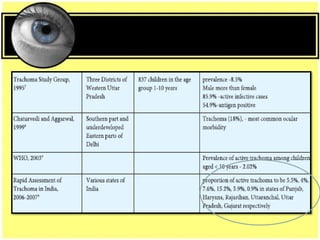
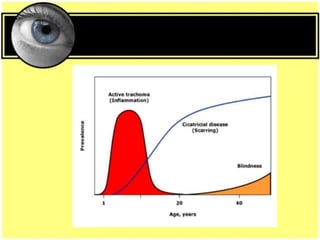
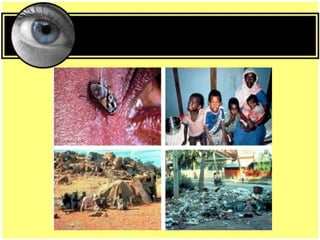
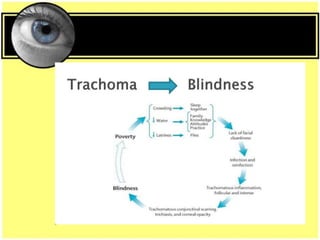
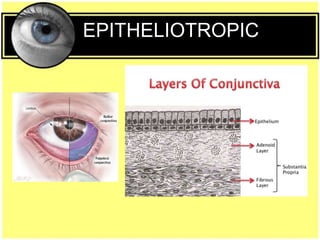
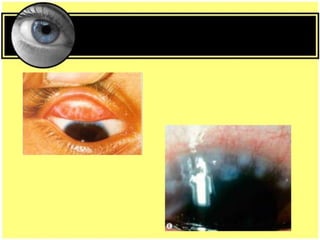
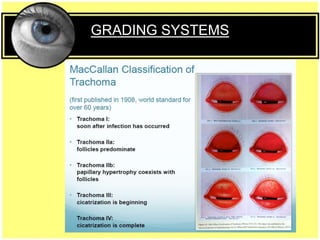
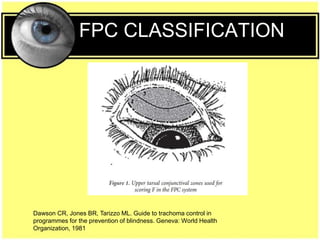
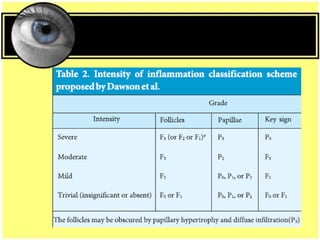
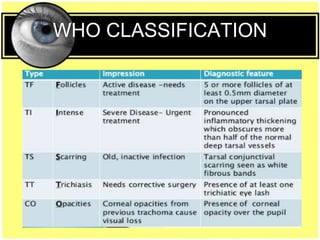
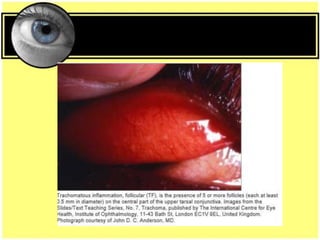
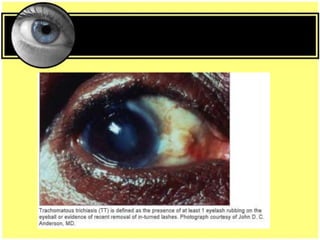
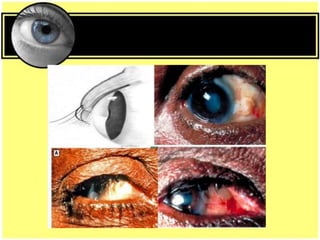
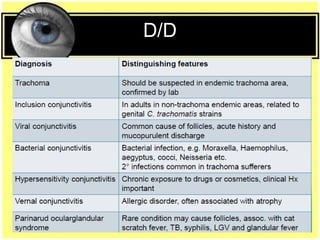
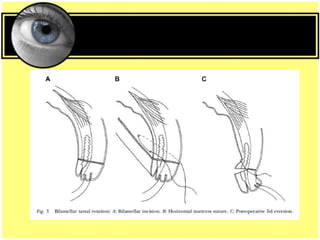
This document provides an overview of trachoma, the leading infectious cause of preventable blindness worldwide. It is caused by repeated infections with Chlamydia trachomatis bacteria, which leads to conjunctival scarring and trichiasis (turned in eyelashes). The highest prevalence is in parts of Africa, the Middle East, South Asia, and South America. Trachoma is diagnosed clinically and managed using the SAFE strategy of surgery, antibiotics, facial cleanliness and environmental improvement.