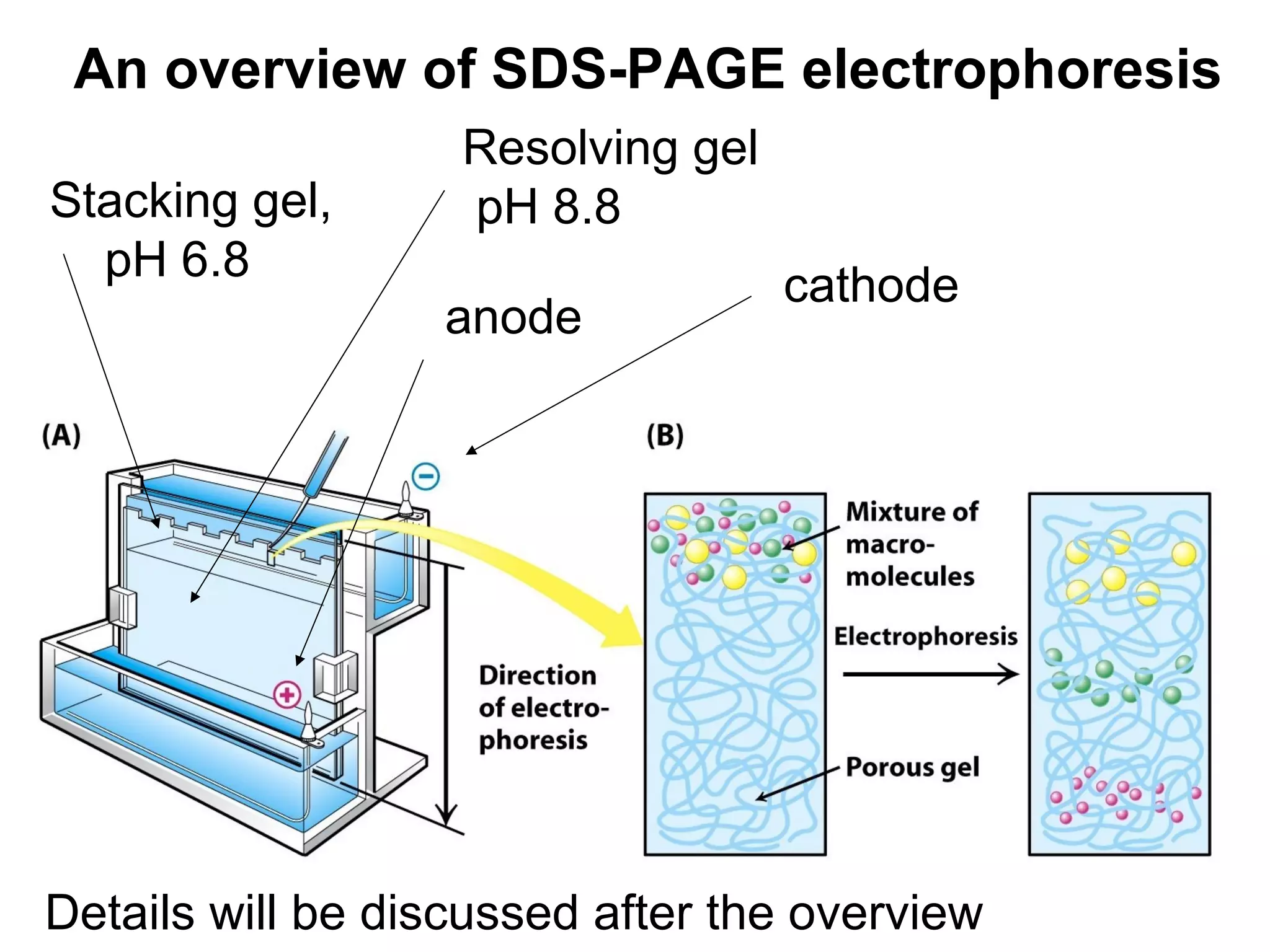
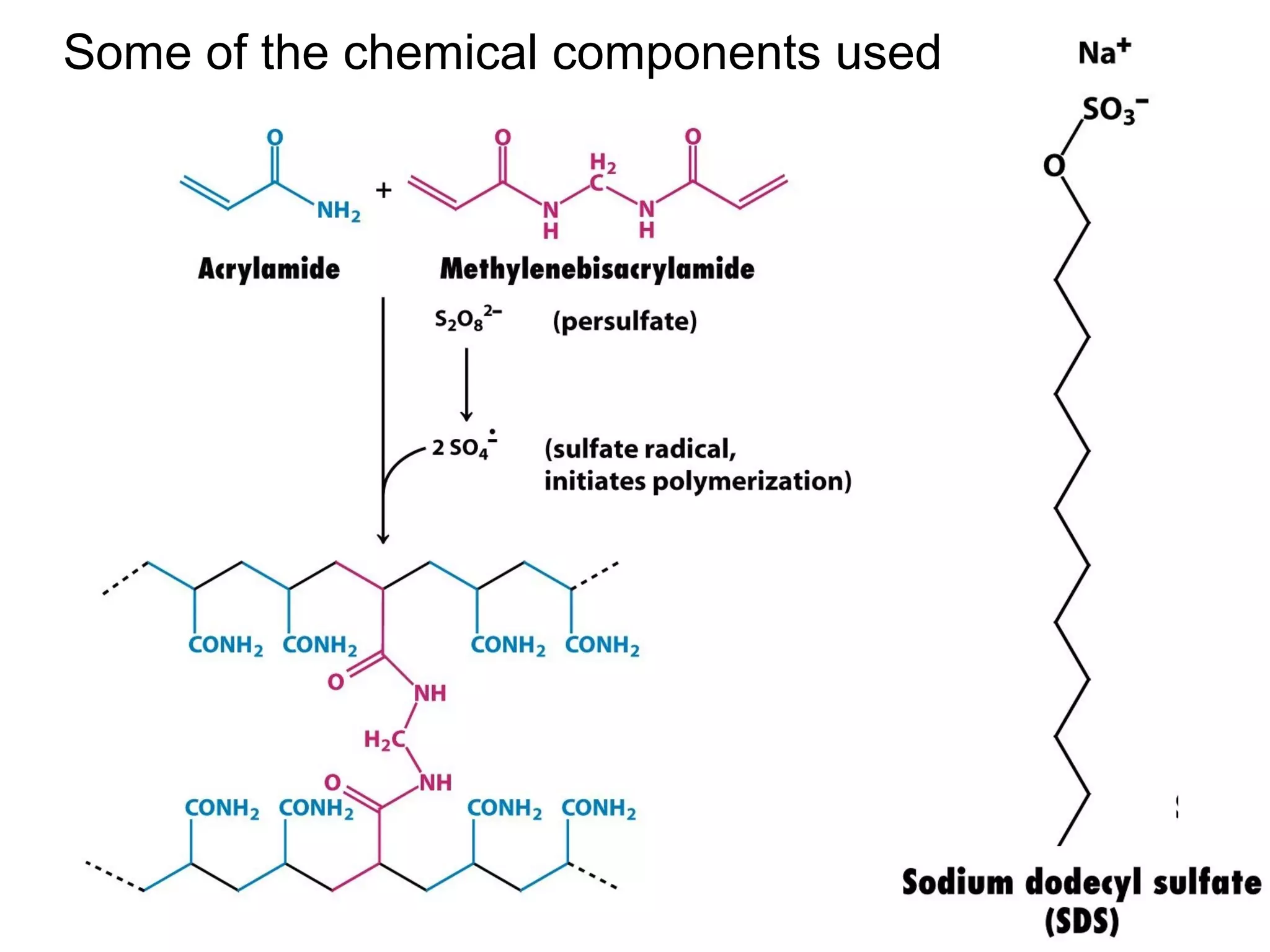
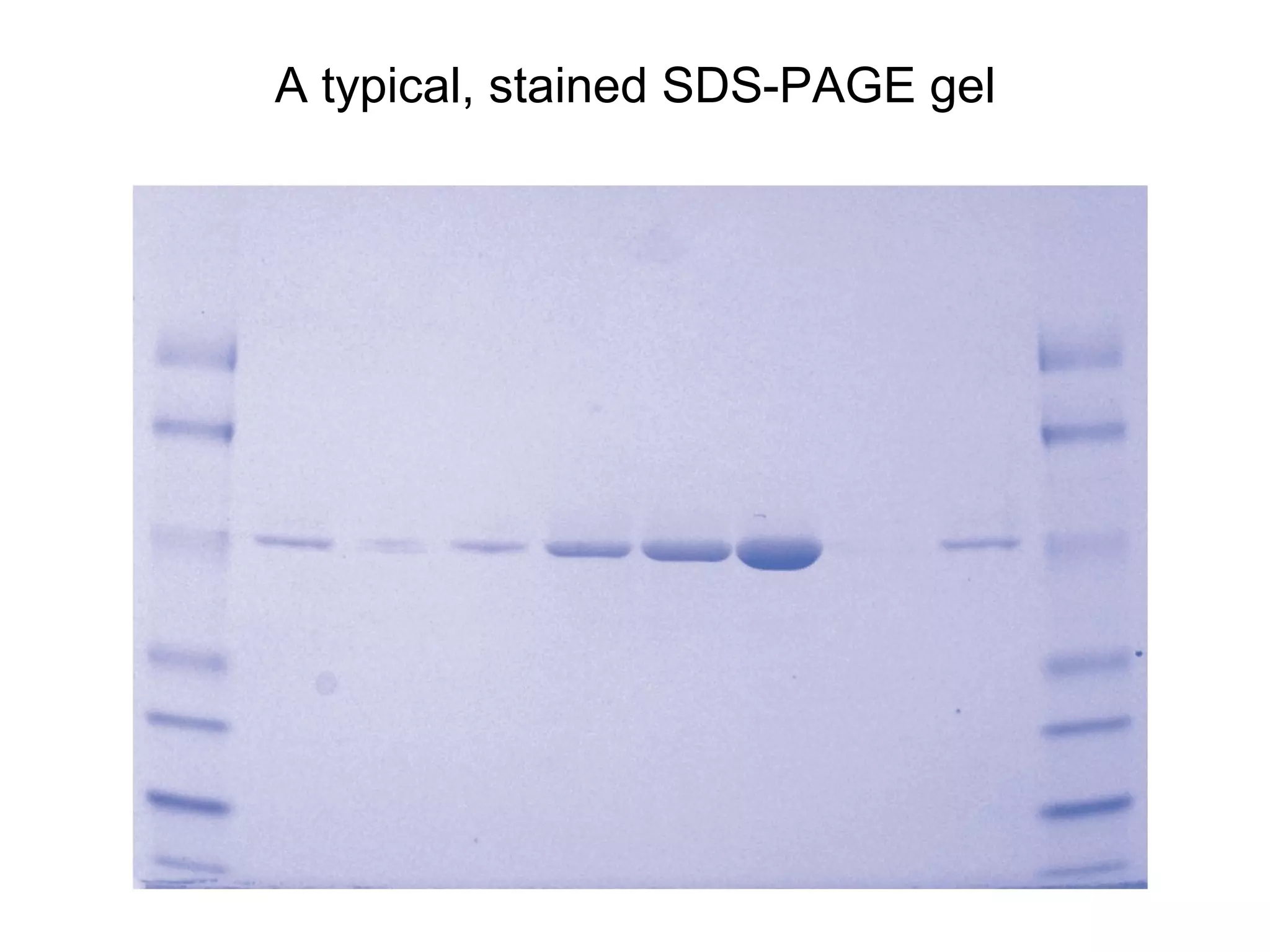
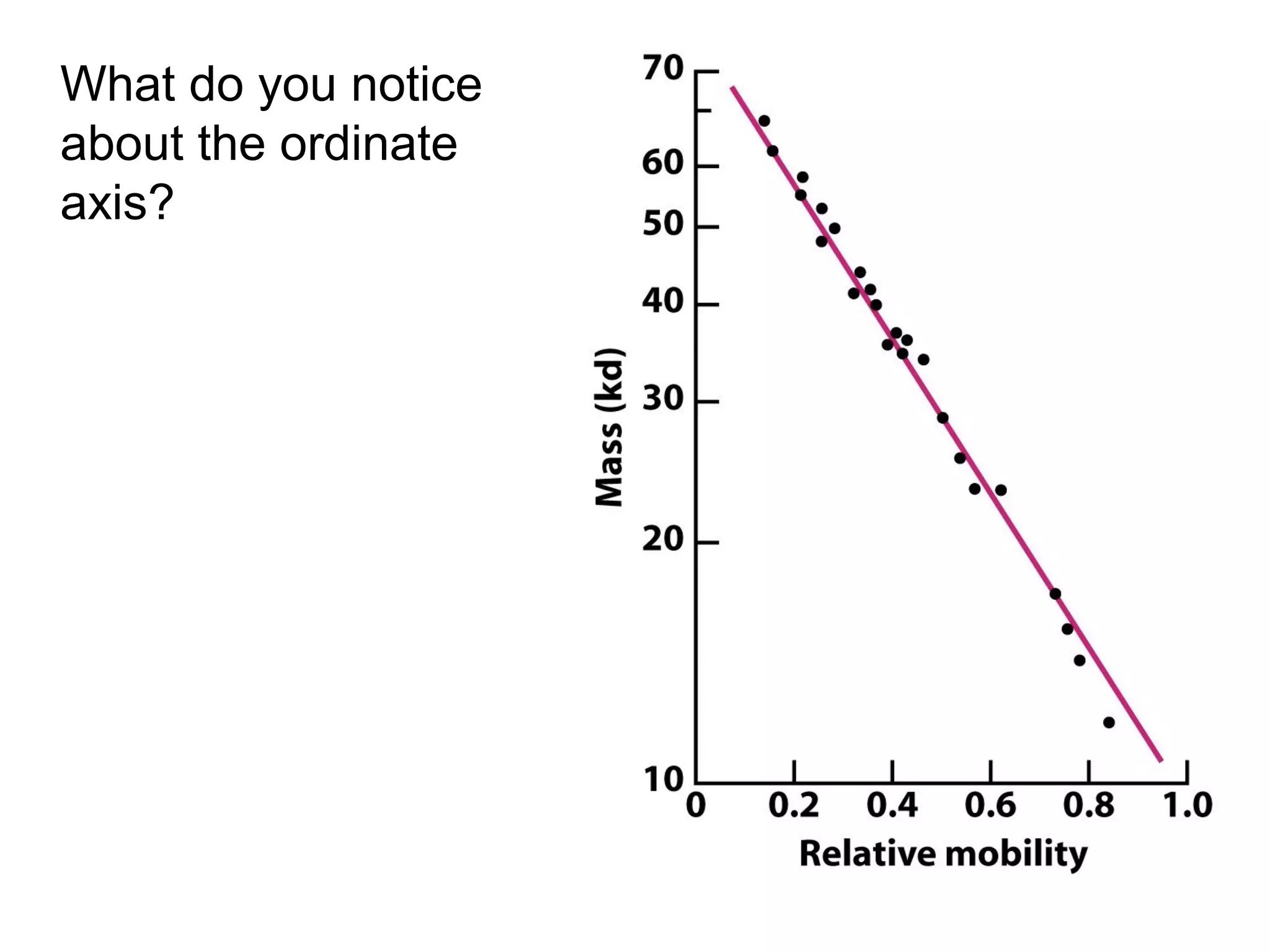
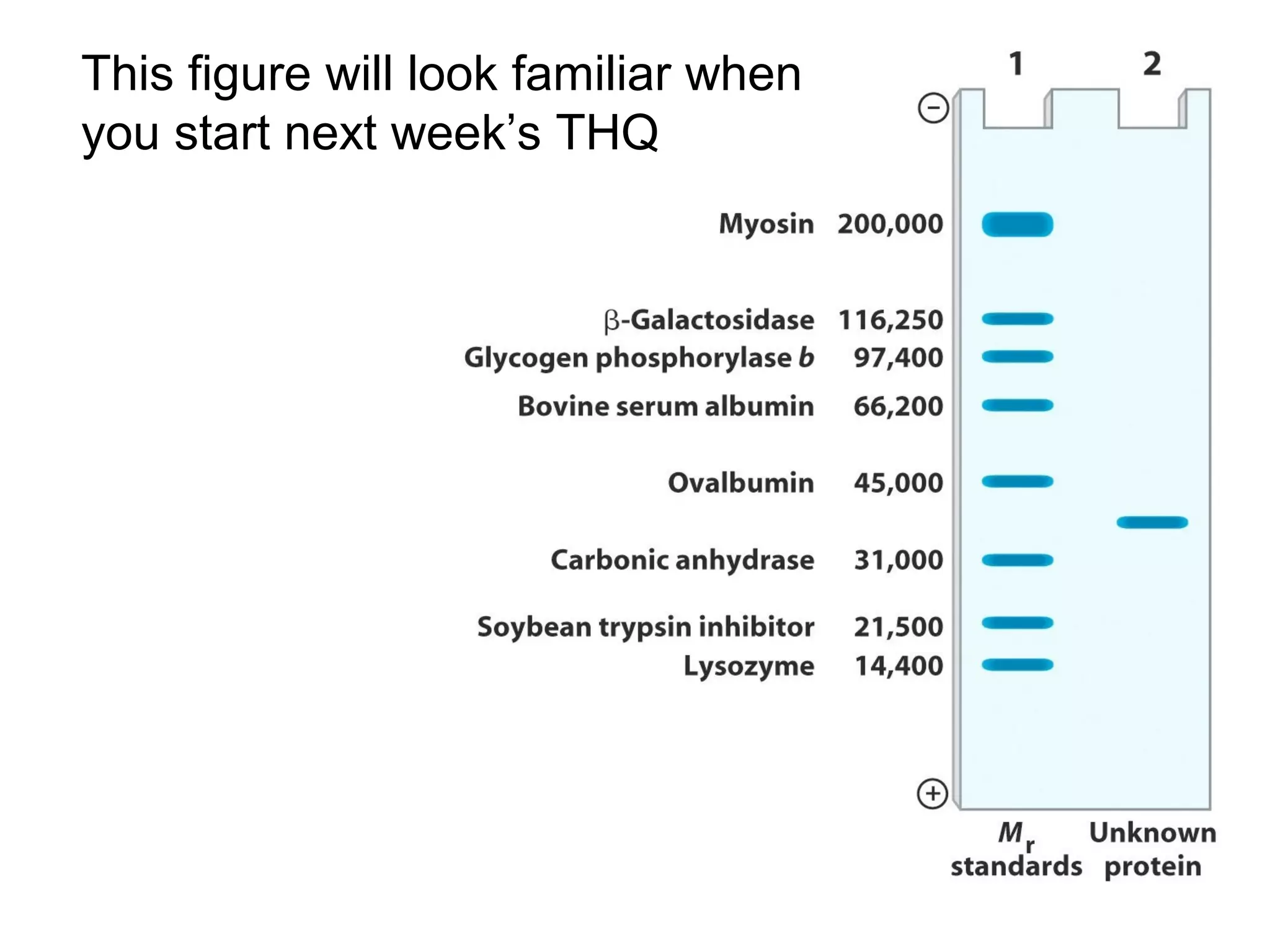
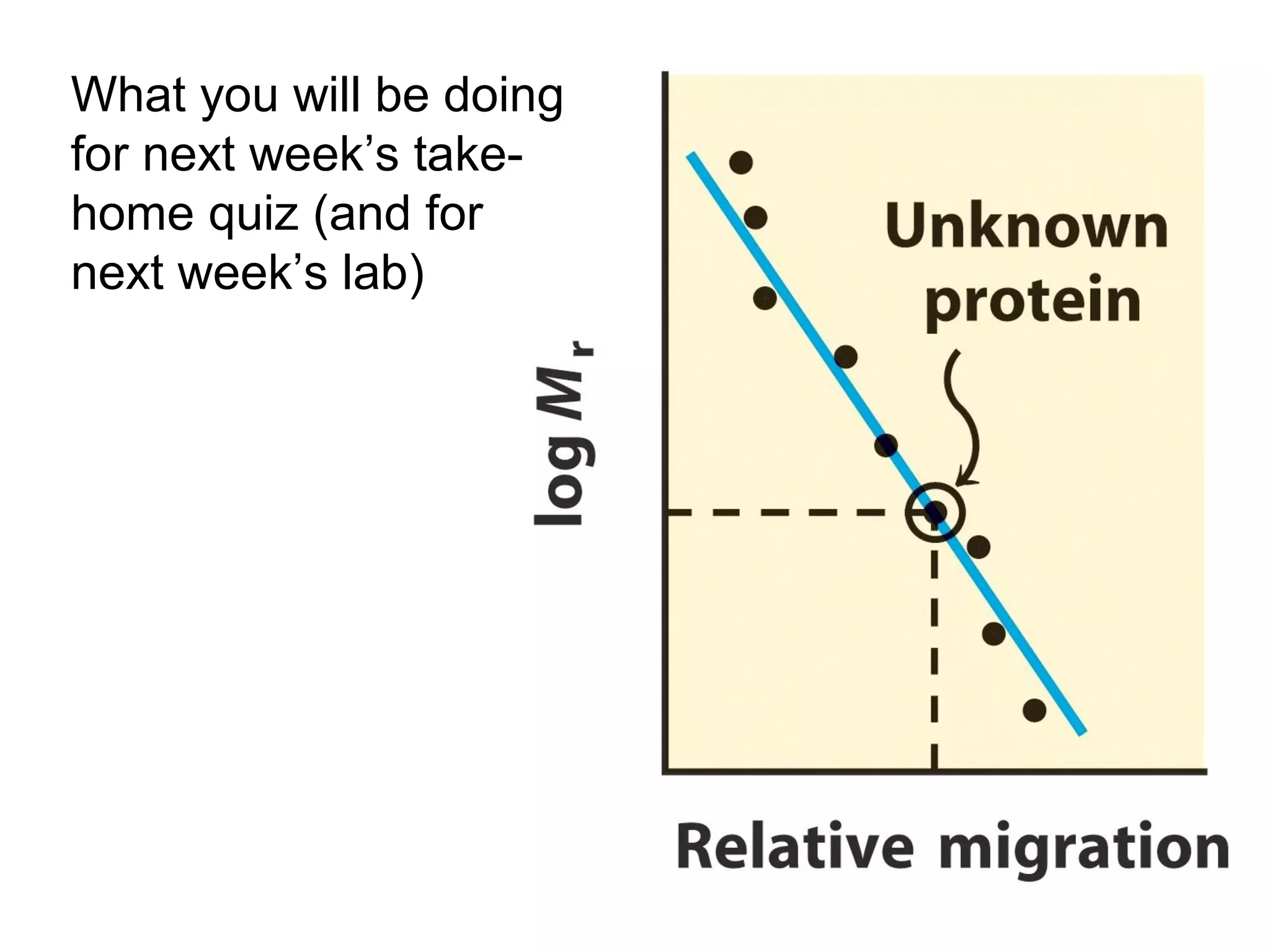
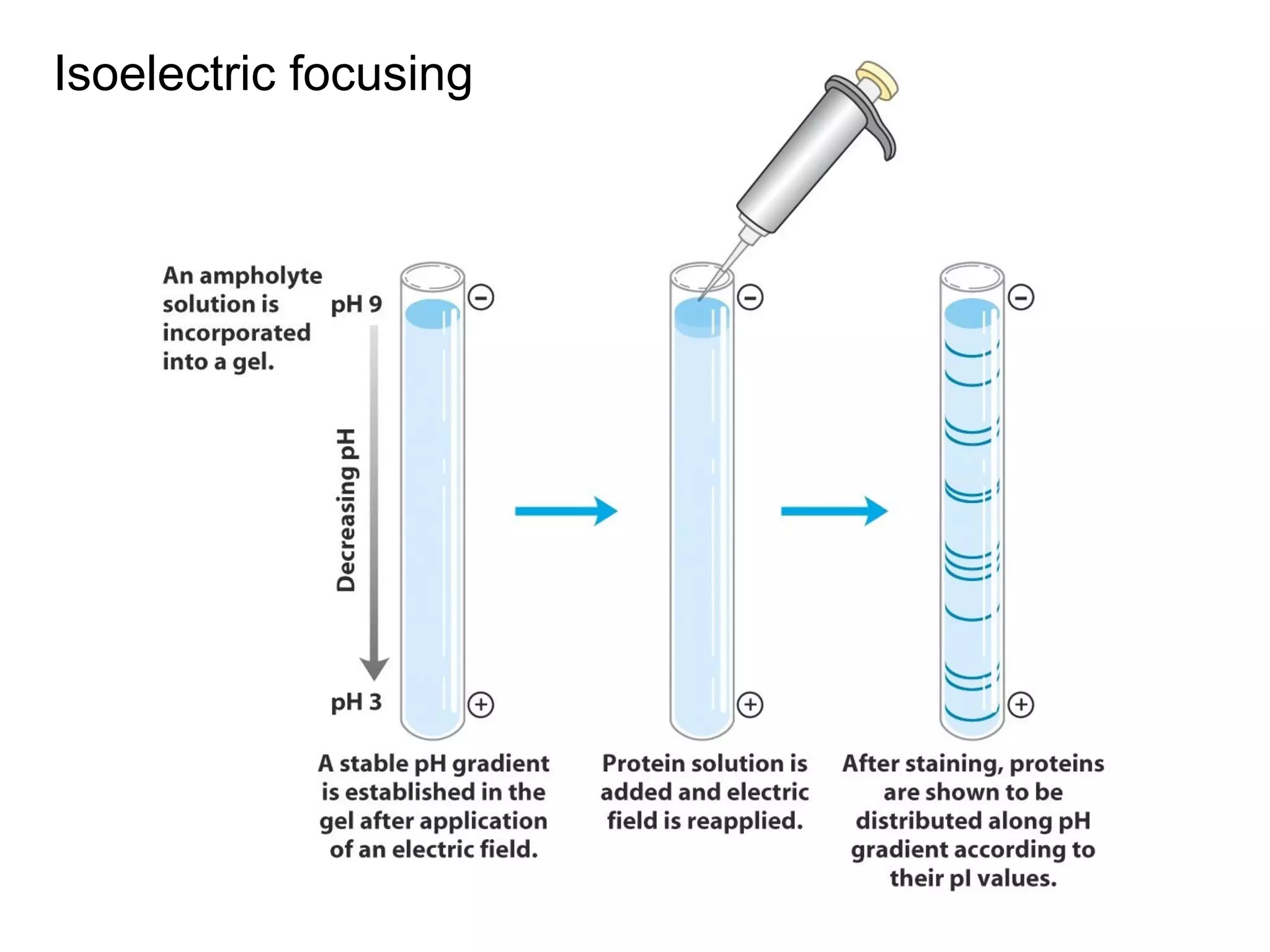
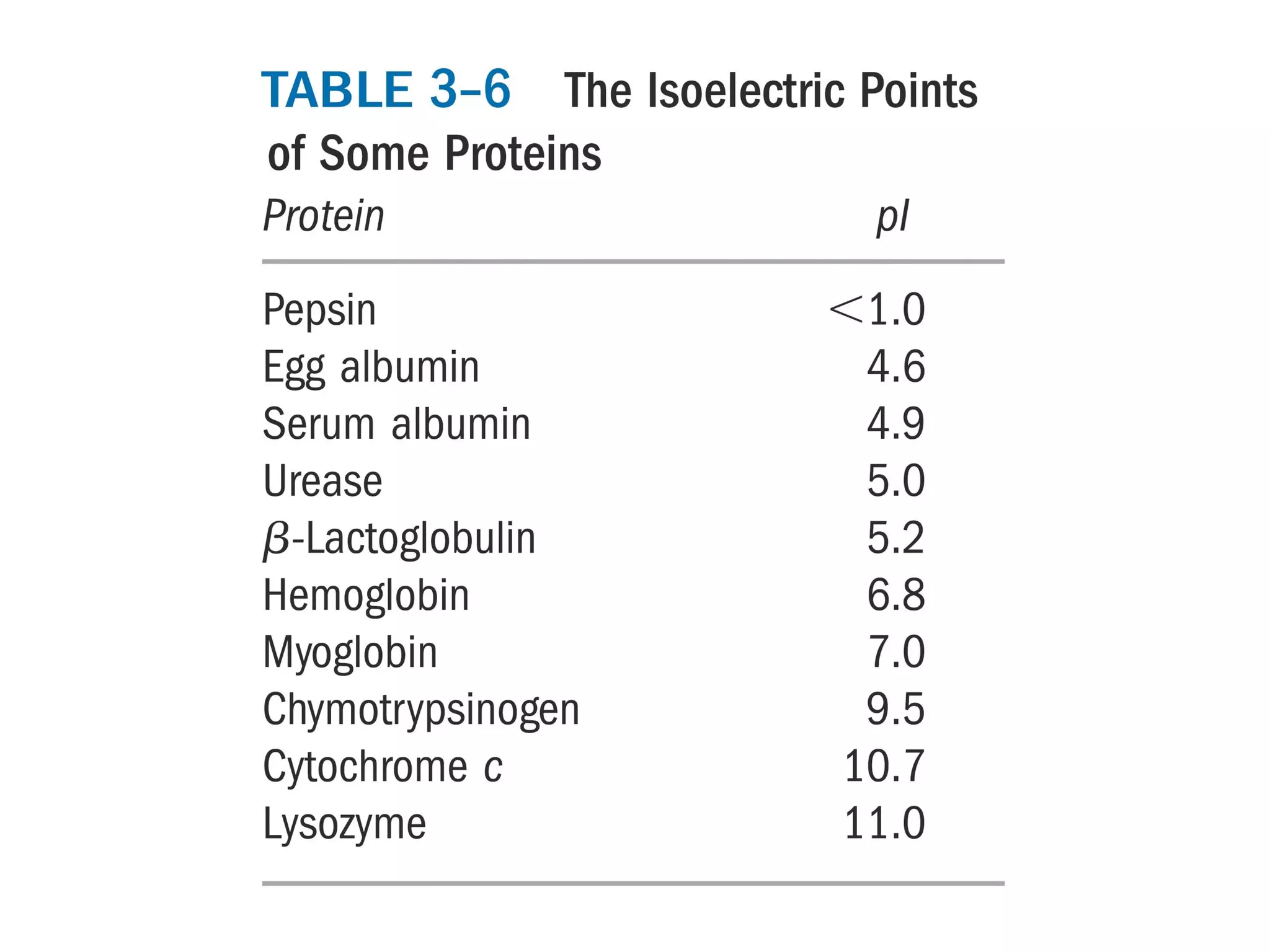
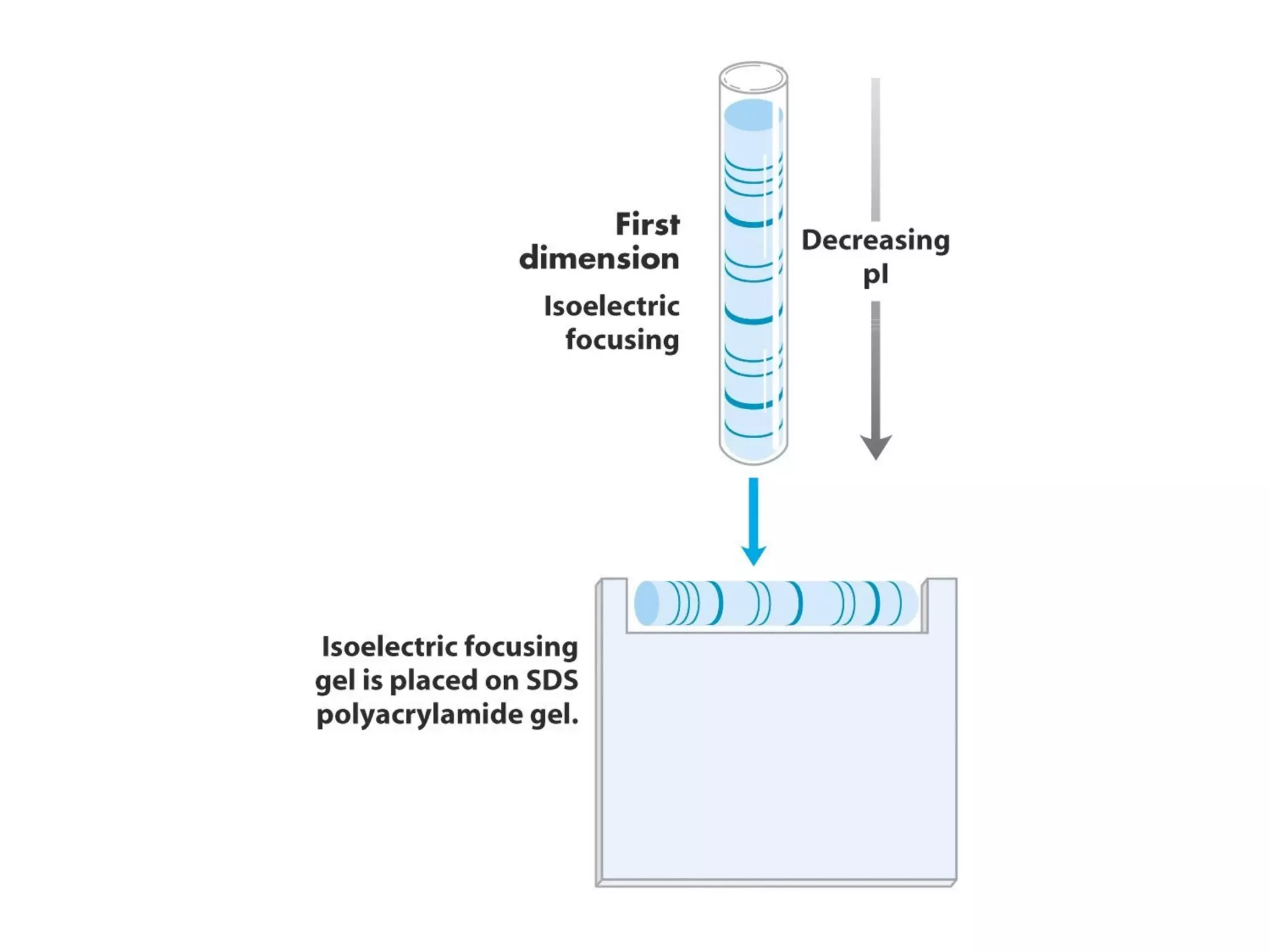
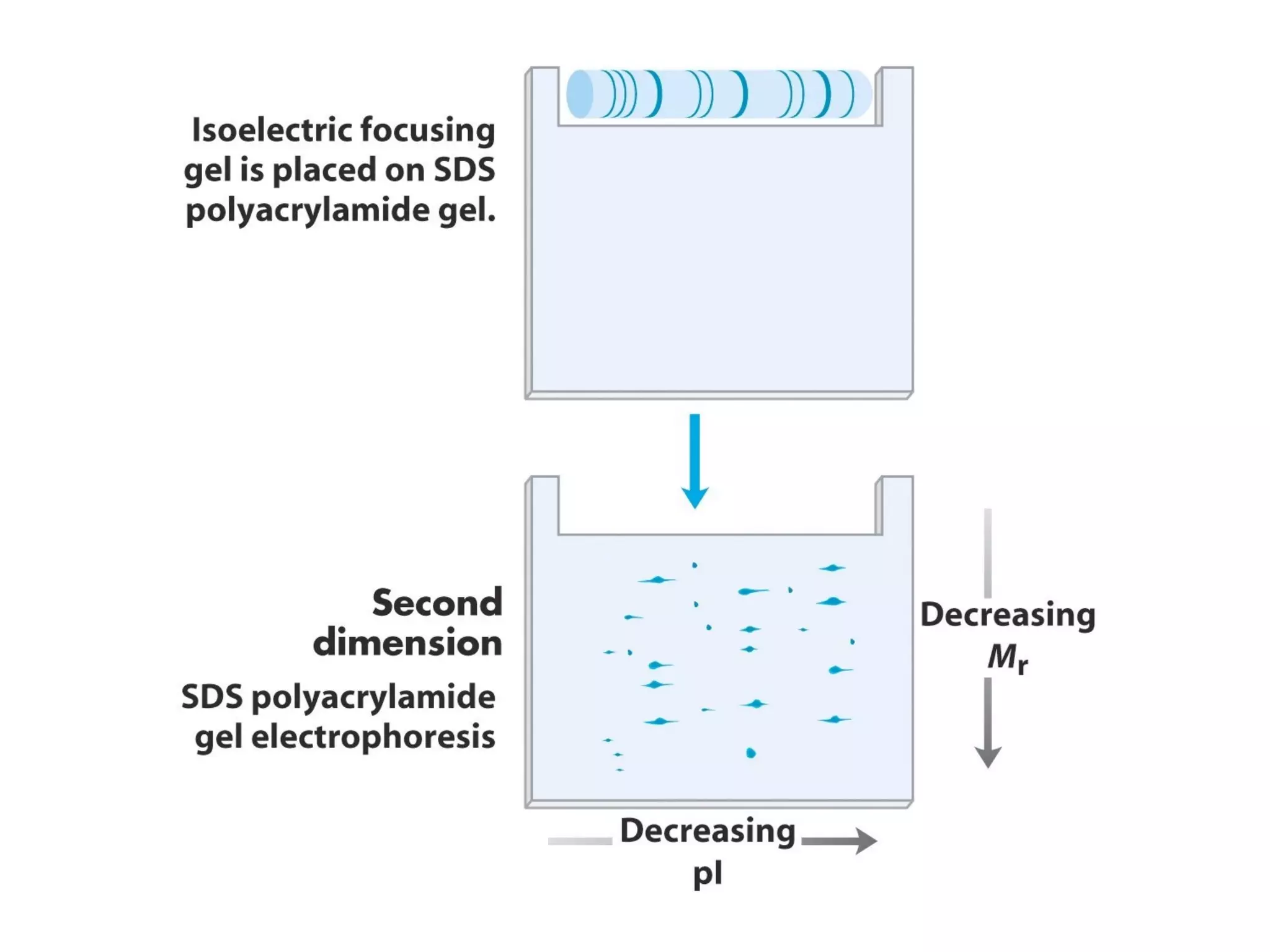
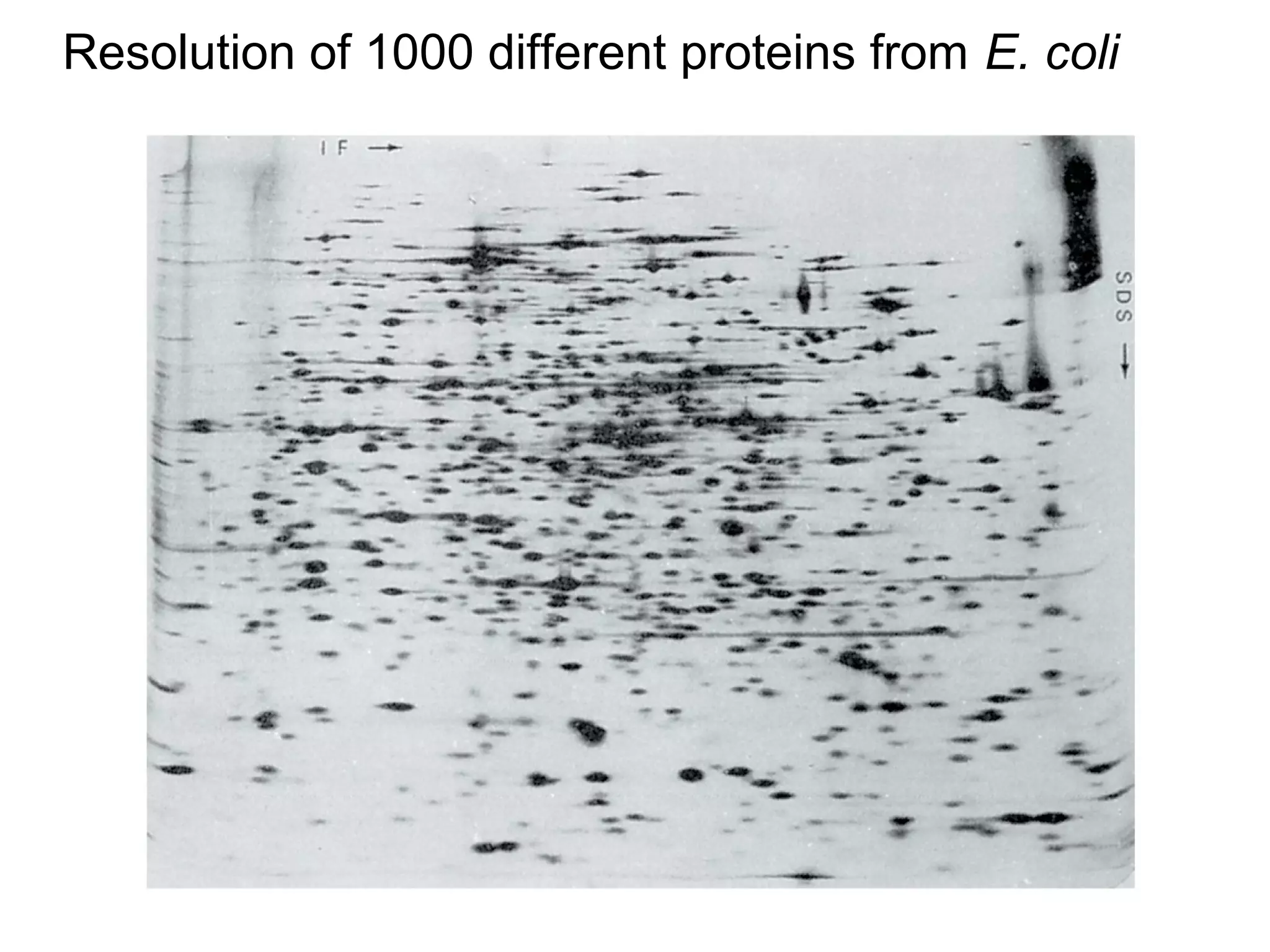
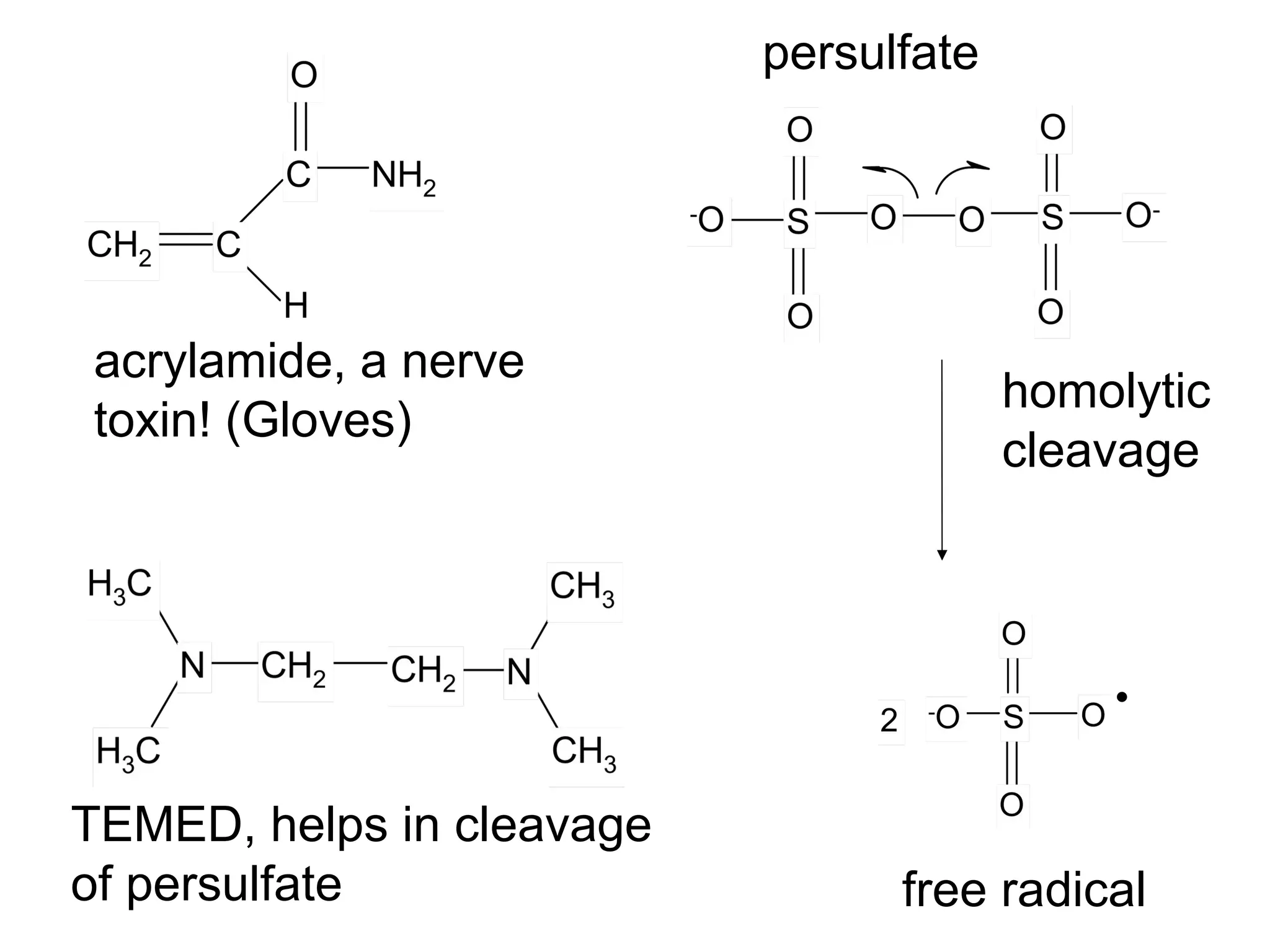
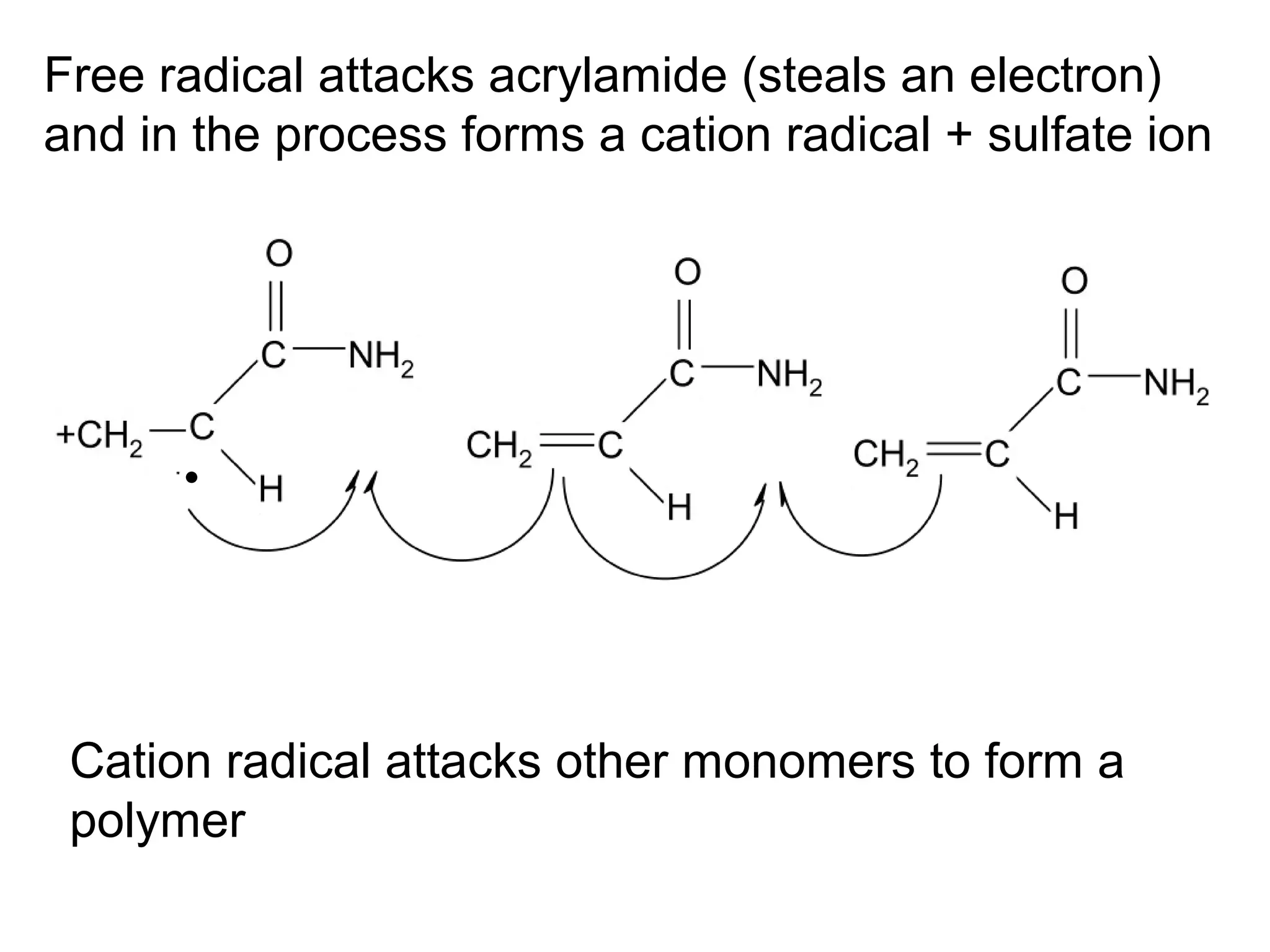
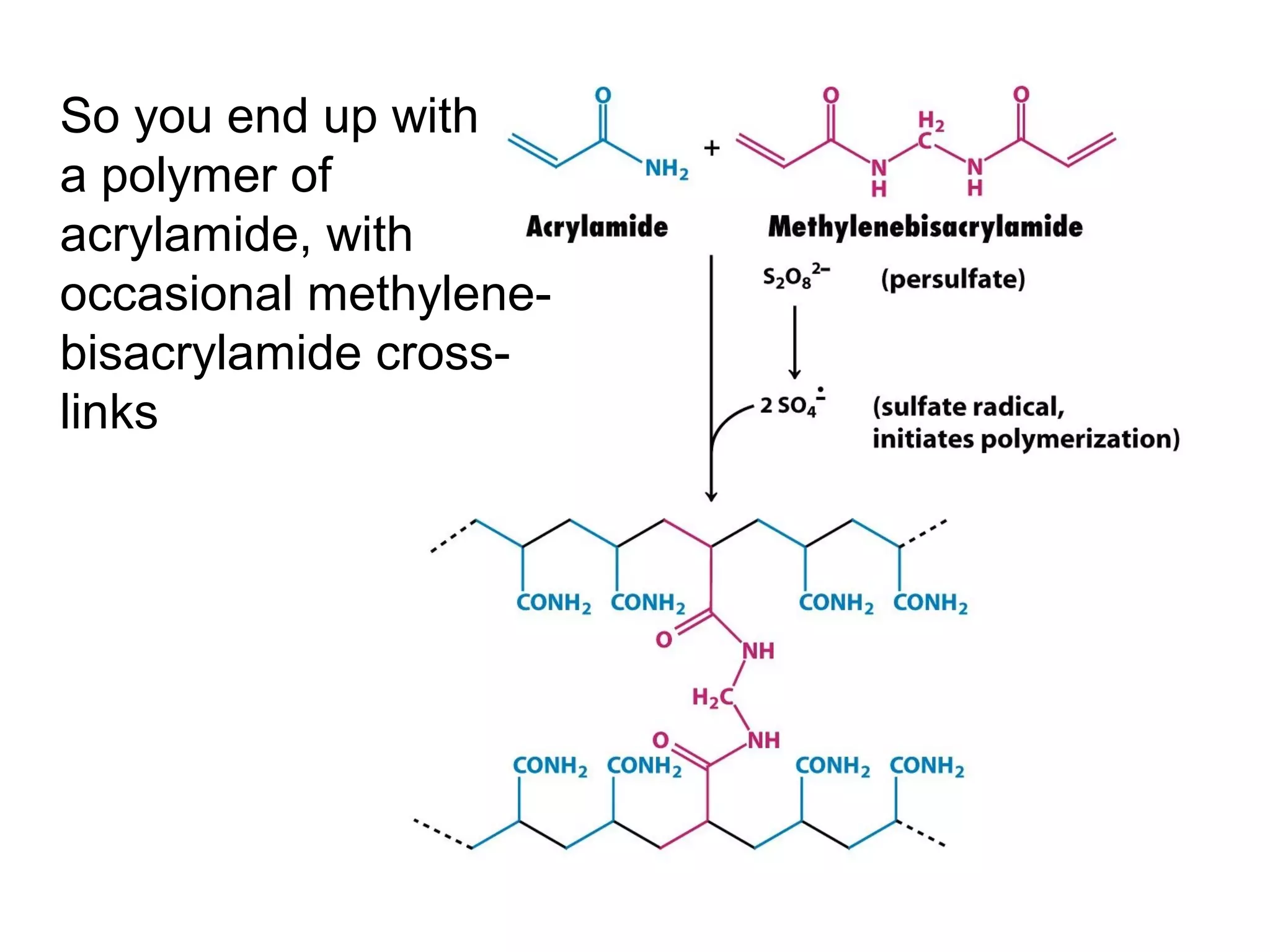
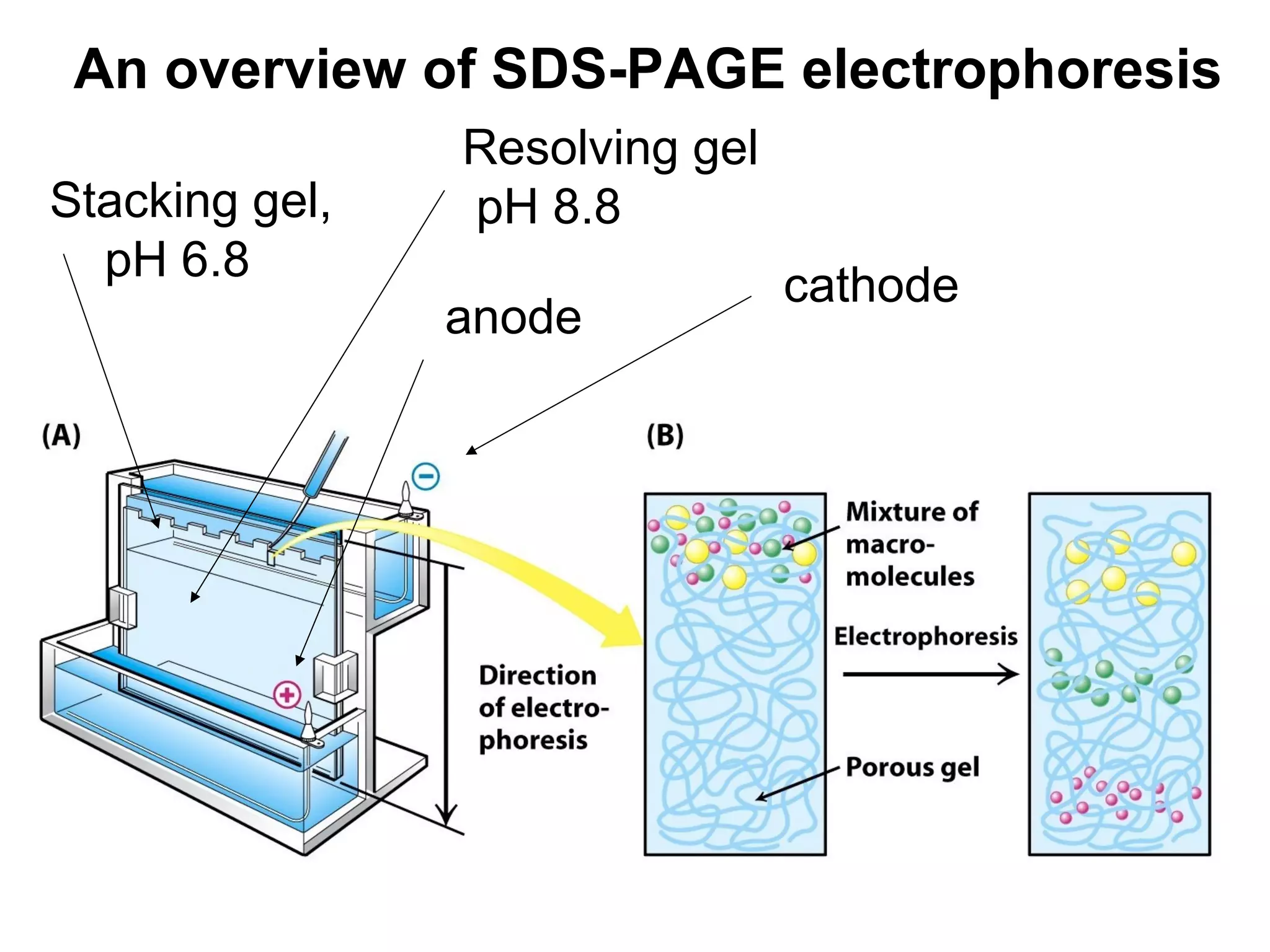
SDS-PAGE electrophoresis is a technique used to separate proteins by size. It involves running proteins through a stacking gel and resolving gel with an electric current. The stacking gel concentrates the proteins into a narrow band before entering the resolving gel, which separates the proteins based on size differences. Key components of SDS-PAGE include SDS to impart identical charge-to-mass ratios to proteins, reducing agents to unfold proteins, and polyacrylamide gels which sieve proteins during electrophoresis based on their size.