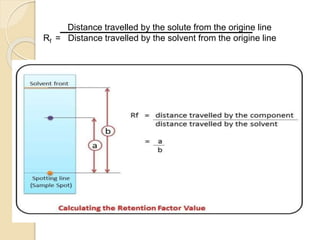
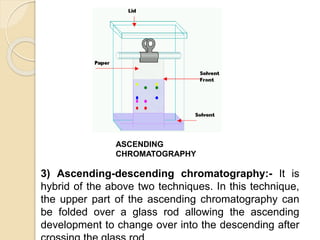
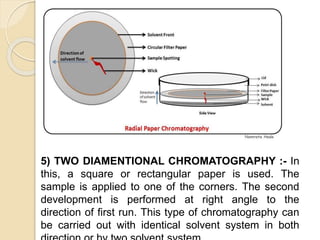
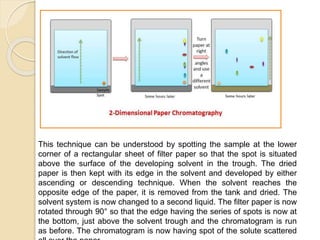
The document provides a comprehensive overview of paper chromatography, covering its principles, types, methodologies, and applications. It explains the importance of factors like the choice of filter paper, development solvent, and spotting techniques in achieving effective separation of substances. Additionally, it highlights the various techniques used in paper chromatography, such as ascending, descending, and two-dimensional chromatography, as well as their practical applications in analyzing organic and biochemical products.