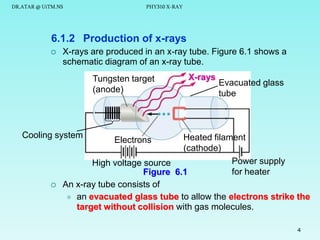
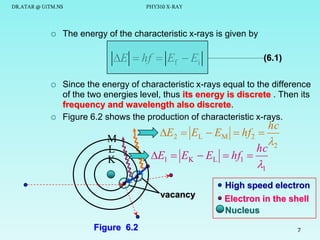
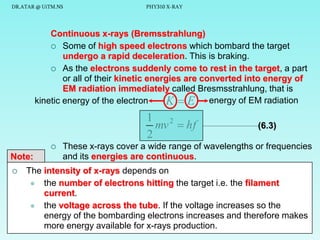
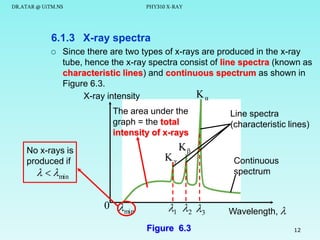
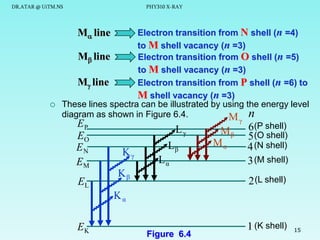
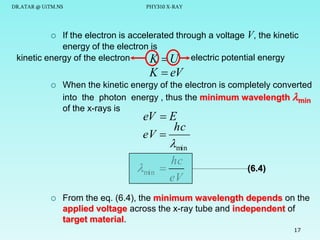
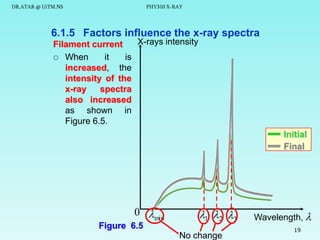
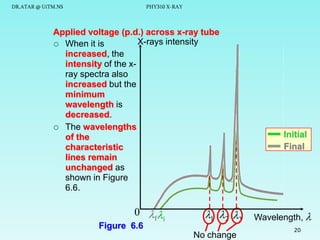
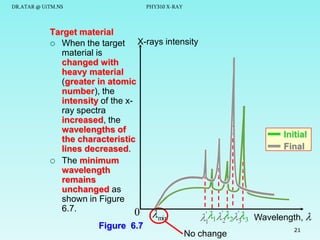
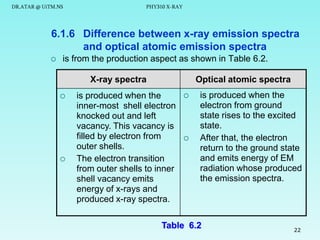
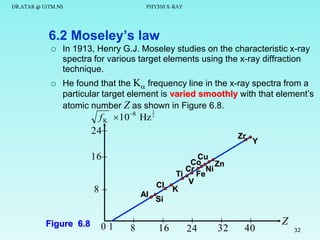
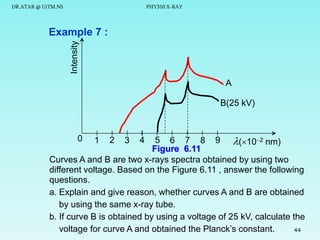
This document discusses x-rays and their production and spectra. It begins by defining x-rays as electromagnetic radiation produced when high-energy electrons bombard atoms. X-rays were discovered in 1895 by Wilhelm Röntgen. The document then discusses how x-rays are produced in an x-ray tube, where high-speed electrons are accelerated toward a metal target. This produces two types of x-rays: characteristic x-rays formed by electron transitions within atoms, and continuous x-rays formed when electrons are decelerated. The x-ray spectrum consists of characteristic line spectra and a continuous spectrum. It provides formulas for calculating x-ray energies and wavelengths.