Oxygen Transfer in Fermentation Processes
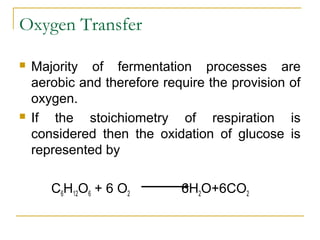
- 1. Oxygen Transfer Majority of fermentation processes are aerobic and therefore require the provision of oxygen. If the stoichiometry of respiration is considered then the oxidation of glucose is represented by C6H12O6 + 6 O2 6H2O+6CO2
- 2. Thus 192 gms of O2 is required for 180 gm glucose. Also oxygen is required for product formation ex: for pencillin production 2.2 gms of oxygen is required for 1g pencillin formation Microorganisms take up the substrate/ nutrient from liquid Hence both O2 and glucose have to be available in liquid Solubility of oxygen in water is 1.26 mmol/l at 25o C OR approx 8 mg/l This will reduce further in the presence of salt or acid This solubility is 6000 times lesser than the solubility of glucose
- 3. Thus it is not possible to supply entire oxygen required for batch fermentation in one addition. Oxygen will be supplied continuously at the rate of demand by the microorganism. Oxygen is consumed quickly in aerobic cultures. For actively respirating yeast with 109 cells /ml oxygen in liquid have to be replaced 12 times in minute to keep up the cellular demand. O2 in the liquid is measured as Dissolved oxygen concentration
- 4. OUR = qo X OUR = oxygen uptake rate qo= specific oxygen uptake rate X = biomass concentration Cell concentration increases oxygen requirement increases Upto certain DO concentration in the liquid the specific oxygen uptake rate increases with DO and remain constant after that. E.coli = 0.008 mmol/l S.cereviseae = 0.004 mmol/l Pencillium sp = 0.022 mmol/l Time qo X qo X qo DO 1
- 5. Flux = resistance X driving force NA = kLa X (C*-C) NA = Oxygen transfer rate kLa = Volumetric Mass transfer coefficient C*-C = Concentration gradient. The concentration gradient is small because of poor solubility of oxygen. Hence it is not easy task. 2
- 6. 1 2 3 4 5 6 7 8 1.1. Transfer from the interior of the bubble to the gas liquid interfaceTransfer from the interior of the bubble to the gas liquid interface 2.2. Movement across the gas liquid interfaceMovement across the gas liquid interface 3.3. Diffusion through relatively stagnant film surrounding the bubbleDiffusion through relatively stagnant film surrounding the bubble 4.4. Transport through the bulk liquidTransport through the bulk liquid 5.5. Diffusion through the relatively stagnant film surrounding the cellsDiffusion through the relatively stagnant film surrounding the cells 6.6. Movement across the liquid cell interfaceMovement across the liquid cell interface 7.7. If the cells are in a floc, clump diffusion through the solid to theIf the cells are in a floc, clump diffusion through the solid to the individual cellindividual cell 8.8. Transport through the cytoplasm to the site of the reactionTransport through the cytoplasm to the site of the reaction Note: Resistance due to the gas boundary layer on the inside of the bubble has been neglected. If the cells are individually suspended step 7 disappears.
- 7. Magnitude of various mass transfer resistance depend on the composition and rheological properties of the liquid, mixing intensity, bubble size, cell clump size, etc., For most of the bioreactors the following analysis is valid Step 1 – relatively fast Step 2 – negligible resistance Step 3- major resistance
- 8. Step 4 – In well mixed fermenter concentration gradients in the bulk liquid are minimised and resistance is small. However rapid mixing is affected in the viscous broths. In this case the bulk liquid resistance is important example: Xanthan gum production, high viscous mycelial fermentation etc.,
- 9. Step 5: Because single cells are much smaller than the gas bubbles, the liquid film surrounding each cell is much thinner than that around the bubbles and its effect on mass transfer can generally be neglected. On the other hand if the cells form large clumps, liquid film resistance can be significant example Citric acid production. A.niger form pellets.
- 10. Step 6 – Very small Step 7 – When the cells are in clumps, intraparticle resistance is significant as oxygen has to diffuse through the solid pellet. Magnitude of this depend on the size of the clumps. Step 8 – Intracellular transfer resistance is small since distance is very small.
- 11. At steady state the rate of oxygen transfer from the bubbles must be equal to the rate of oxygen consumption by the cells. Equations 1 and 2 will be equal. kLa (C*-C)= qo X kLa is used to characterize the oxygen mass transfer capability If it is small then the ability of the reactor to deliver oxygen is small. At steady state if stirrer speed is increased i.e kLa is increased (Raising stirrer speed will reduce the thickness of the boundary layer surrounding the bubble) the dissolved oxygen concentration increases At the same time if the cell concentration is increased at constant kLa the DO will decrease 3
- 12. Equation 3 can be used for deriving relationships for fermenters. For a given set of operating conditions the maximum rate of oxygen transfer occurs when the driving force is maximum. i.e. C*-C is highest. In otherwords when C = 0 Sub in eqn 3 for obtaining maximum cell concentration supported by the reactor Xmax = kLa C*/qo If Xmax is lower than the required cell concentration in fermentation then kLa should be improved. (kLa)crit =qoX/(C*-Ccrit)
- 13. FACTORS AFFECTING OXYGEN TRANSFER OTR ∝ kL a (C*-C)
- 14. Bubbles Efficiency of OTR depends to large extent on the characteristic of bubble Bubble behaviour mostly affect kLa In stirred fermenter air is sparged under the impeller In lab fermenters very good mixing will be there hence bubbles in the system are frequently subject to distortions (10-20 KW/m3 ) In contrast in industrial fermenters most of the time bubbles are freely floating after initial dispersion (0.5 – 5 KW/m3 ) Reason P/V value are low in large fermenters
- 15. The most important property of air bubbles in fermenter is its size Smaller the size Greater is the interfacial area a Slow bubble rise velocity and stays for longer time in fermenter giving more time for oxygen transfer Create high gas holdup (ε) ε = VG/(VL+VG) VG – Vol of gas bubbles in reactor VL – Vol of liquid
- 16. Interfacial area largely depend on gas holdup. It varies between 0.01 to 0.2 One side it is desirable to have smaller bubbles. But bubbles less than 1 mm dia can become nuisance in bioreactors O2 concentration in the bubbles immediately transferred to medium and attains equilibrium. These bubbles staying in the reactor is of no use.
- 17. Bubble size also affects kL Bubbles less than 2-3 mm dia acts as rigid spheres. This lowers kL values On the other hand bubbles greater in size have relatively mobile surfaces. These bubbles are able to wobble and move in spirals during free rise Due to this they have beneficial effect 2-3 mm bubbles 3-4 X 10-4 m/s kL values Bubble size reduced kL reduces to 1 X 10-4 Above 3 mm kL values are constant
- 18. Air bubbles are formed at the sparger Sparger design varies from open pipe, porous diffusers, perforated pipes and complex injectors Normally air flow rates used are 0.5 to 1.5 vvm The effect of air flow rate on kLa values is given in the diagram Aeration and agitation 0.50 1.0
- 19. This air flow rate is maintained during scaleup If the impeller is unable to disperse the incoming air then OTR decreases extremely due to impeller flooding. Flooding is the phenomena where the air flow dominates the flow pattern Air velocityStirrer speed
- 20. The degree of agitation played major role in oxygen transfer Agitation increases the air available by dispersing the air in the culture in the form of bubbles It delays escape of air bubbles from the system It prevents coalescence of bubbles to bigger ones. It decreases the thickness of liquid film at the gas liquid interface by creating turbulence in the culture
- 21. To avoid flooding minimum impeller tip speed for dispersion of air bubbles 1.5 – 2.5 m/s Flooding could be avoided if F/ND3 < 0.3 N2 D/g F – Volumetric air flow rate N – stirrer speed D – stirrer diameter g – accelaration due to gravity
- 22. Viscosity changes flow properties such as surface tension etc will affect kLa Increase in viscosity decreases kLa Increase in viscosity may occur due to biomass in case of fungal mycelia formation or some products such as polysachharides production Broth Viscosity
- 23. High degree of aeration and agitation will result in foam formation. Foaming reduces oxygen transfer. Air bubbles entrapped in the foam and again and again they recirculate in the medium. This will result in oxygen depleted bubbles residing in the system To control foam antifoam agents are added. Antifoam agents
- 24. Most of the antifoams are surface tension lowering substances This will result in rigid bubble formation and resistance to oxygen transfer. Also antifoams in the liquid may favour coalescence of bubbles in freely moving areas which again will decrease oxygen transfer. OTR can be reduced dramatically even by factor of 10.
- 25. Normally salts suppress the coalescence of bubbles hence it favors OTR Increase in suspended solids will decrease OTR in High cell density cultivation Temperature increase beyond 40 o C will decrease oxygen solubility hence OTR Vessel geometry will influence OTR. If H/D ratio is more bubble residence time is more and hence OTR may increase. Other factors
- 26. kLa and Power consumption A large number of empirical relationships have been developed between kLa, power consumption and superficial gas velocity kLa = k(P/V)x Vs y P –Power absorption V- Volume of the reactor Vs – Superficial airvelocity k,x,y – empirical constants
- 27. Value of x is dependant on the size of the vessel Laboratory it is 0.95 Pilot plant – 0.67 Production fermenter – 0.5 k – 0.026 x- 0.4 y- 0.5
- 28. Major Factors in ScaleupMajor Factors in Scaleup Inoculum developmentInoculum development SterilizationSterilization Environmental parametersEnvironmental parameters Nutrient availabilityNutrient availability pHpH TemperatureTemperature Dissolved oxygen concentrationDissolved oxygen concentration Shear conditionsShear conditions Dissolved CODissolved CO22 concentrationconcentration Foam productionFoam production
- 29. AerationAeration AgitationAgitation COCO22 Bulk mixingBulk mixing FoamFoam CostCost ShearShear OO22
- 30. Steps in scaleupSteps in scaleup Identification of the principalIdentification of the principal environmental domain affected by theenvironmental domain affected by the aeration and agitationaeration and agitation Identification of the process variable whichIdentification of the process variable which affects the identified environmentalaffects the identified environmental domaindomain Calculation of the value of the processCalculation of the value of the process variable to be used on the large scalevariable to be used on the large scale
- 31. Process VariableProcess Variable CharacteristicsCharacteristics affectedaffected Power consumptionPower consumption per unit volumeper unit volume Oxygen Transfer RateOxygen Transfer Rate Impeller tip speedImpeller tip speed Shear RateShear Rate Volumetric air flow rateVolumetric air flow rate Oxygen Transfer RateOxygen Transfer Rate Pumping ratePumping rate Mixing timeMixing time Reynolds numberReynolds number Heat transferHeat transfer
- 32. Criterion usedCriterion used in scale upin scale up from 80 tofrom 80 to 10000 l10000 l Effect on the operating conditionsEffect on the operating conditions on the large scaleon the large scale PP P/VP/V FlowFlow min-1 vol-1min-1 vol-1 NDiNDi P/VP/V 125125 1.01.0 0.340.34 1.71.7 FlowFlow min-1 vol-1min-1 vol-1 31253125 25.025.0 1.01.0 5.05.0 NDiNDi 2525 0.20.2 0.20.2 1.01.0 ReynoldsReynolds numbernumber 0.20.2 0.00160.0016 0.040.04 0.20.2
- 33. Scale down methodScale down method Medium designMedium design Medium sterilizationMedium sterilization Inoculation proceduresInoculation procedures Number of generationsNumber of generations MixingMixing Oxygen transfer rateOxygen transfer rate
