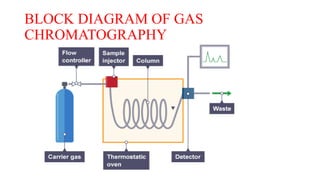
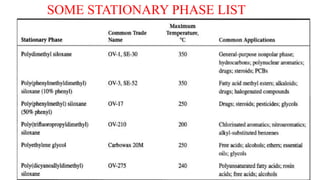
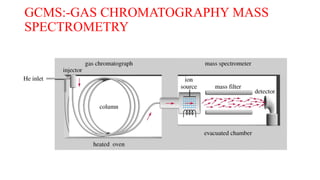
This document discusses gas chromatography. It begins by defining chromatography and describing different chromatography techniques. It then focuses on gas chromatography, explaining that the mobile phase is a carrier gas and the stationary phase is a liquid or polymer coating inside a column. Key components of a gas chromatograph are described, including the carrier gas, injector, column, temperature control, stationary phases, and detectors. The document discusses how gas chromatography can be used for qualitative analysis of compounds and lists some advantages and disadvantages. It concludes by mentioning gas chromatography-mass spectrometry as a modern approach.