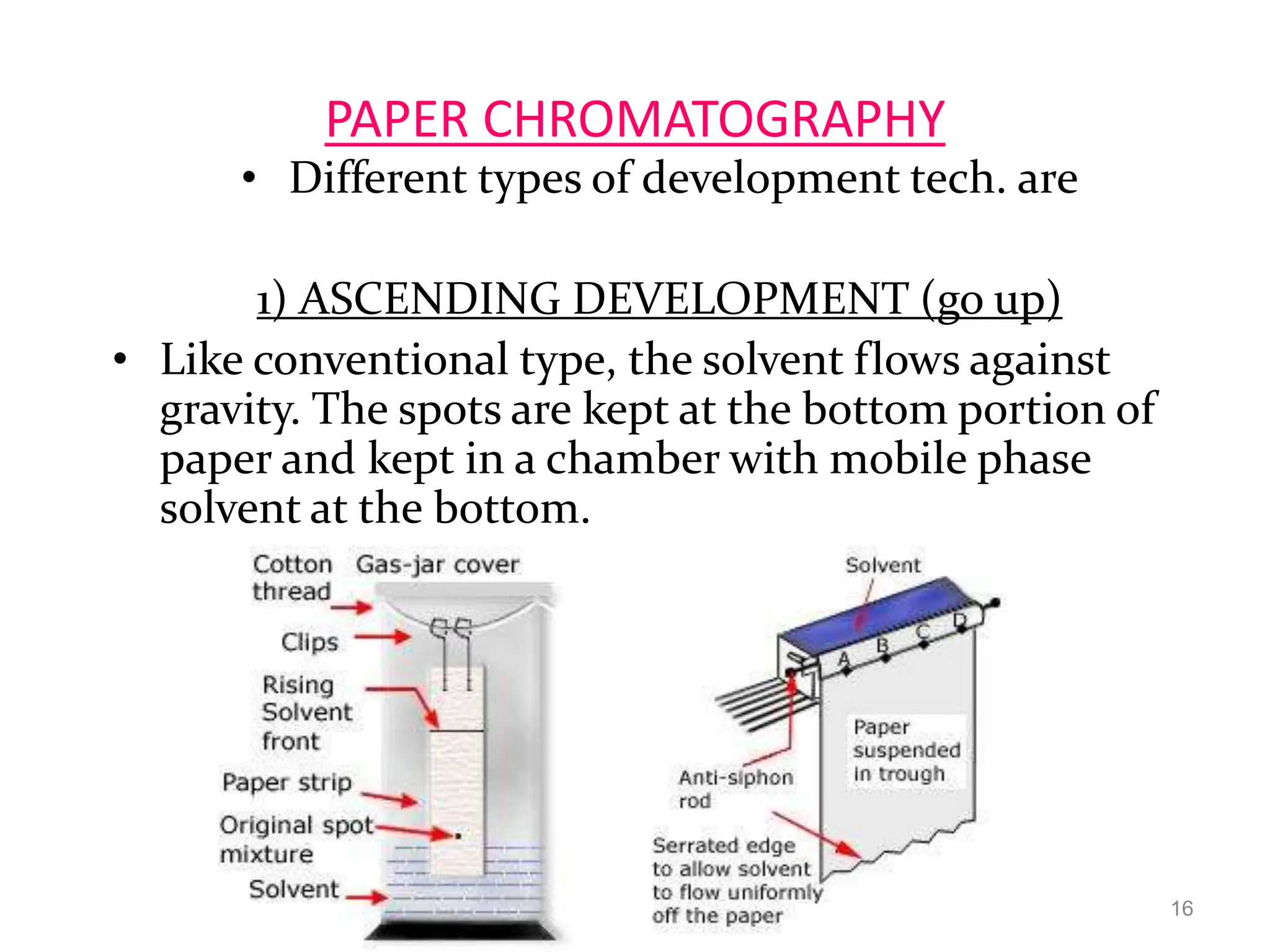
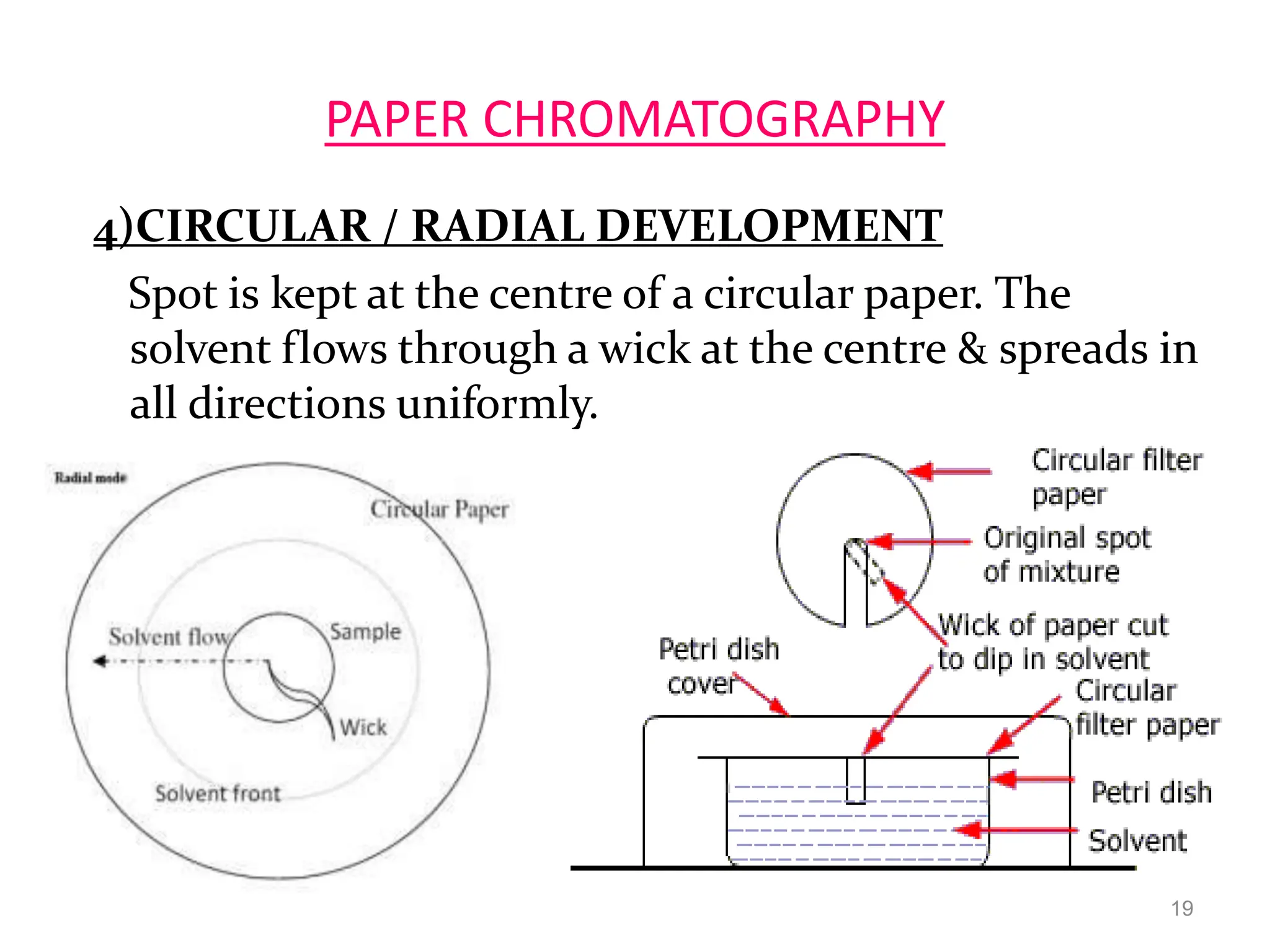
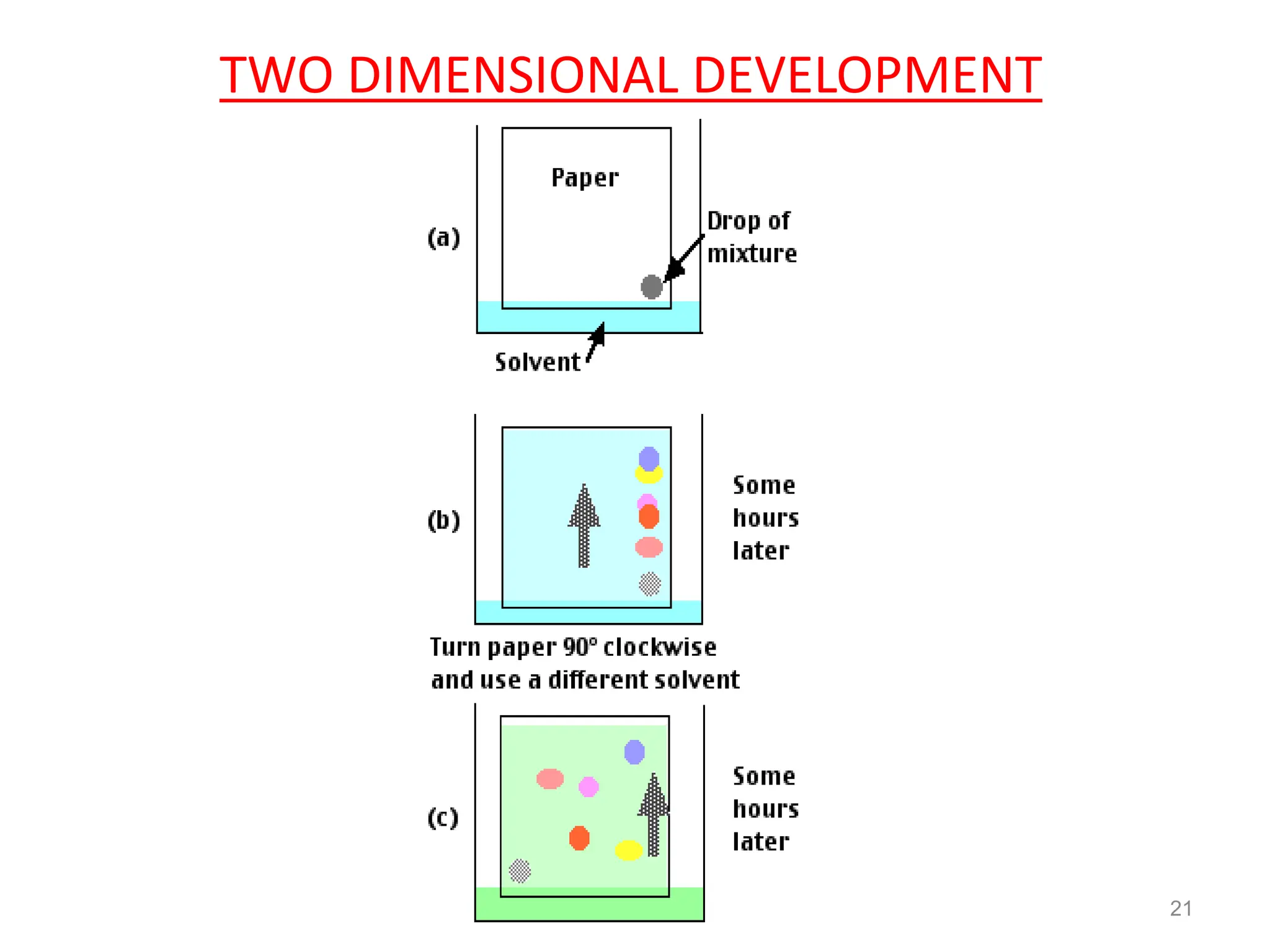
Paper chromatography is a technique used to separate and analyze mixtures that relies on the differential migration of compounds across paper. It works on the principle that some compounds interact more with the stationary phase (paper) and others interact more with the mobile phase (solvent). The sample is applied to the paper and then placed in a solvent, allowing the compounds to separate into distinct spots. Paper chromatography is simple, inexpensive, and has good resolving power, making it useful for analyzing mixtures like drugs, metabolites, proteins, and more.