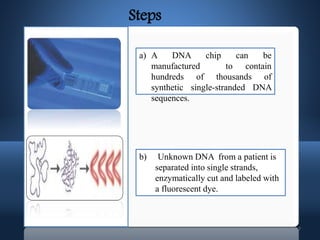
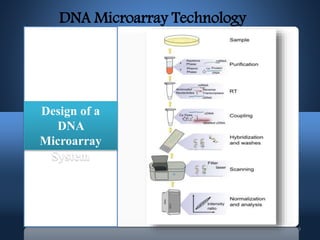
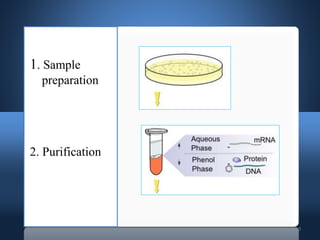
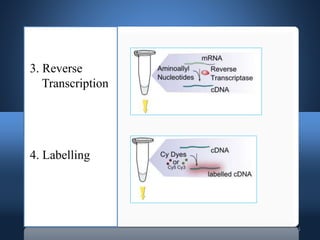
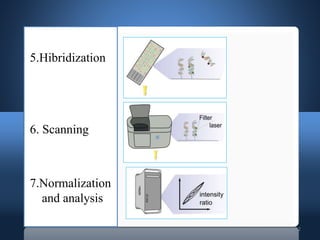
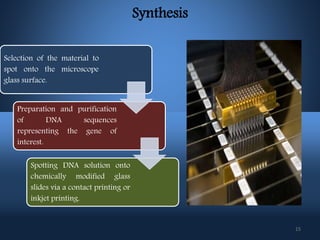
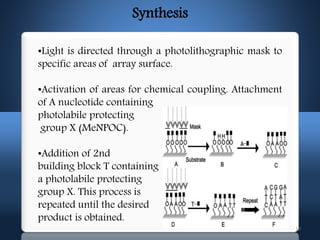
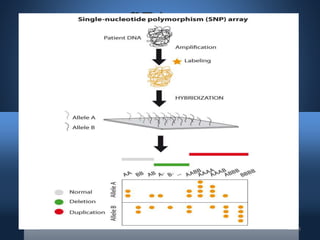
DNA microarrays, also known as DNA chips, allow researchers to analyze large numbers of DNA sequences simultaneously. They contain thousands of unique DNA probes immobilized on a solid surface in an organized grid pattern. Unknown DNA samples are labeled with fluorescent dyes and hybridized to the probes on the chip. The bound DNA is then detected using lasers and analyzed by a computer to determine which genes are present or expressed in the sample. There are two main types of DNA chips - cDNA-based chips containing amplified cDNA probes, and oligonucleotide-based chips containing short, synthesized DNA sequences. DNA microarrays have many applications including gene expression profiling, comparative genomics, disease diagnosis, and drug discovery.