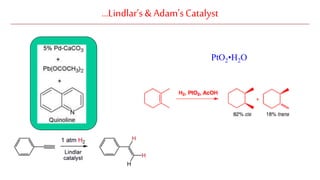
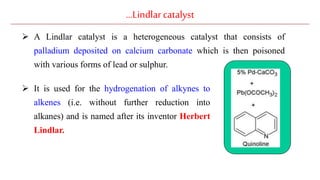
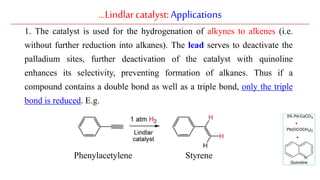
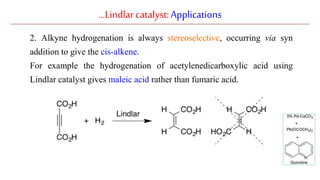
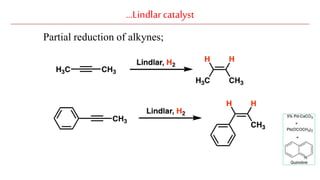
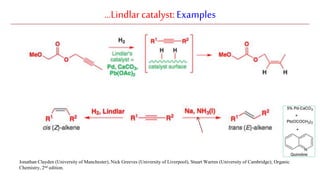
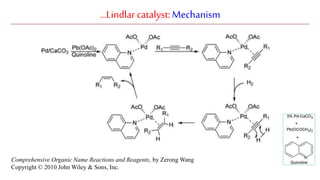
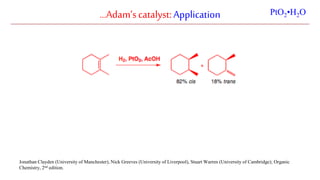
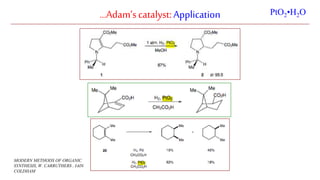
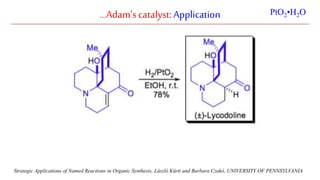
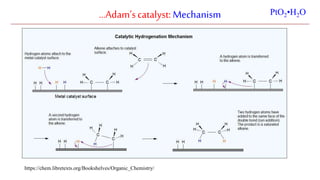
The document discusses Lindlar's catalyst and Adam's catalyst. Lindlar's catalyst consists of palladium deposited on calcium carbonate and poisoned with lead or sulfur. It is used to selectively hydrogenate alkynes to alkenes without full reduction to alkanes. Adam's catalyst, also known as platinum dioxide PtO2•H2O, is used for hydrogenation, hydrogenolysis, dehydrogenation, and oxidation reactions. It hydrogenates alkenes with syn stereochemistry and is commonly used to reduce ketones to alcohols and nitro compounds to amines. Both catalysts provide selective hydrogenation under mild conditions.