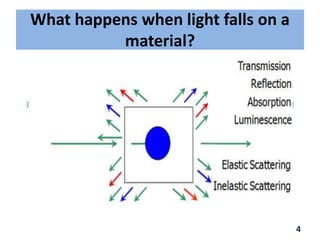
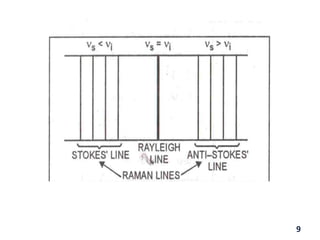
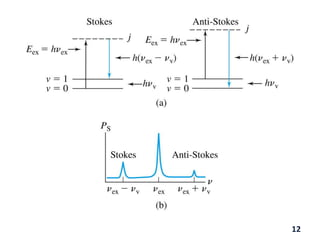
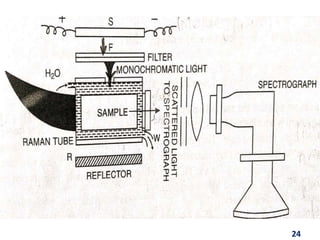
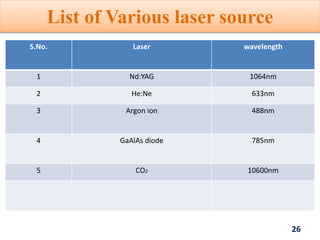
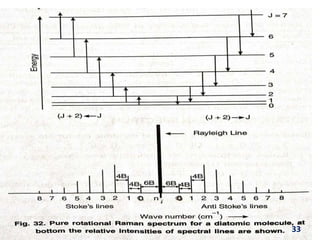
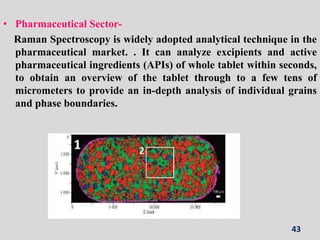
Raman spectroscopy involves the scattering of light by molecules, contrasting with absorption techniques. It differentiates between Rayleigh scattering, Stokes lines, and anti-Stokes lines and has applications across various fields including inorganic, organic, physical, pharmaceutical, forensic, and biological sciences. Despite its advantages, such as the ability to analyze gases, liquids, and solids, it faces limitations like high instrumentation costs and sensitivity to sample quality.