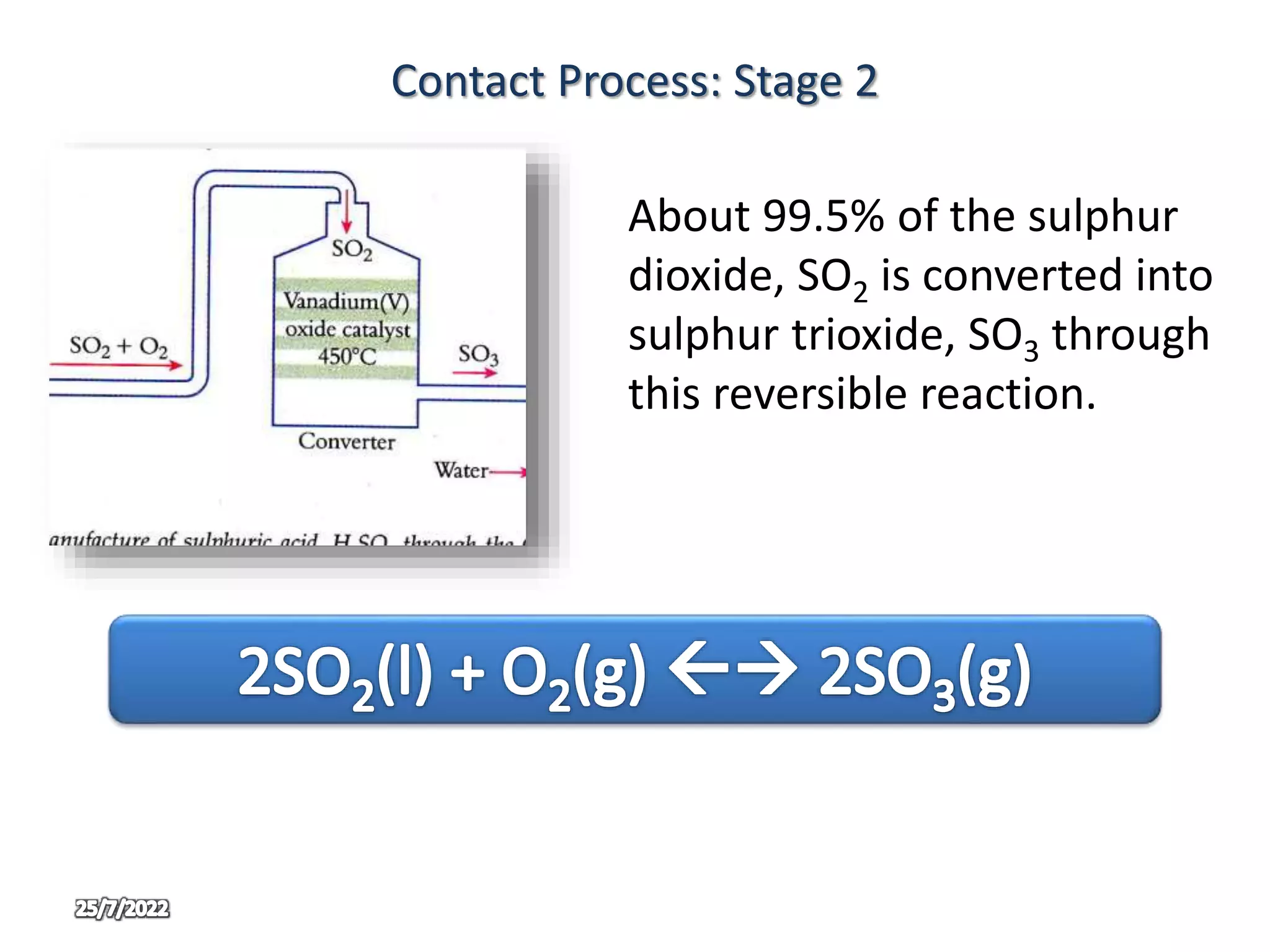
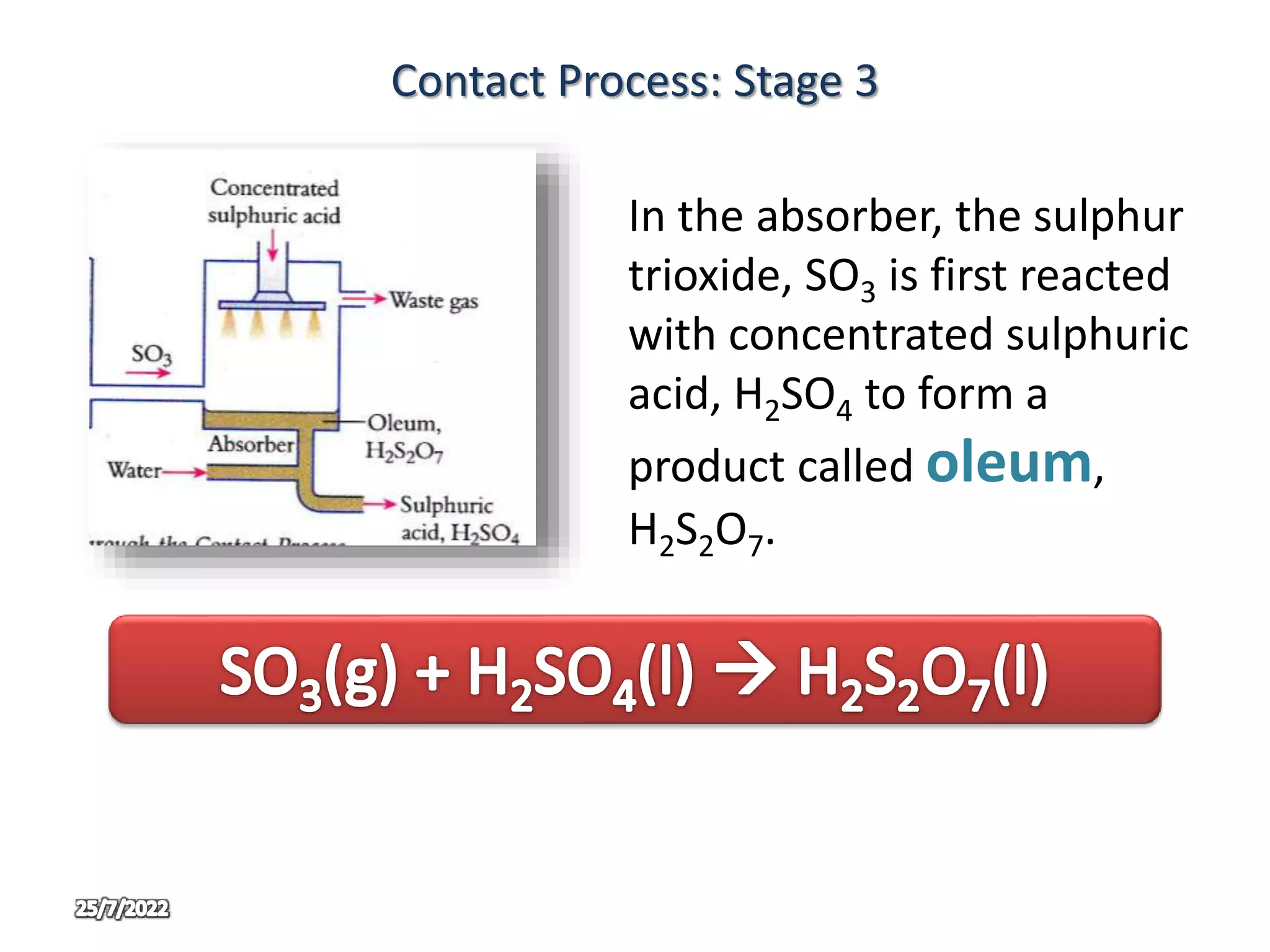
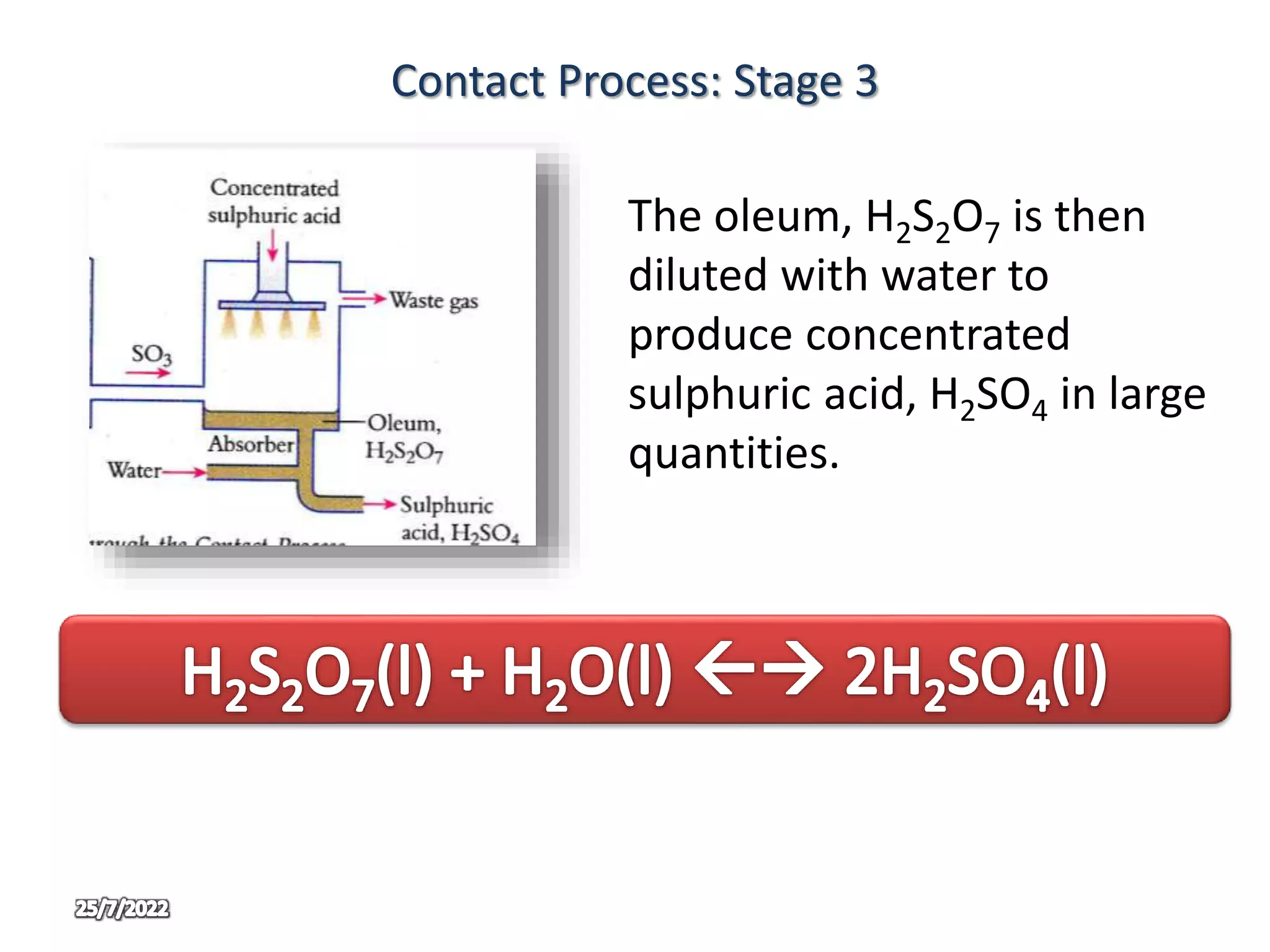
The contact process is used to manufacture sulphuric acid through three main stages. In stage one, sulphur is burned in air to produce sulphur dioxide. In stage two, sulphur dioxide and oxygen are passed over a vanadium oxide catalyst at 450°C to produce sulphur trioxide. In stage three, the sulphur trioxide first reacts with concentrated sulphuric acid to form oleum, which is then diluted with water to produce concentrated sulphuric acid in large quantities. The use of oleum prevents the too violent reaction that would occur if sulphur trioxide reacted directly with water.












![FAQ 1
• The two reaction in the third stage are equivalent to
adding Sulphur trioxide, SO3, directly to water
• Then why can’t we just skipped concentrated
sulphuric acid step [thus not forming oleum]?](https://image.slidesharecdn.com/contactprocess-220725034246-6fe0c98b/75/The-contact-process-pptx-16-2048.jpg)

