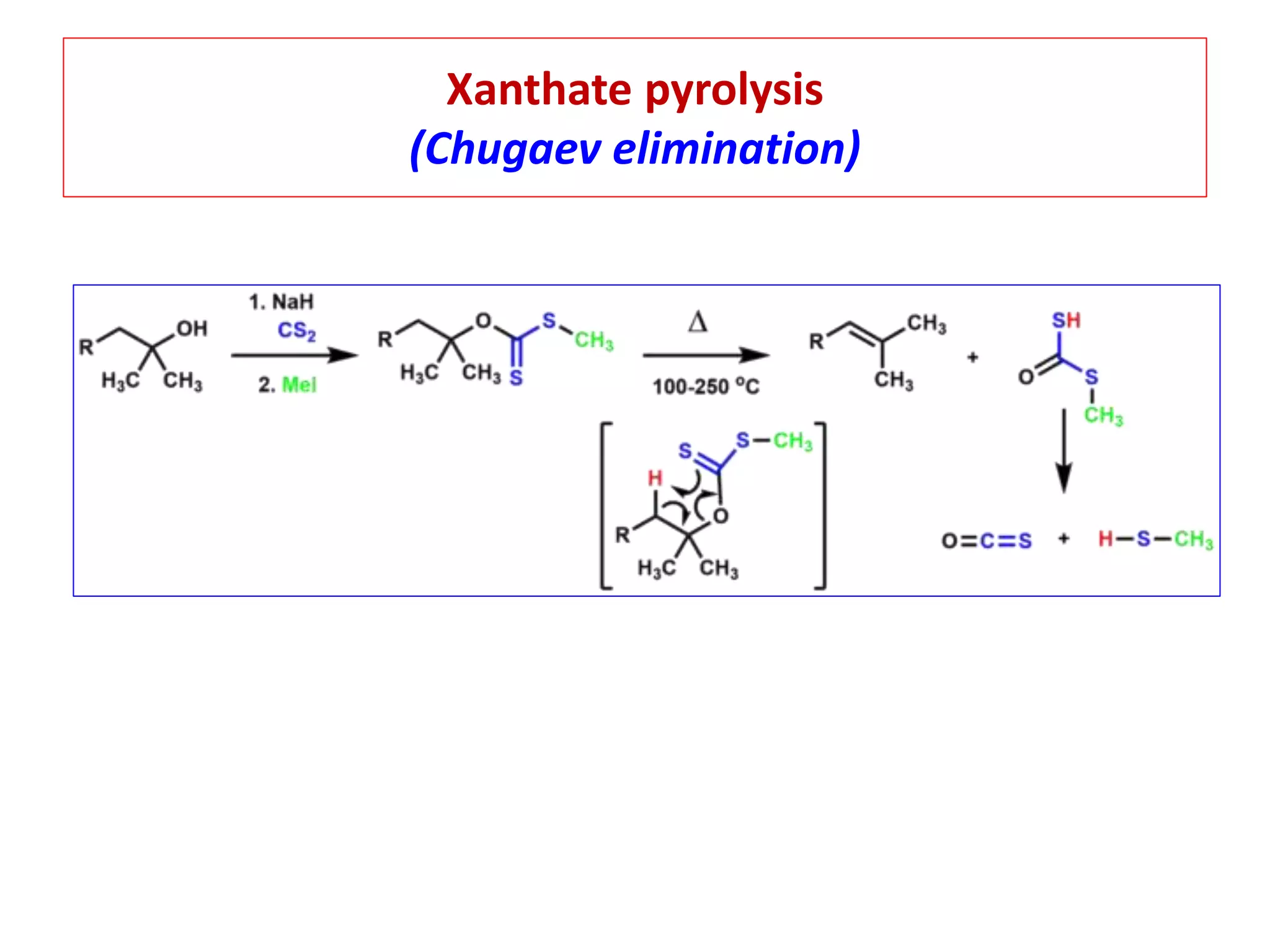
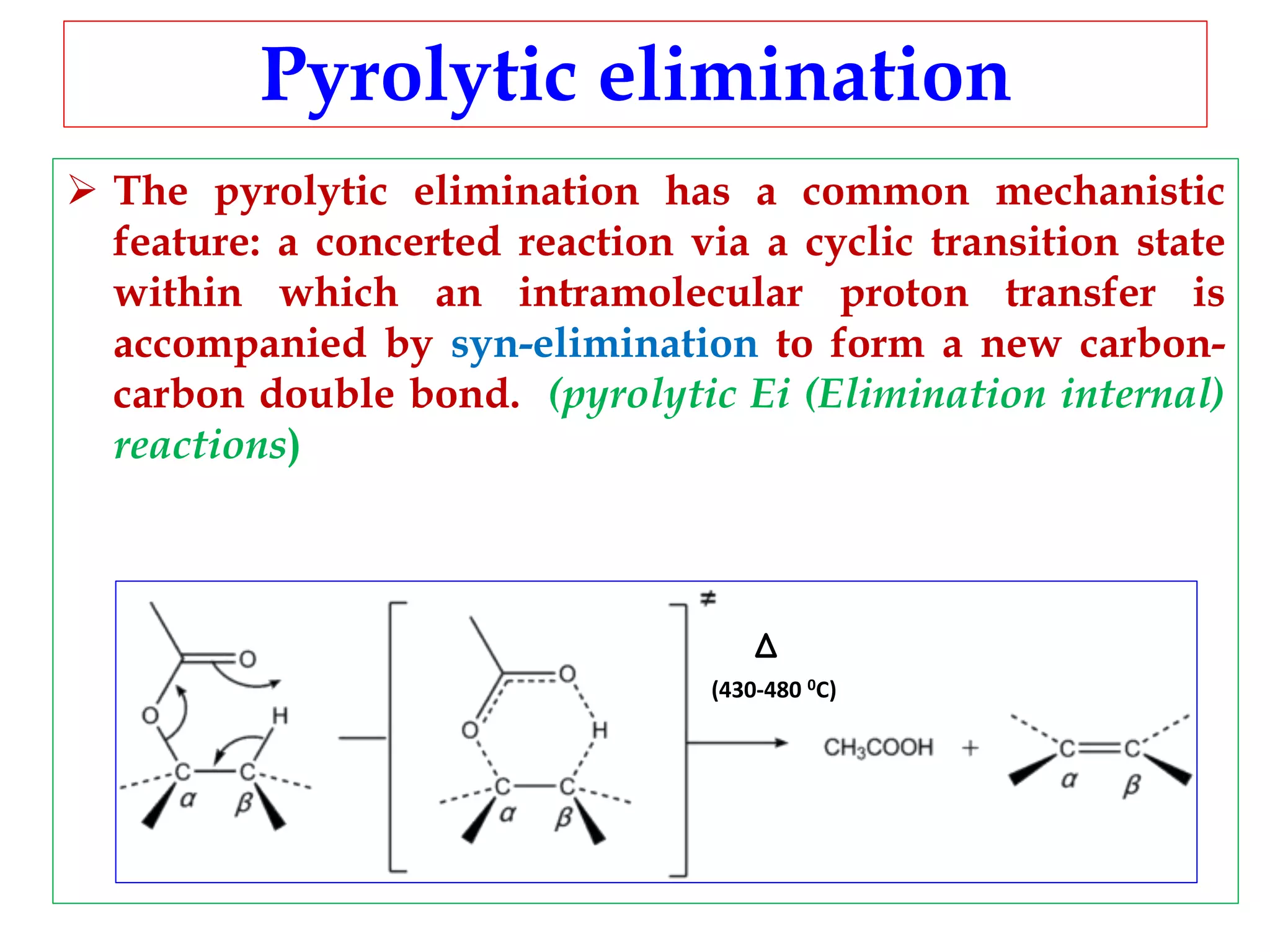
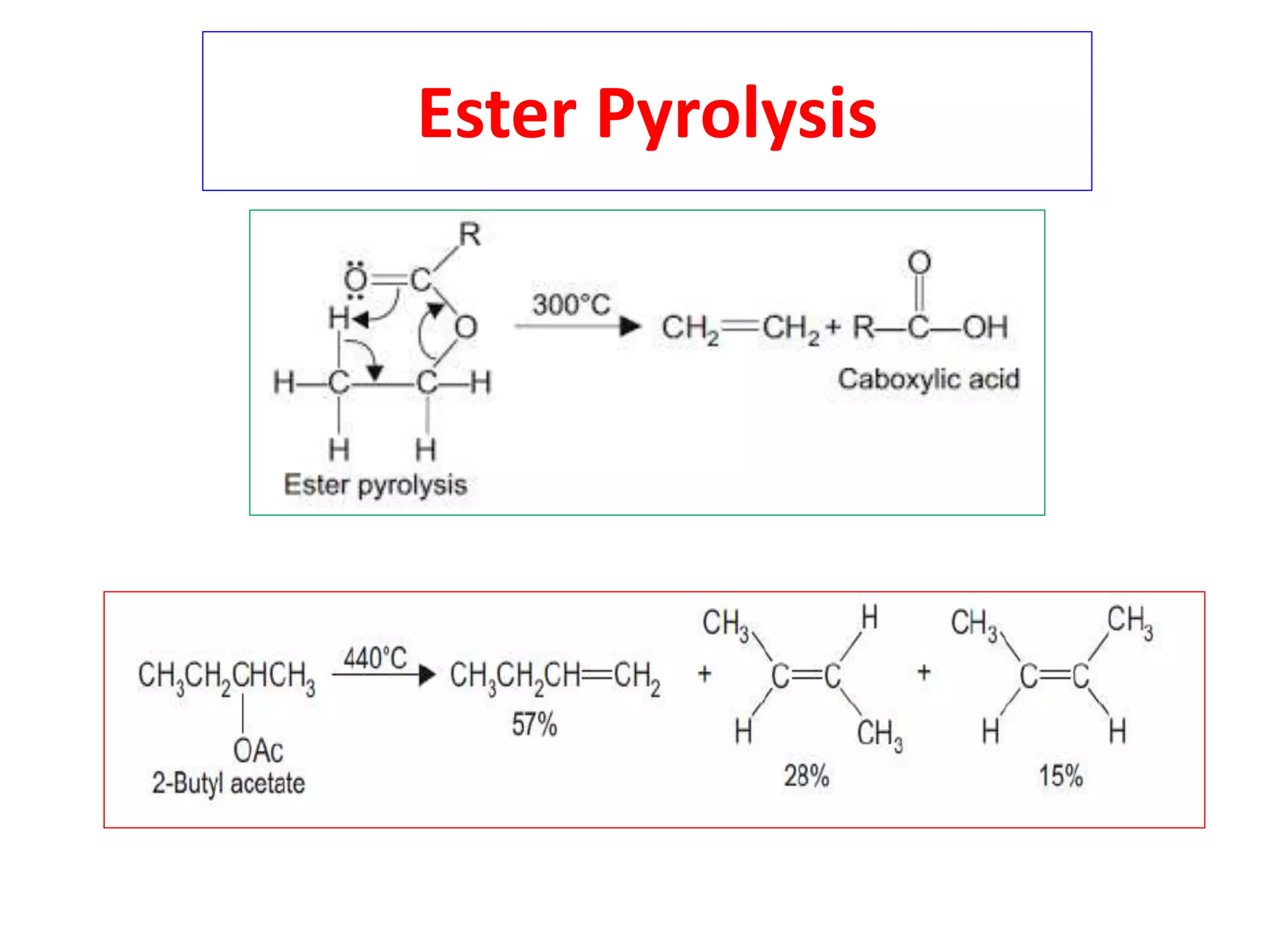
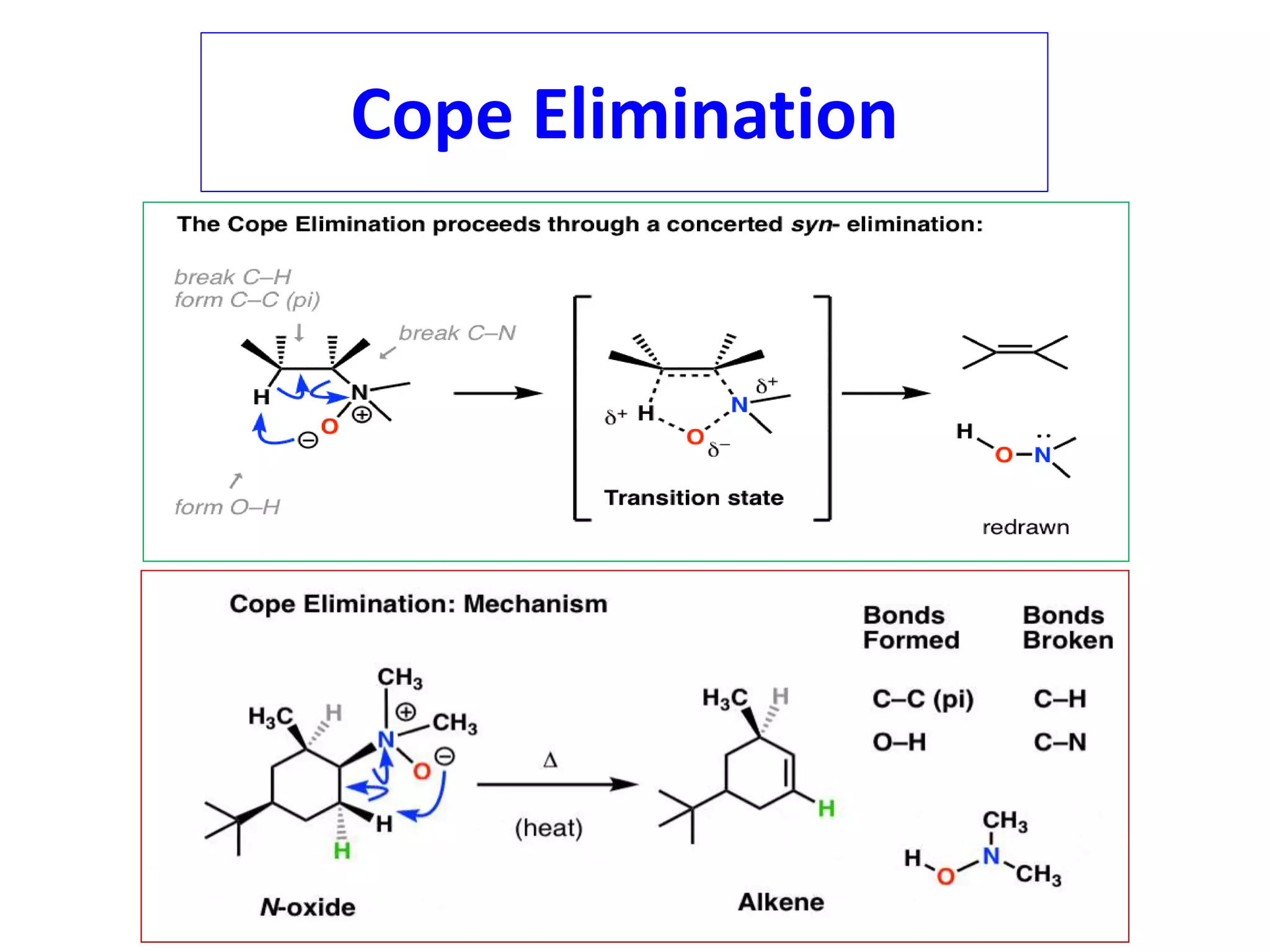
Pyrolytic elimination reactions involve the application of heat to induce an elimination reaction in an organic substrate without the need for an external base or solvent. This type of elimination proceeds through a concerted, syn-elimination via a cyclic transition state that allows for an intramolecular proton transfer and the formation of a new carbon-carbon double bond. Specific examples of pyrolytic eliminations discussed in the document include the conversion of esters to carboxylic acids and alkenes, eliminations in alicyclic systems, Cope eliminations, sulfoxide eliminations, xanthate pyrolysis, and selenoxide eliminations.







![ β-hydroxy phenylsulfoxides were found to undergo thermal
elimination through a 5-membered cyclic transition state, yielding
β-keto esters and methyl ketones after tautomerization.
Allylic alcohols can be formed from β-hydroxy phenylsulfoxides
that contain a β’-hydrogen through an Ei mechanism, tending to
give the β,γ-unsaturation.
1,3-Dienes were found to be formed upon the treatment of an
allylic alcohol with an aryl sulfide in the presence
of triethylamine. Initially, a sulfenate ester is formed followed by
a [2,3]-sigmatropic rearrangement to afford an allylic sulfoxide
which undergoes thermal syn elimination to yield the 1,3-diene.](https://image.slidesharecdn.com/pyrolyticeliminationreactionorganicchemistry-iii-msc33ct21-200420072945/75/Pyrolytic-elimination-reaction-11-2048.jpg)