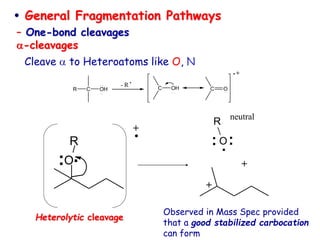
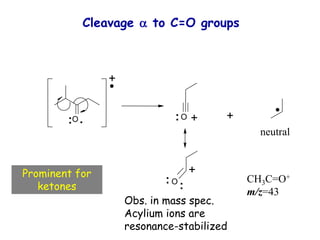
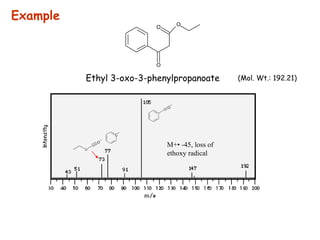
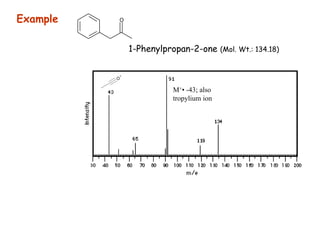
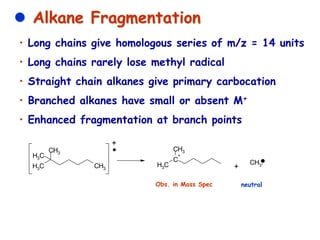
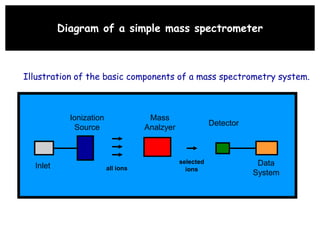
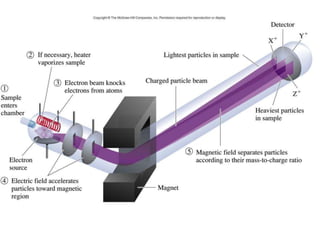
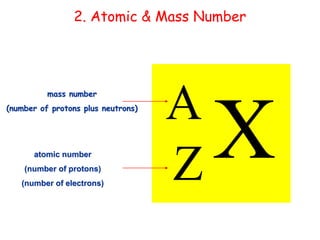
Mass spectrometry is an analytical technique used to separate molecular species by their mass, providing insights like molecular weight, structure, and isotopic distribution. It is applied in various fields such as pharmaceutical analysis, environmental testing, and forensic science. Mass spectrometry involves ion generation, acceleration, analysis, and detection to produce a spectrum that reveals the abundance of ions relative to their mass-to-charge ratio.









![What’s in a Mass Spectrum?
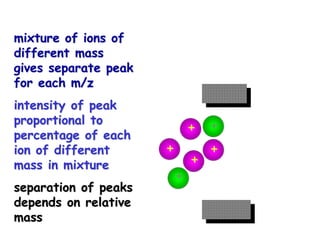
Mass, as m/z. Z is the charge, and for doubly charged ions (often seen in
macromolecules), masses show up at half their proper value
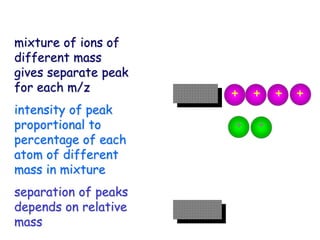
High
mass
[M+H]+(CI)
Or M•+ (EI)
“molecular ion”
Unit mass
spacing
Fragment Ions Derived from
molecular ion
or higher
weight
fragments
In CI, adduct ions,
[M+reagent gas]+](https://image.slidesharecdn.com/finalmassspectroscopy-230607205059-9d1980ec/85/MASS-SPECTROSCOPY-ppt-14-320.jpg)










![In
ten
sity
(%)
0
20
40
60
80
100
Mass [amu]
111.95 112.00 112.05 112.10
In
ten
sity
(%)
0
20
40
60
80
100
Mass [amu]
111.95 112.00 112.05 112.10
In
ten
sity
(%)
0
20
40
60
80
100
Mass [amu]
111.95 112.00 112.05 112.10
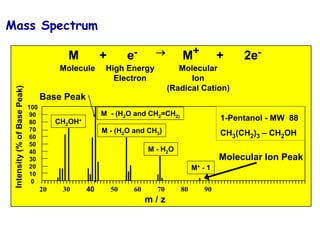
RP= 3,000 RP= 5,000 RP= 7,000
All resolving powers are FWHM
C6H5OF
C6H5Cl
Resolving Power Example](https://image.slidesharecdn.com/finalmassspectroscopy-230607205059-9d1980ec/85/MASS-SPECTROSCOPY-ppt-31-320.jpg)

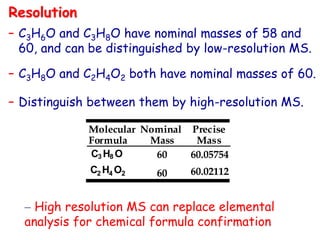
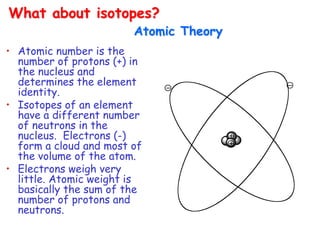

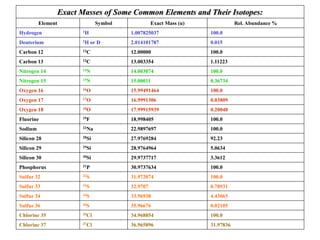
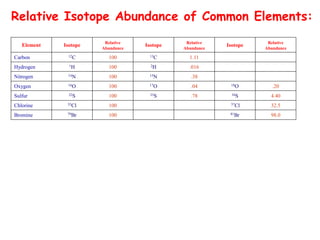
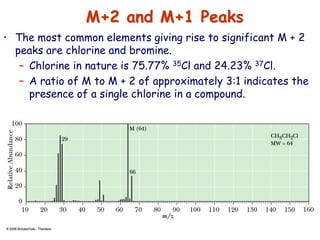
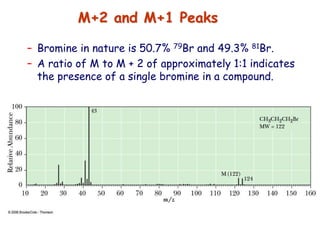


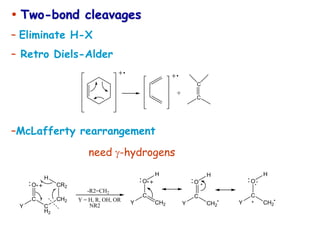
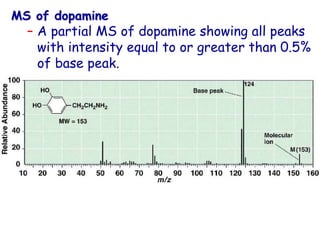
![• Better carbocation wins and predominates “Stevenson’s Rule”
[M·]+ A+ + B· (neutral)
or
B+ + A·
EI
Fragmentation
Stevenson’s Rule:
– For simple bond cleavage, the fragment with lowest
ionization potential takes the charge
(in other words, the most stable ion is formed)](https://image.slidesharecdn.com/finalmassspectroscopy-230607205059-9d1980ec/85/MASS-SPECTROSCOPY-ppt-42-320.jpg)

![The “Even Electron Rule” dictates that even (non-radical)
ions will not fragment to give two radicals (pos• + neutral•)
(CI)
CI
[M+H]+ PH+ + N (neutral)
– Loss of neutral molecules, small stable, from MH+
– Loss of neutrals from protonated fragments
– Subsequent reprotonation after a loss
– Typically there is no ring cleavage (needs radical) or two
bond scissions.
– Depends highly on ion chemistry specifically acid-base
(proton affinities)](https://image.slidesharecdn.com/finalmassspectroscopy-230607205059-9d1980ec/85/MASS-SPECTROSCOPY-ppt-44-320.jpg)