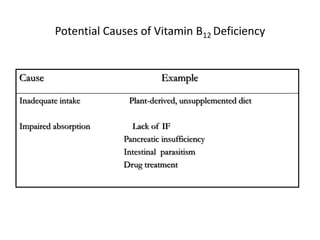
Vitamin B12 is essential for normal cell metabolism and DNA synthesis. Deficiency can result from inadequate dietary intake or impaired absorption. Initial signs include fatigue and pallor, while long-term deficiency leads to megaloblastic anemia and peripheral neuropathy. Diagnosis involves measuring serum B12 levels along with metabolites like methylmalonic acid and homocysteine, which increase with deficiency. The Schilling test evaluates absorption by measuring urinary excretion of radioactive B12.