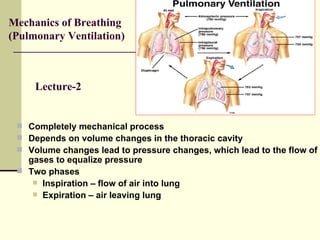
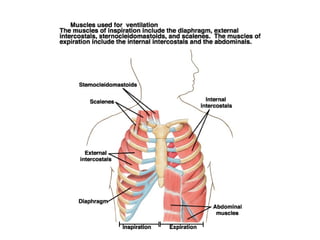
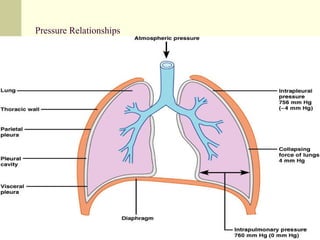
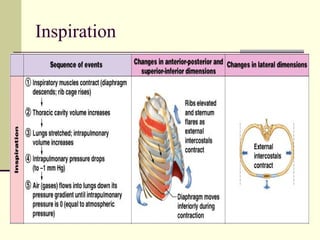
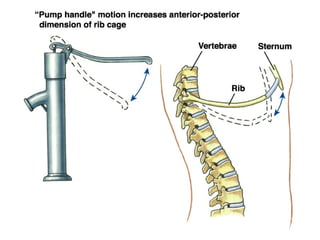
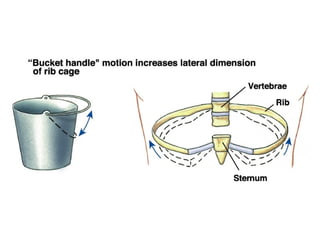
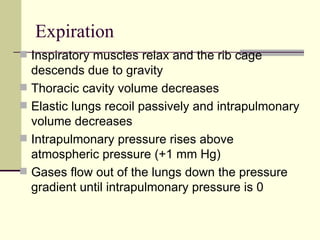
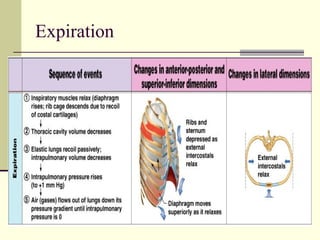
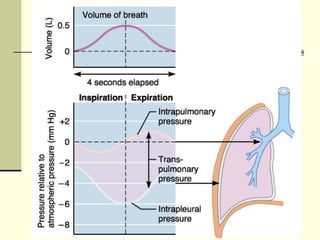
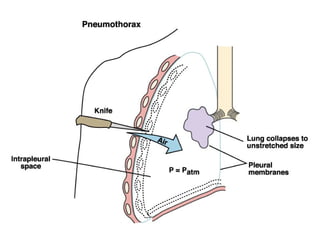
This document discusses the mechanics of breathing and pulmonary ventilation. It is a completely mechanical process that depends on volume changes in the thoracic cavity. These volume changes cause pressure changes, leading to the flow of gases to equalize pressure. There are two phases: inspiration where air flows into the lungs, and expiration where air leaves the lungs. Intrapulmonary pressure and intrapleural pressure fluctuate between these two phases.