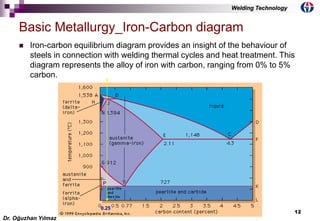
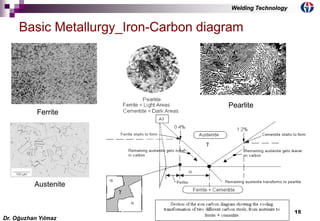
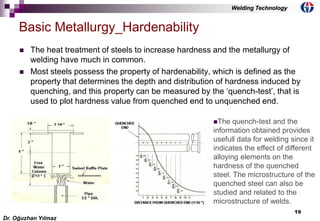
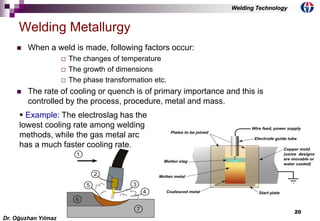
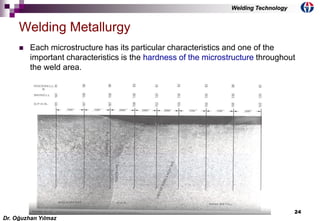
This document discusses welding metallurgy and basic metallurgical concepts relevant to welding. It covers topics like crystalline structures of metals, phase transformations, alloying effects, microstructures like ferrite, pearlite, and martensite, and the influence of cooling rate on microstructure. It also discusses the heat affected zone and issues that can arise from changes in composition and cooling rate near the weld interface.


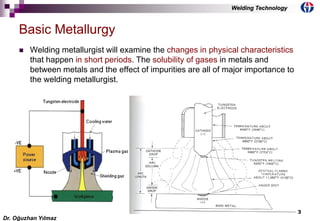

![6
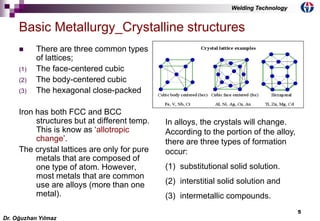
Substitutional solid solution: the atoms of the metal making up
the minor portion of the alloy will at random replace some of
the atoms of the metal making up the majority of the alloy.
Interstitial solid solution: The atoms of the minor metal in the
alloy are much smaller than those in the major lattice, they do
not replace the atoms of the major metal in the lattice but
rather locate in points between or intervening spaces known
as interstices in the lattice.
Intermetallic compounds: the minor metal atoms in the alloy
cannot completely dissolve either interstitially or
substitutionally. They will form the type of chemical compound
the composition of which corresponds roughly to the chemical
formula. This results in the formation of mixed kinds of atomic
groupings consisting of different and complicated crystalline
structure. [Fe3C, Cementite,Iron-Carbide]
Each group with its own crystalline structure is referred to as a
phase.
Basic Metallurgy_Crystalline structures
Dr. Oğuzhan Yılmaz
Welding Technology](https://image.slidesharecdn.com/weldingmetallurgyparti-150422040356-conversion-gate01/85/Welding-metallurgy-part-i-6-320.jpg)