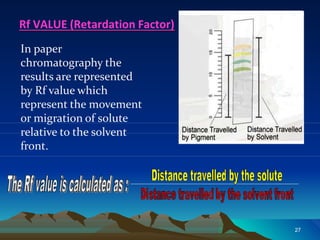
Paper chromatography is a widely used chromatographic technique for isolating, identifying, and quantifying organic and inorganic compounds. It involves the flow of solvents on specially designed filter paper, with two main types: paper adsorption chromatography and paper partition chromatography. The method includes various development techniques and detection methods, offering advantages like simplicity and rapid analysis.