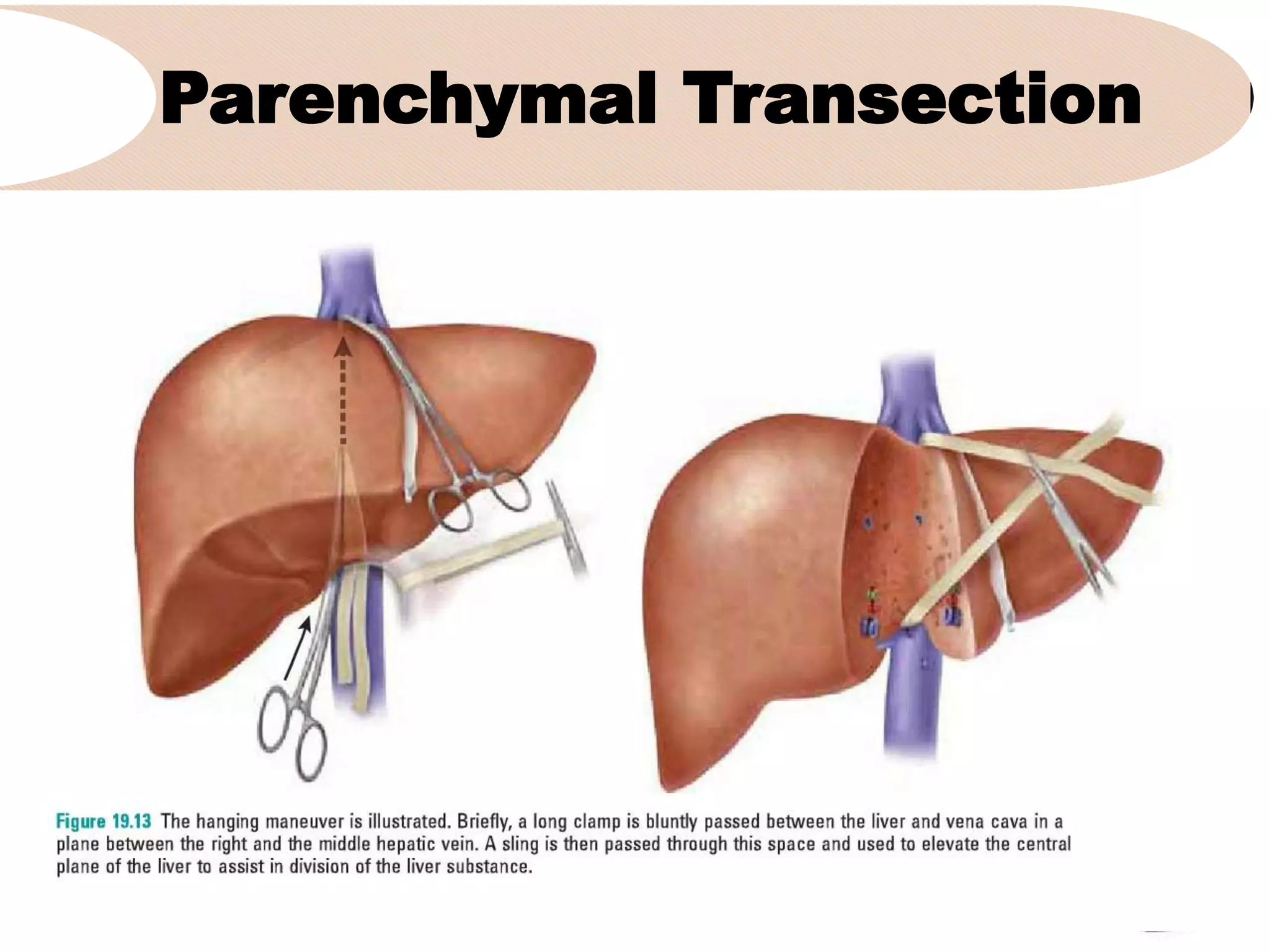
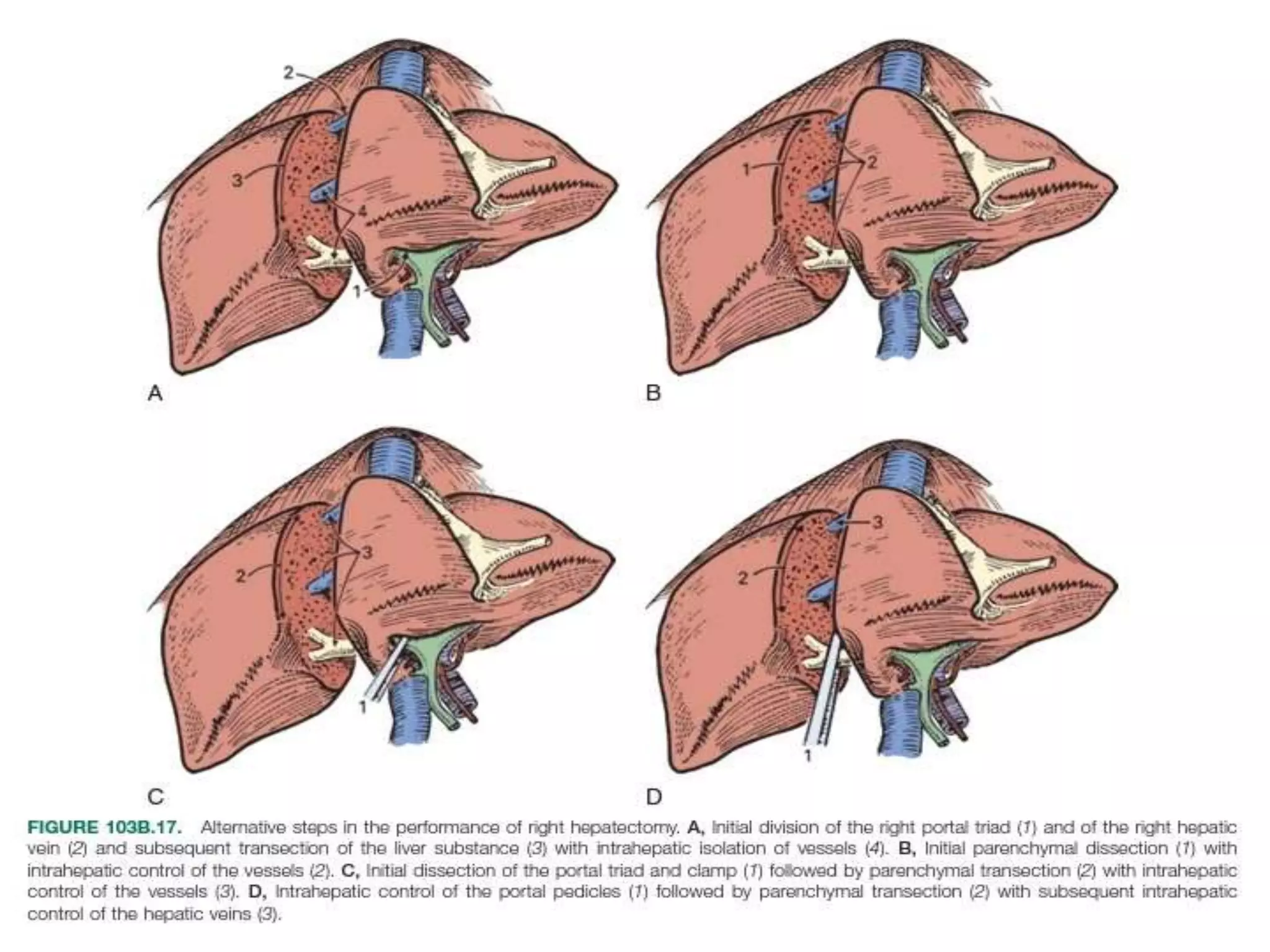
The document provides information about right hepatectomy surgery:
1. It discusses the traditional and Brisbane terminology used to describe right hepatectomy and the indications for the procedure such as primary liver tumors, metastatic tumors, and large benign tumors.
2. It notes some of the challenges with right hepatectomy due to the large volume of the right liver and higher risk of postoperative hepatic failure compared to other procedures.
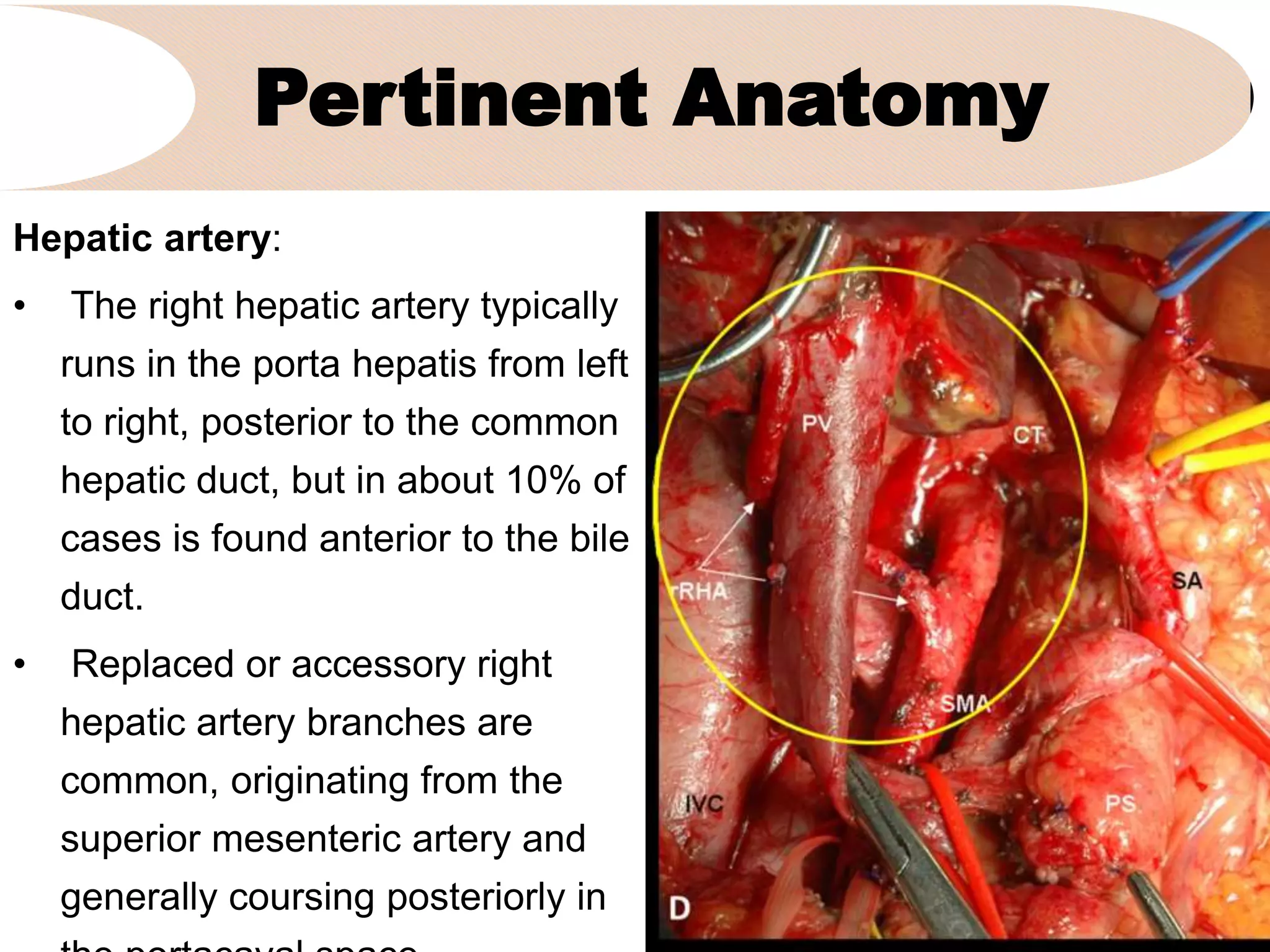
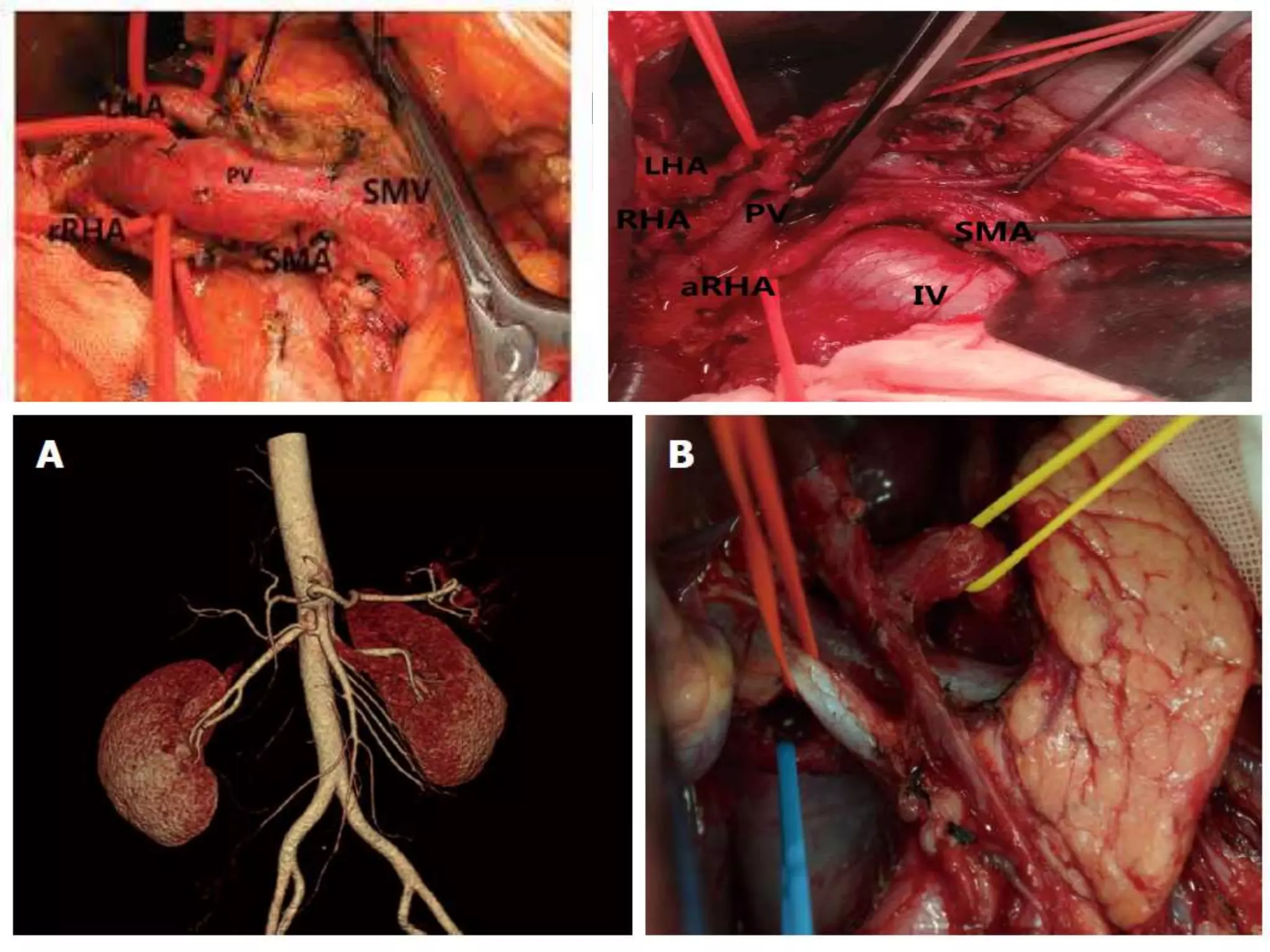
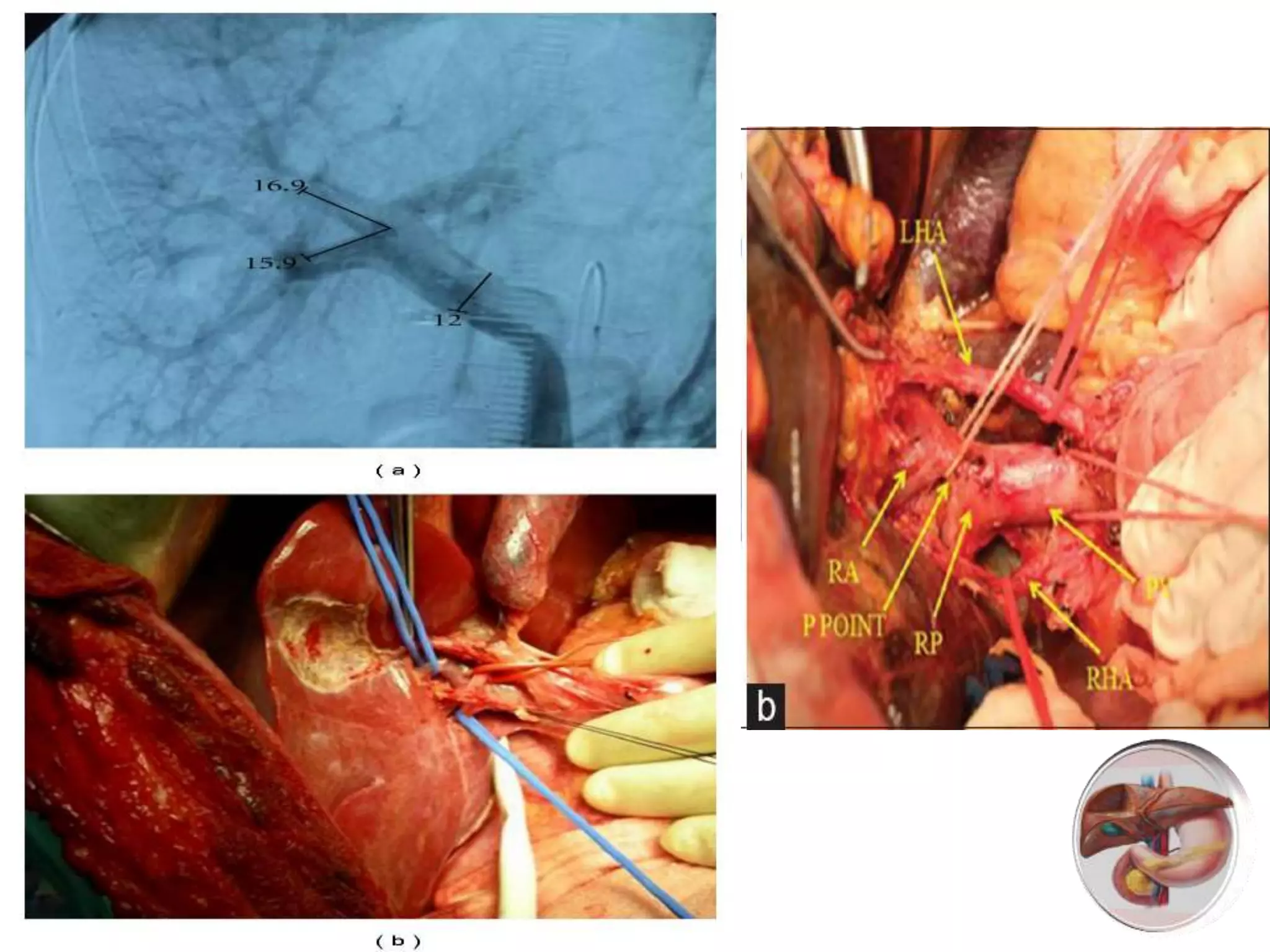
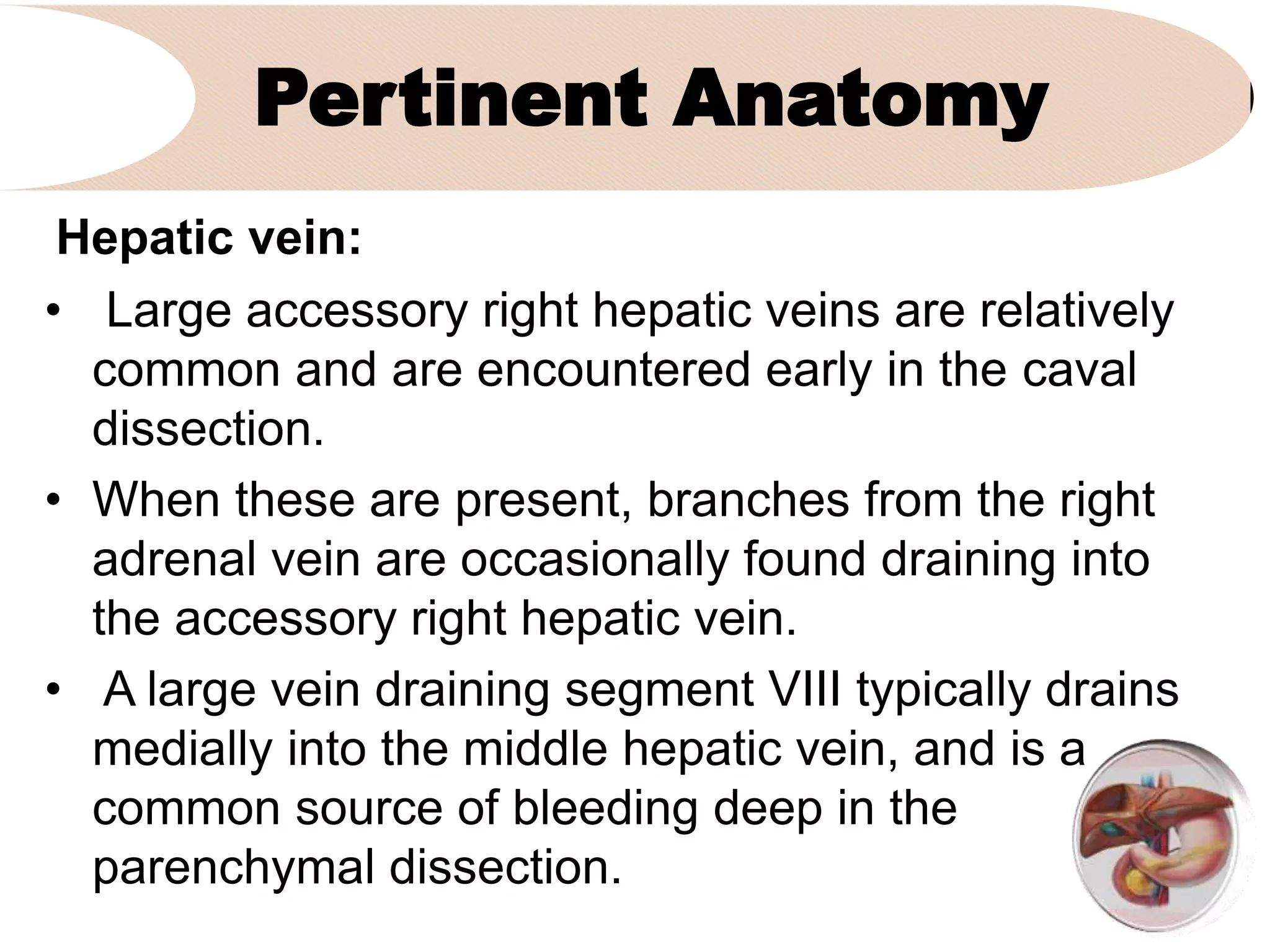
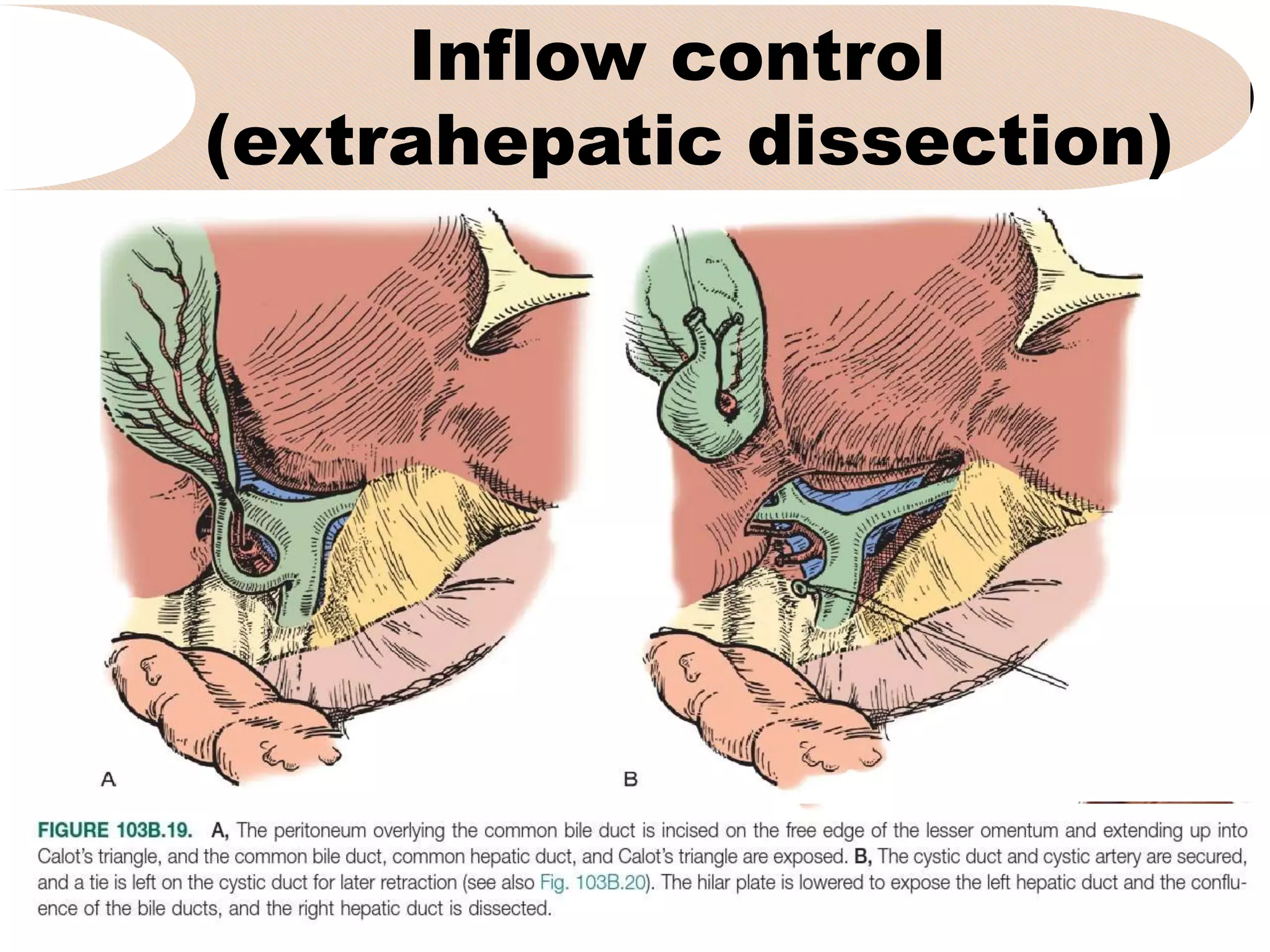
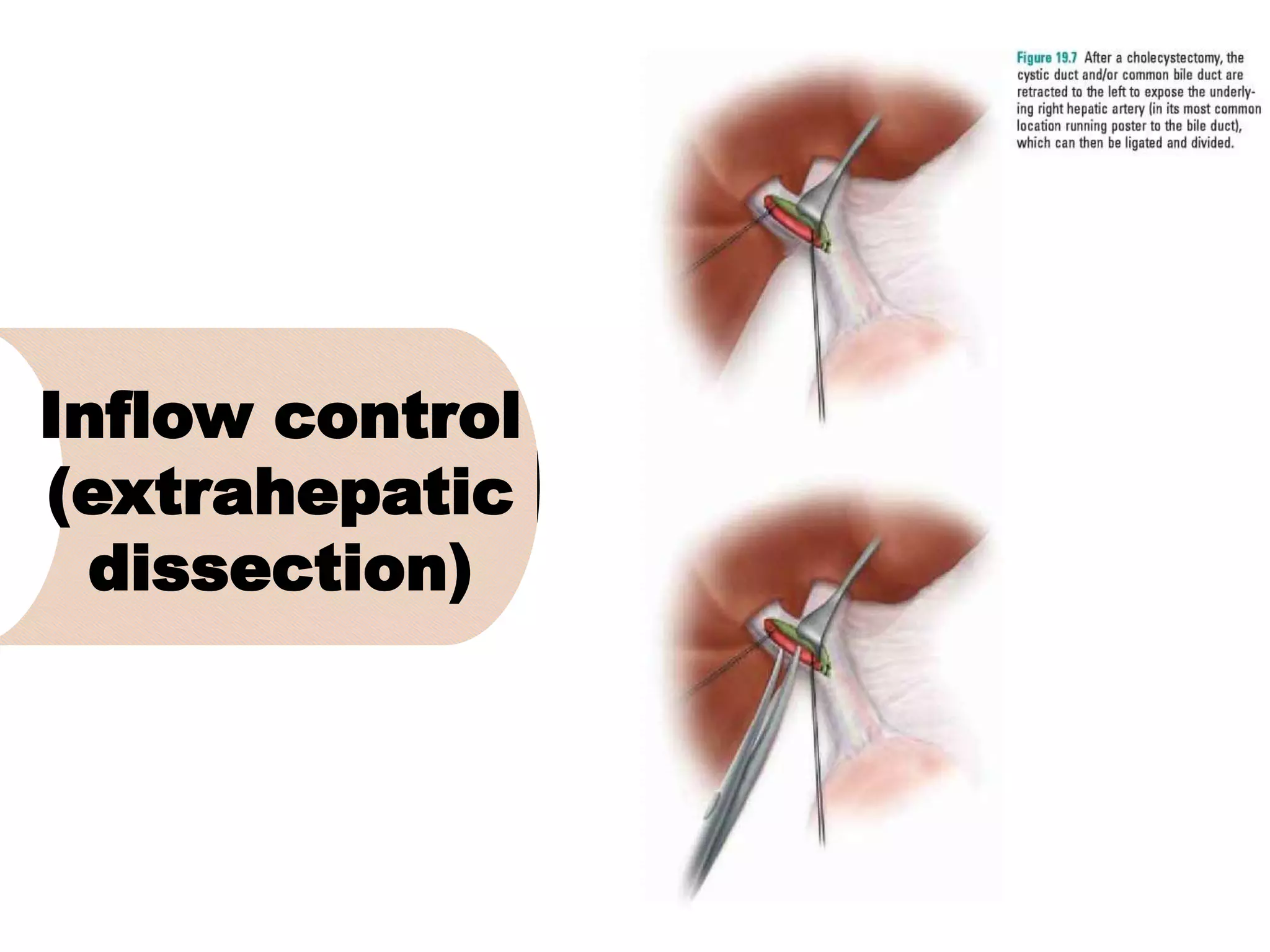
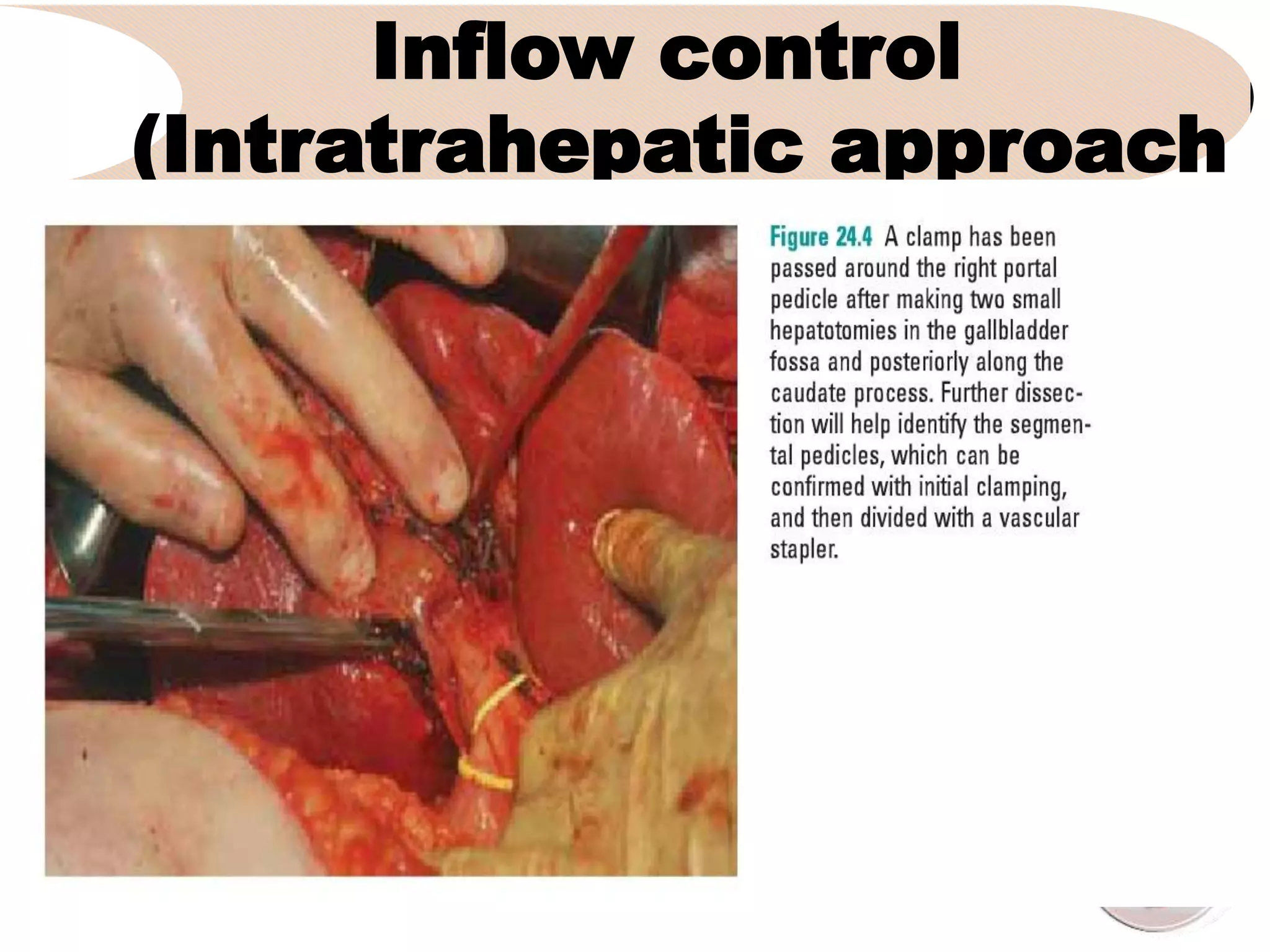
3. It describes the pertinent anatomy of the hepatic artery, portal vein, bile ducts, and hepatic vein that are important to understand for the surgery.
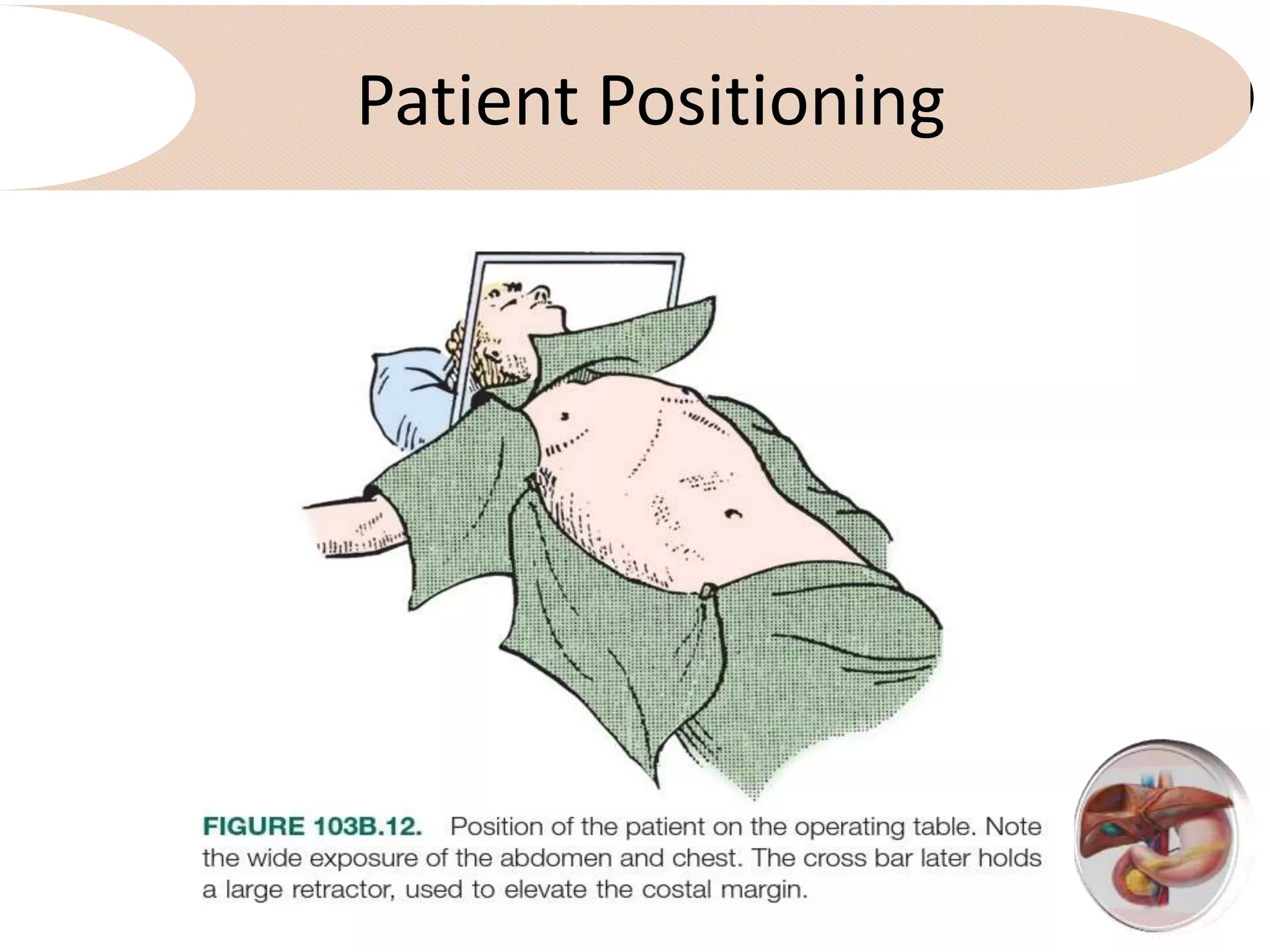
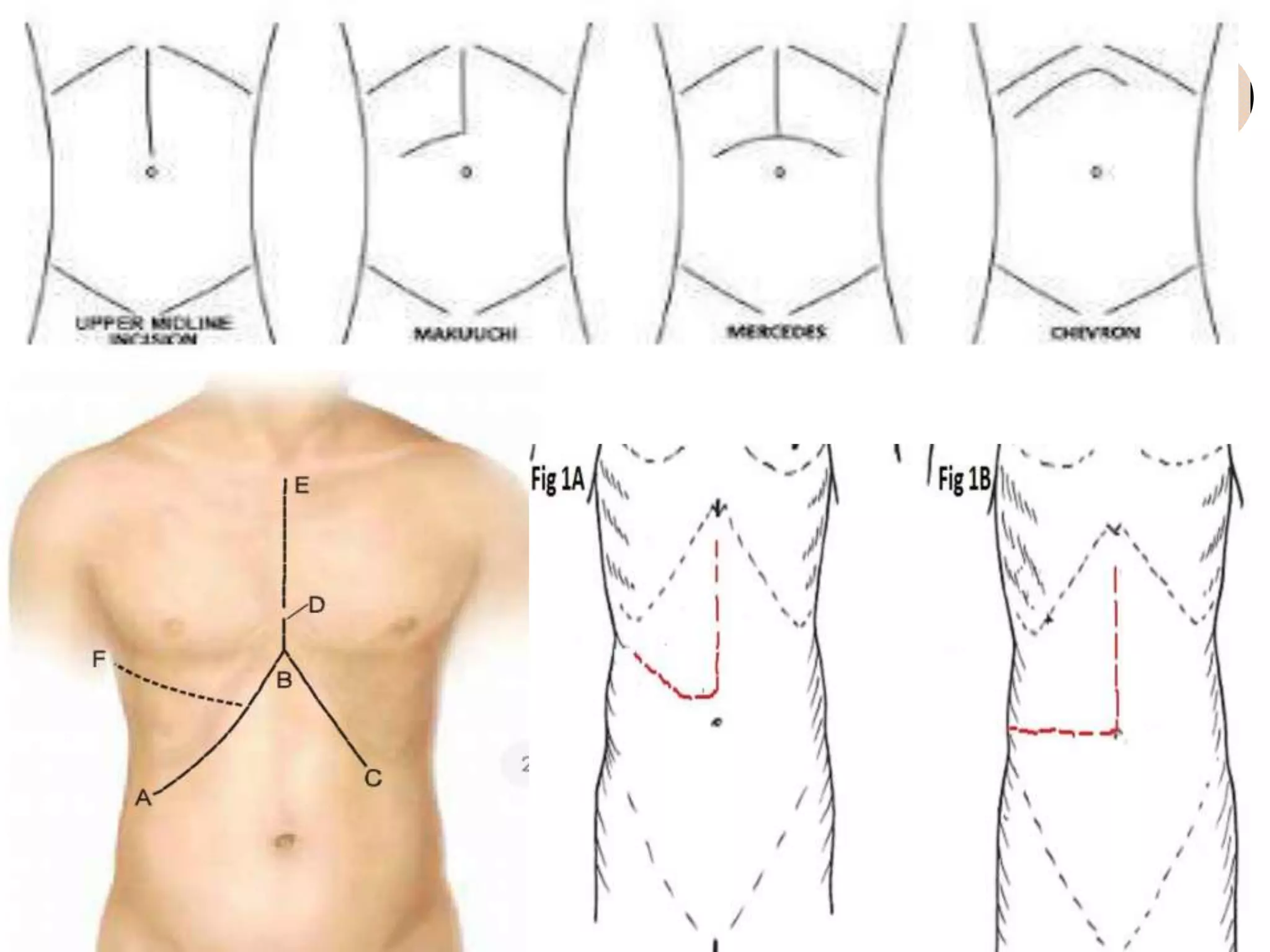
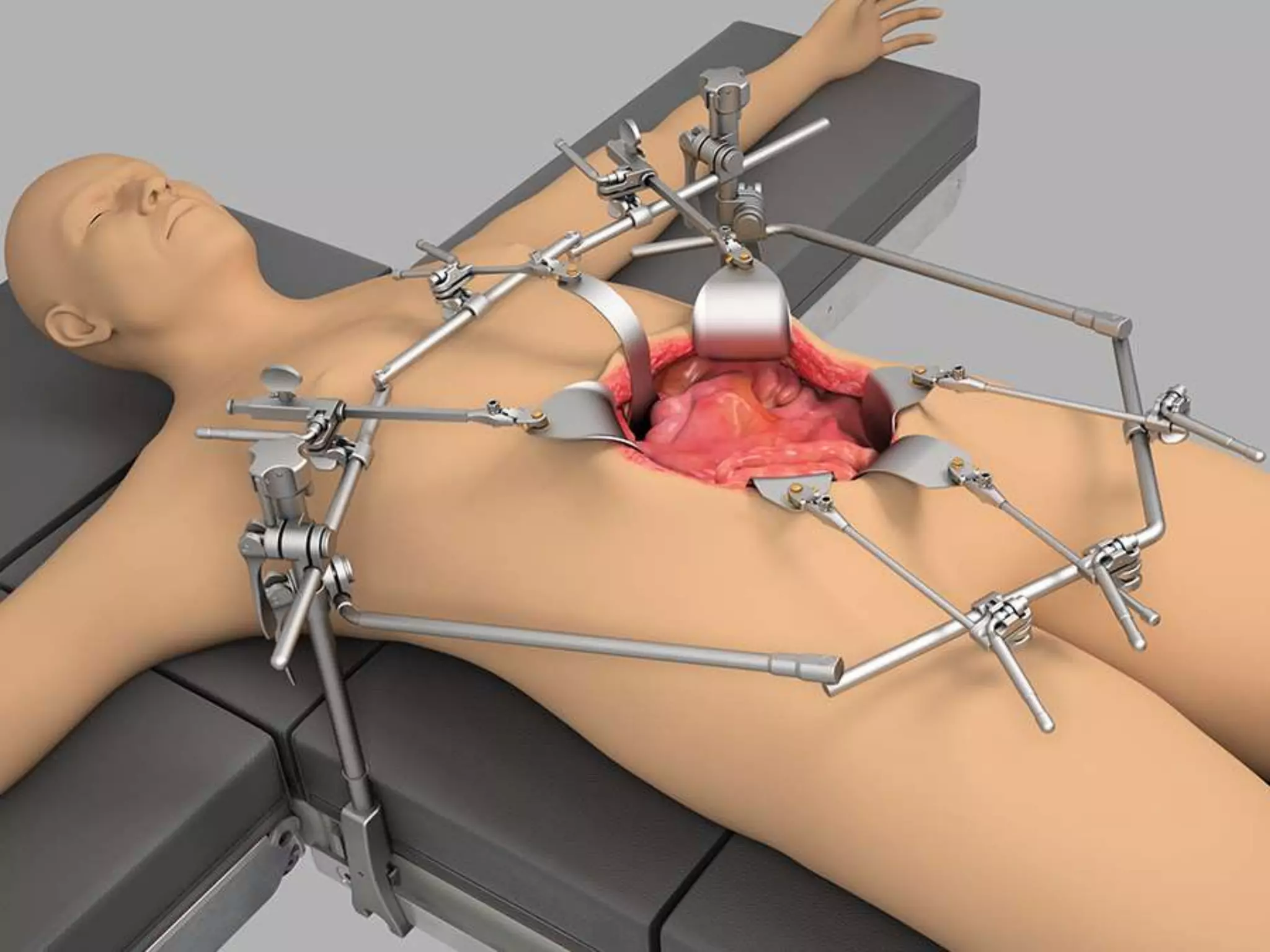
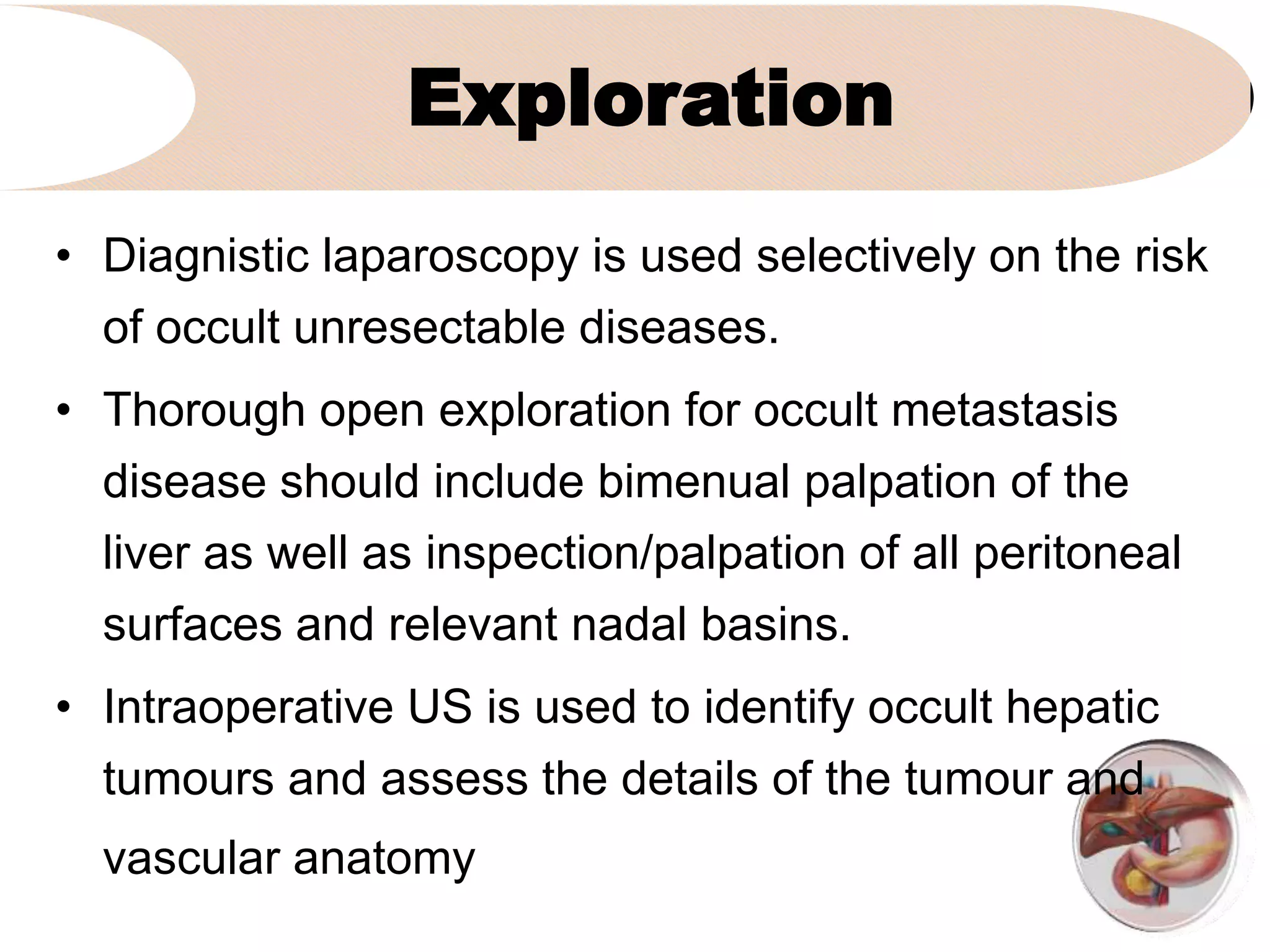
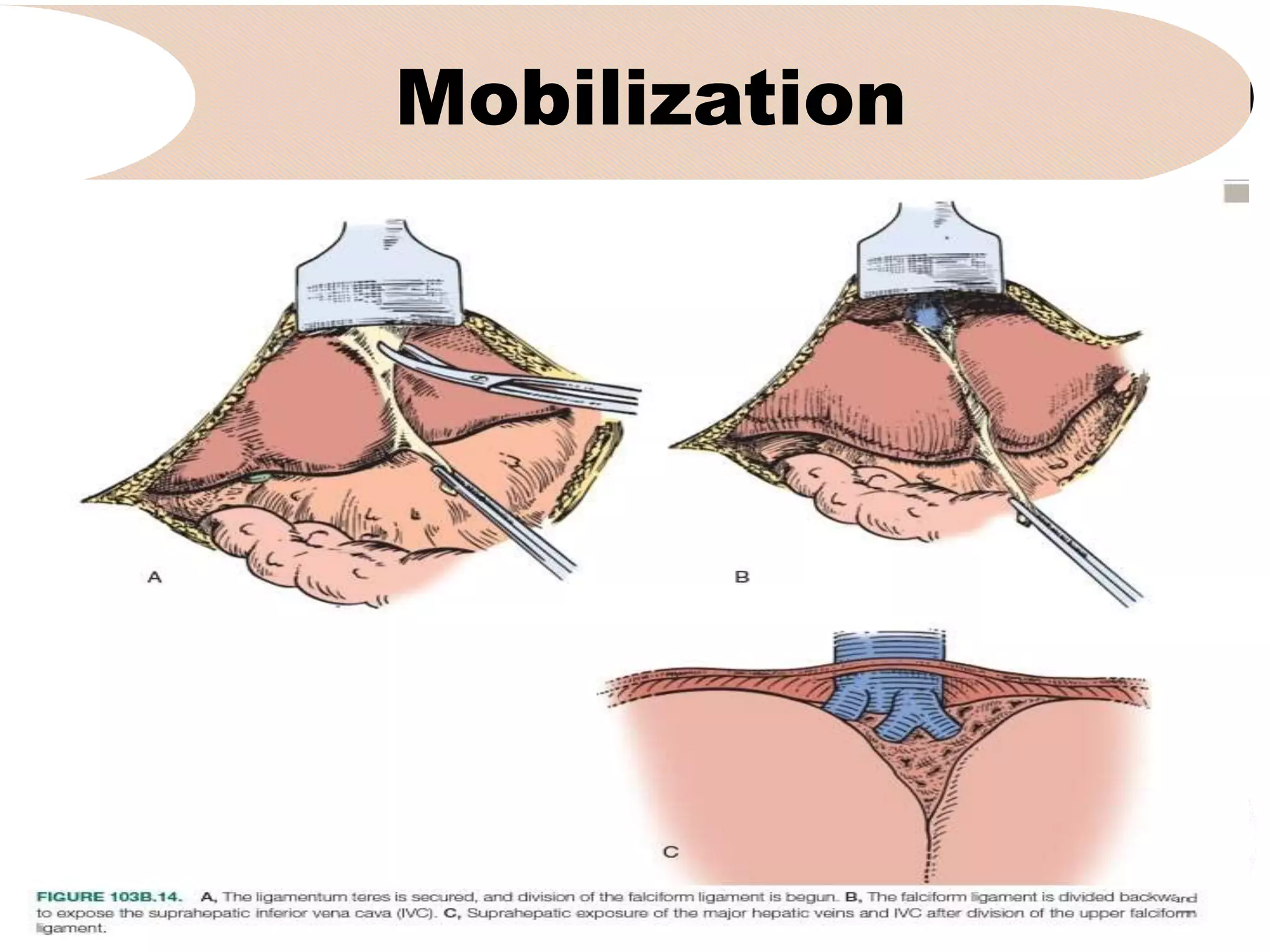
4. It outlines the key steps of the operative procedure including patient positioning, incisions, retraction, exploration, liver mobilization