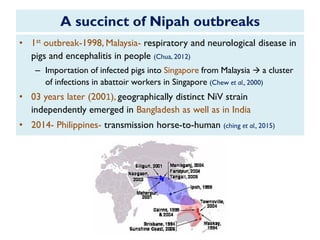
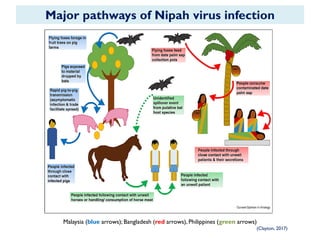
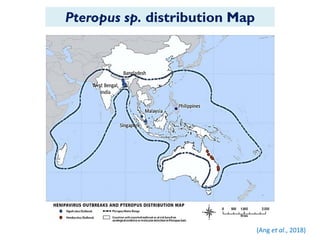
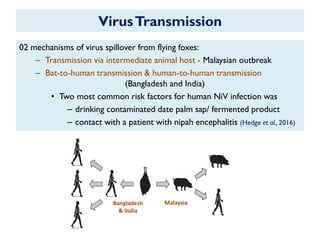
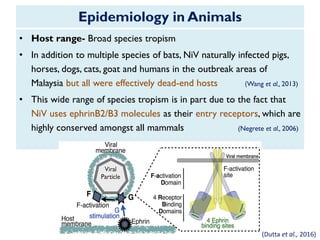
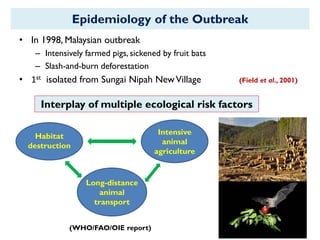
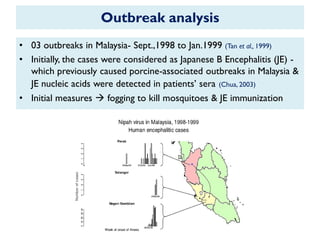
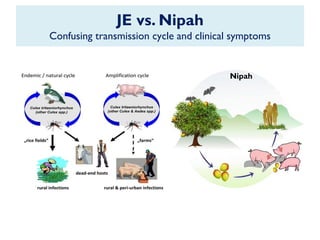
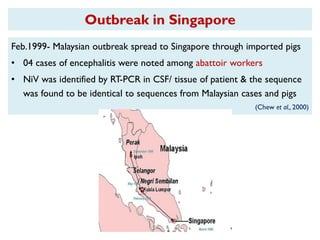
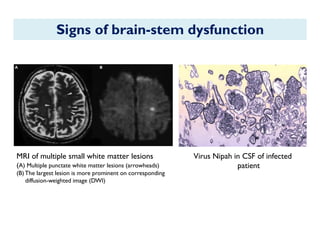
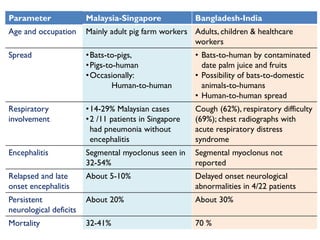
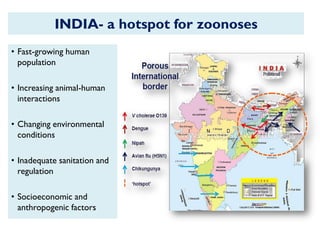
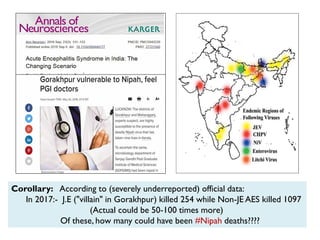
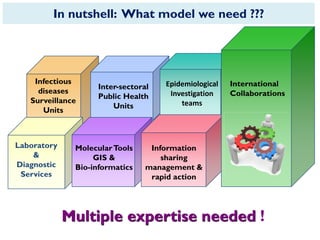
The document provides an overview of Nipah virus, detailing its emergence, characteristics, and outbreaks in various countries, particularly focusing on human and animal health impacts. It discusses the virus's transmission mechanisms from flying foxes to pigs and humans, as well as the severe neurological symptoms associated with infection. Additionally, it highlights key outbreaks in Malaysia, Bangladesh, and India, the epidemiological risks, clinical signs, and treatment approaches for affected individuals.