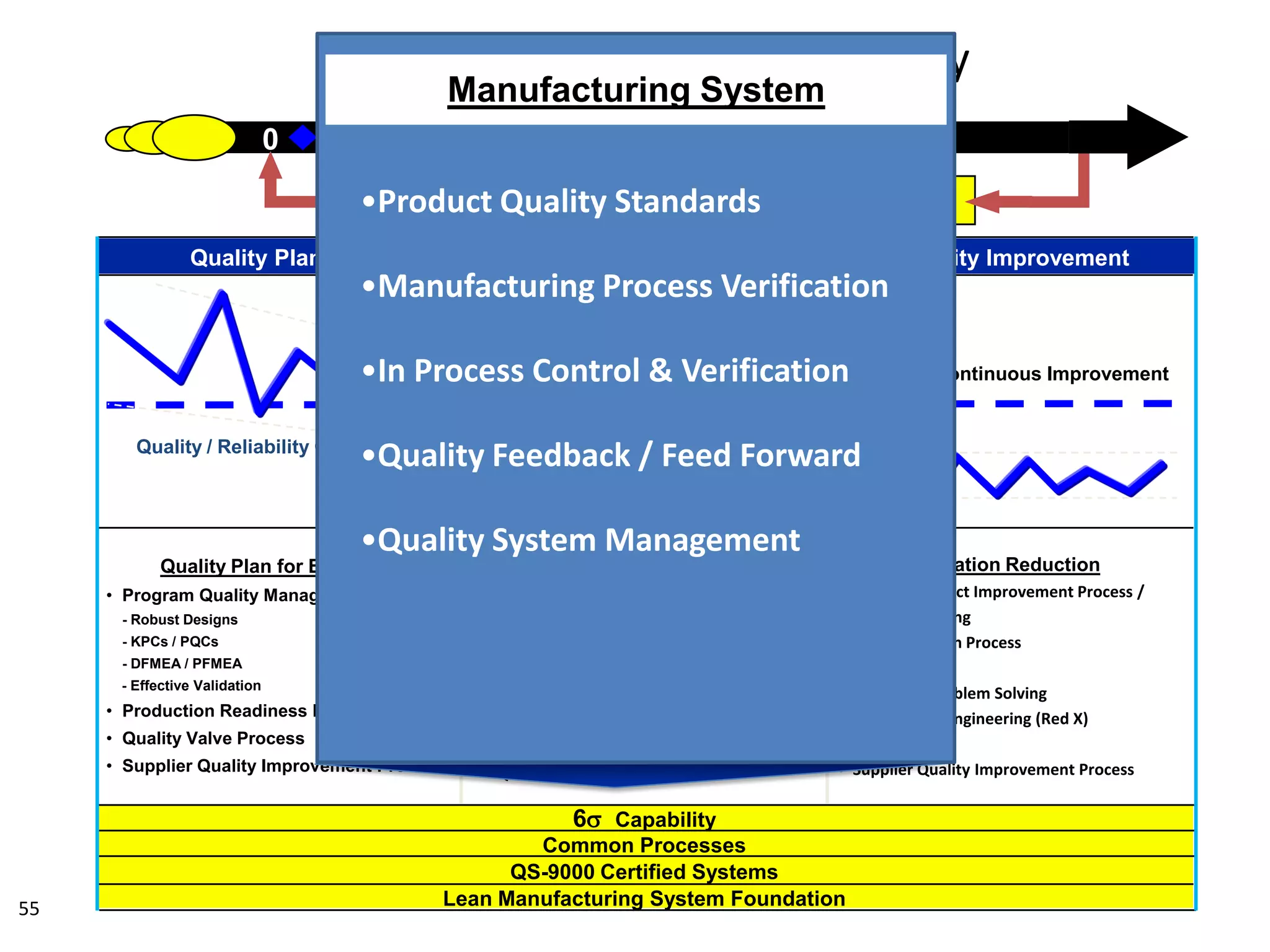
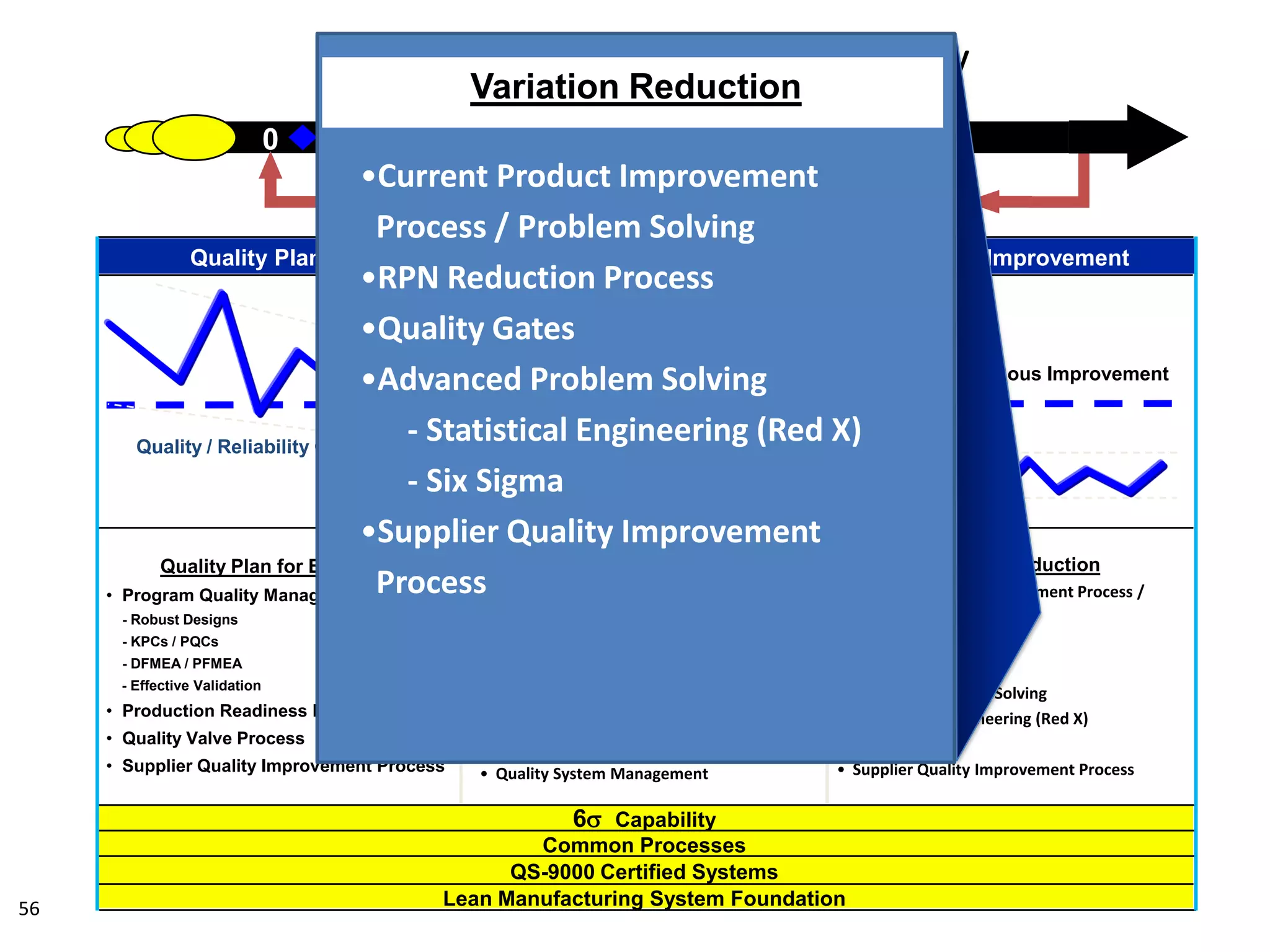
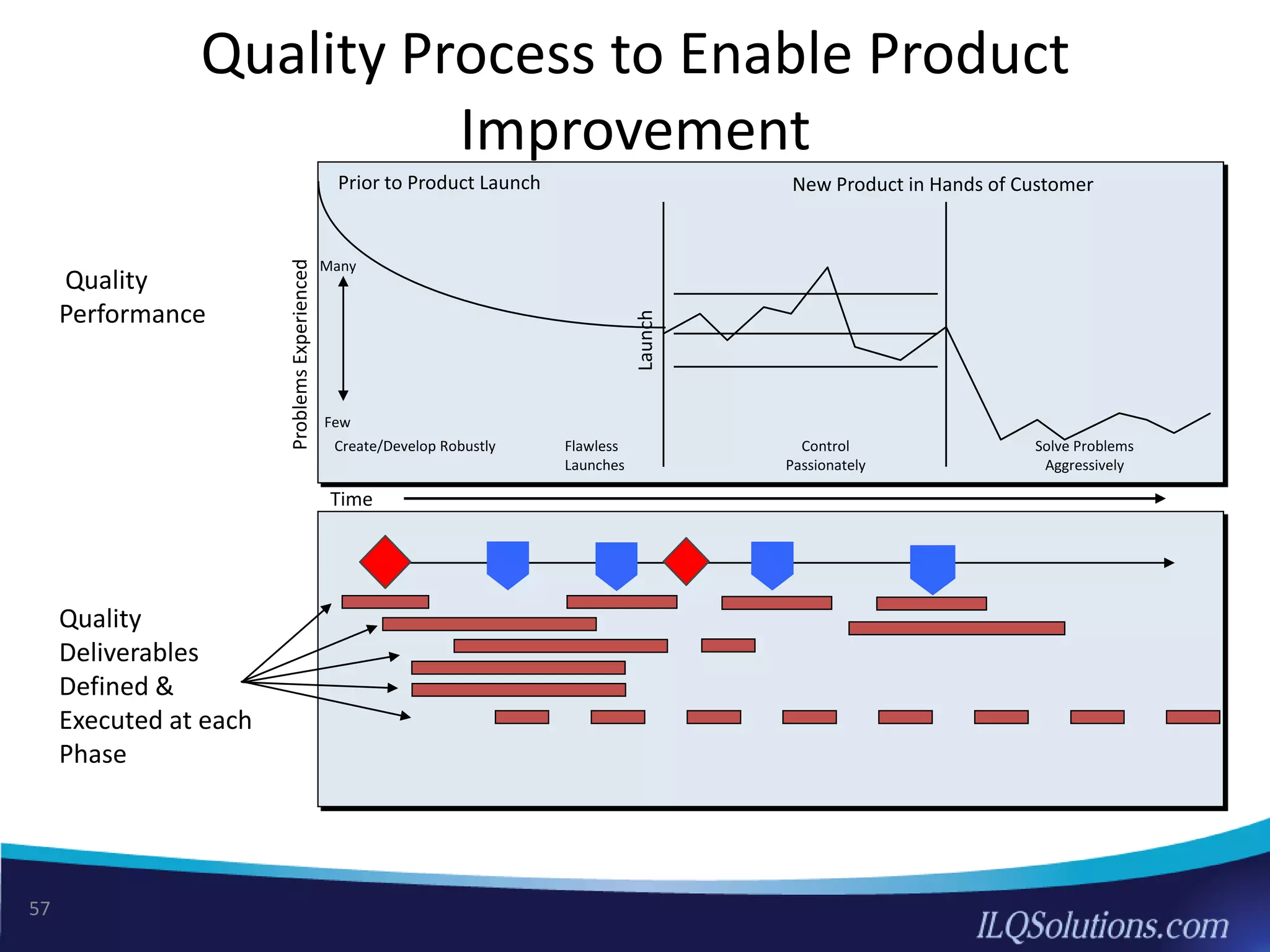
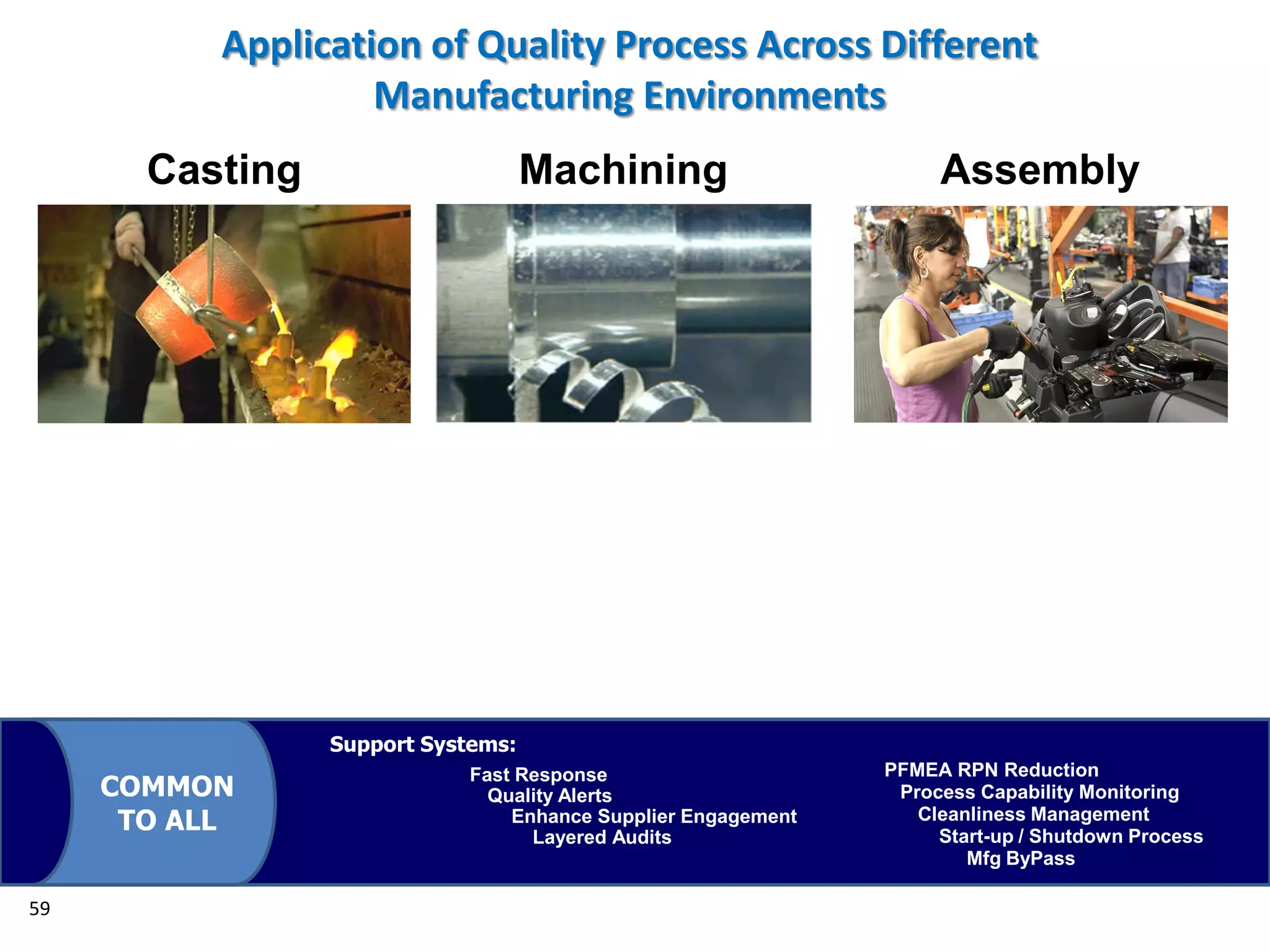
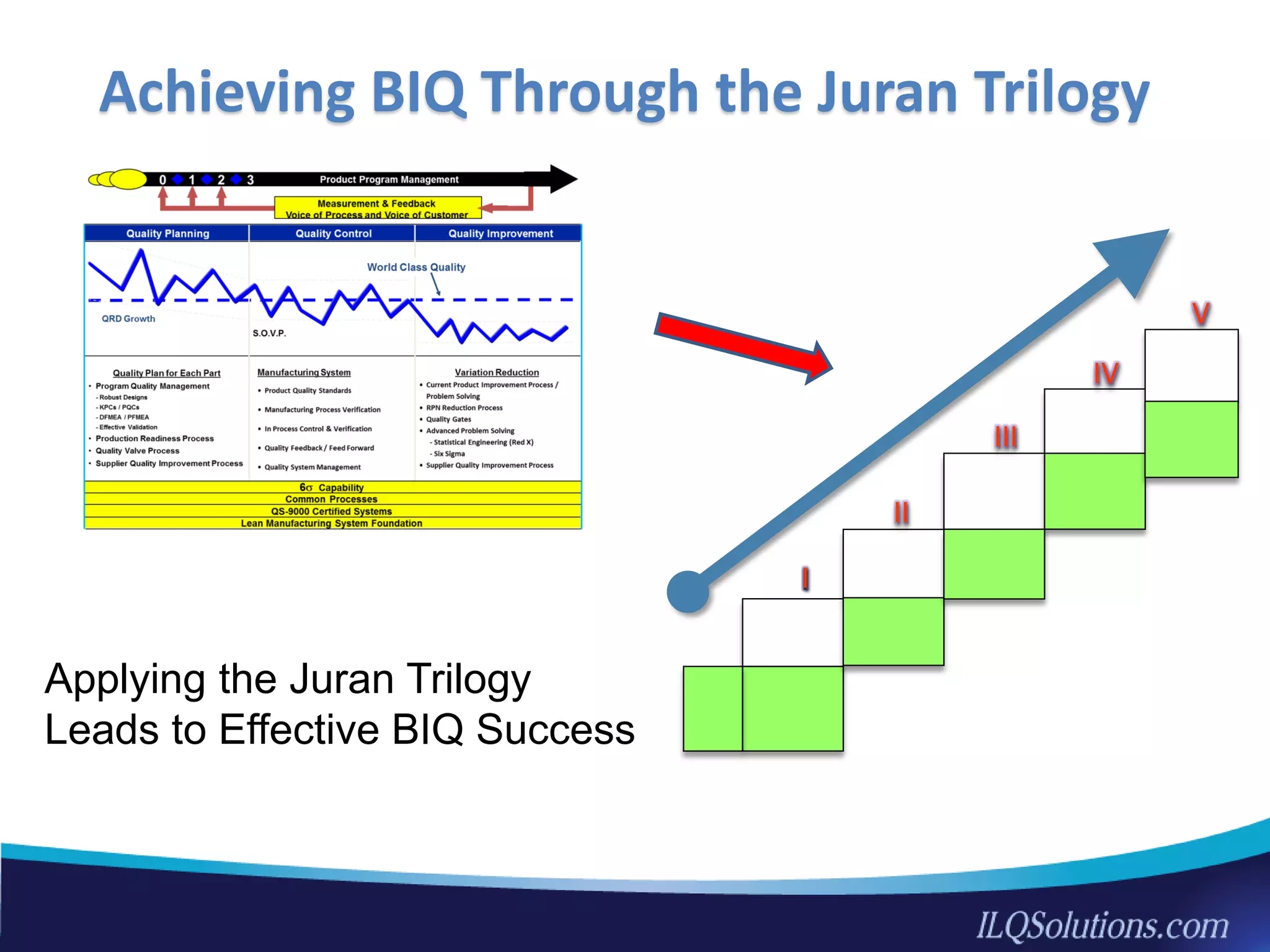
The document discusses achieving built-in quality (BIQ) through the Juran Trilogy, emphasizing its application within lean manufacturing principles. It defines BIQ and explores how to implement its concepts to improve processes, eliminate waste, and enhance product quality. The document highlights leadership roles and provides frameworks for organizations of various sizes to adopt BIQ practices successfully.