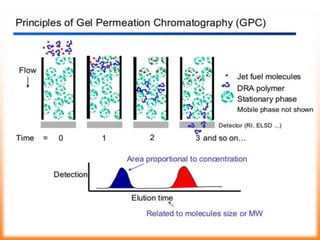
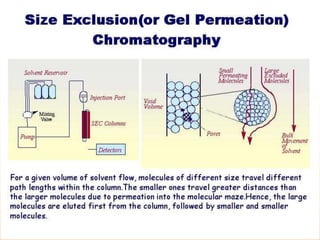
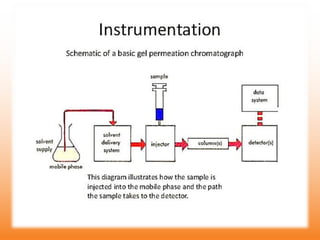
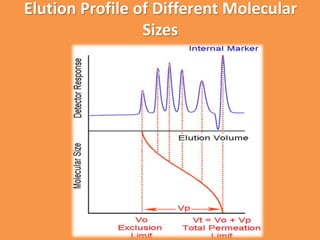
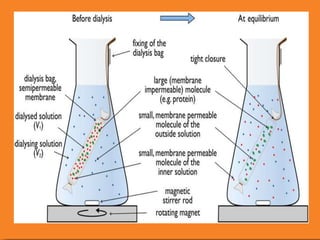
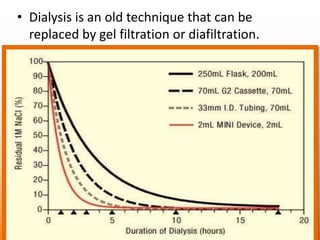
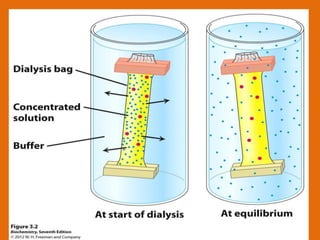
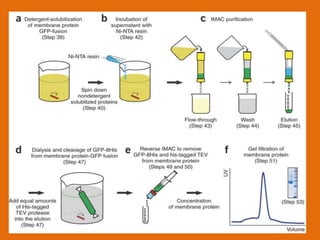
The document discusses product polishing techniques involving gel permeation chromatography (GPC) and dialysis, emphasizing the importance of product safety and purification in biotechnology. GPC separates analytes based on size using a gel and mobile phase, while dialysis removes low molecular weight solutes from solutions using a semi-permeable membrane. Both methods are valuable for characterizing and purifying various substances, although they have distinct advantages and disadvantages.