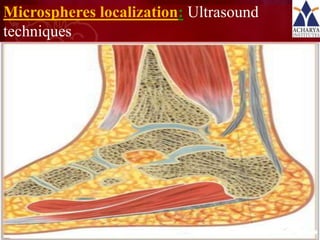
Magnetic microspheres are small particles containing magnetite that can be guided to target tissues using external magnetic fields. They allow drugs to be delivered at lower doses directly to tissues while avoiding toxicity. Microspheres are prepared using methods like phase separation or solvent evaporation to encapsulate drugs and magnetite in carriers like albumin. Particle characteristics and drug release kinetics can be evaluated using techniques like dialysis, imaging, and microscopy. Magnetic microspheres show potential for targeted drug delivery but also have technical challenges for approval.