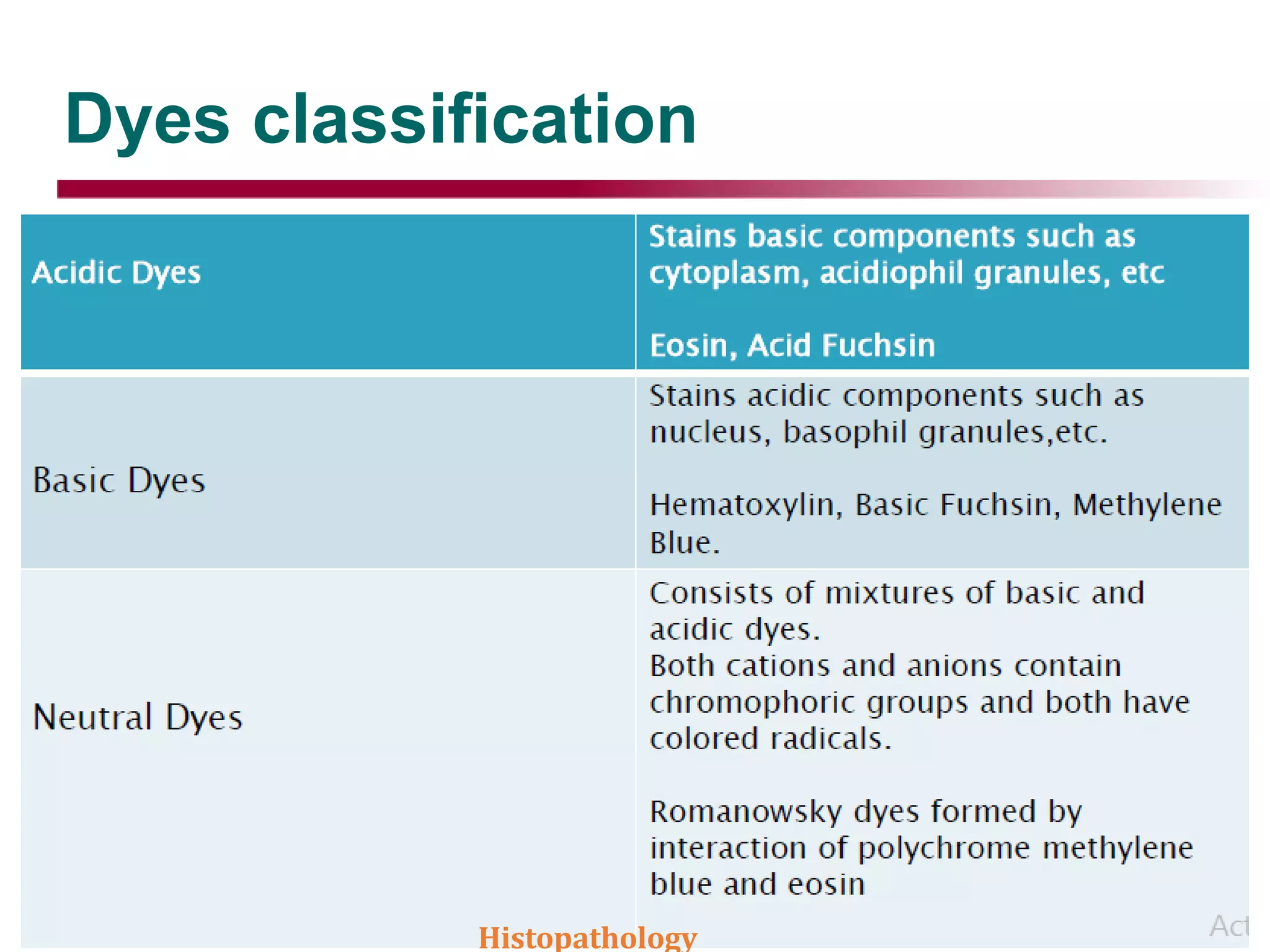
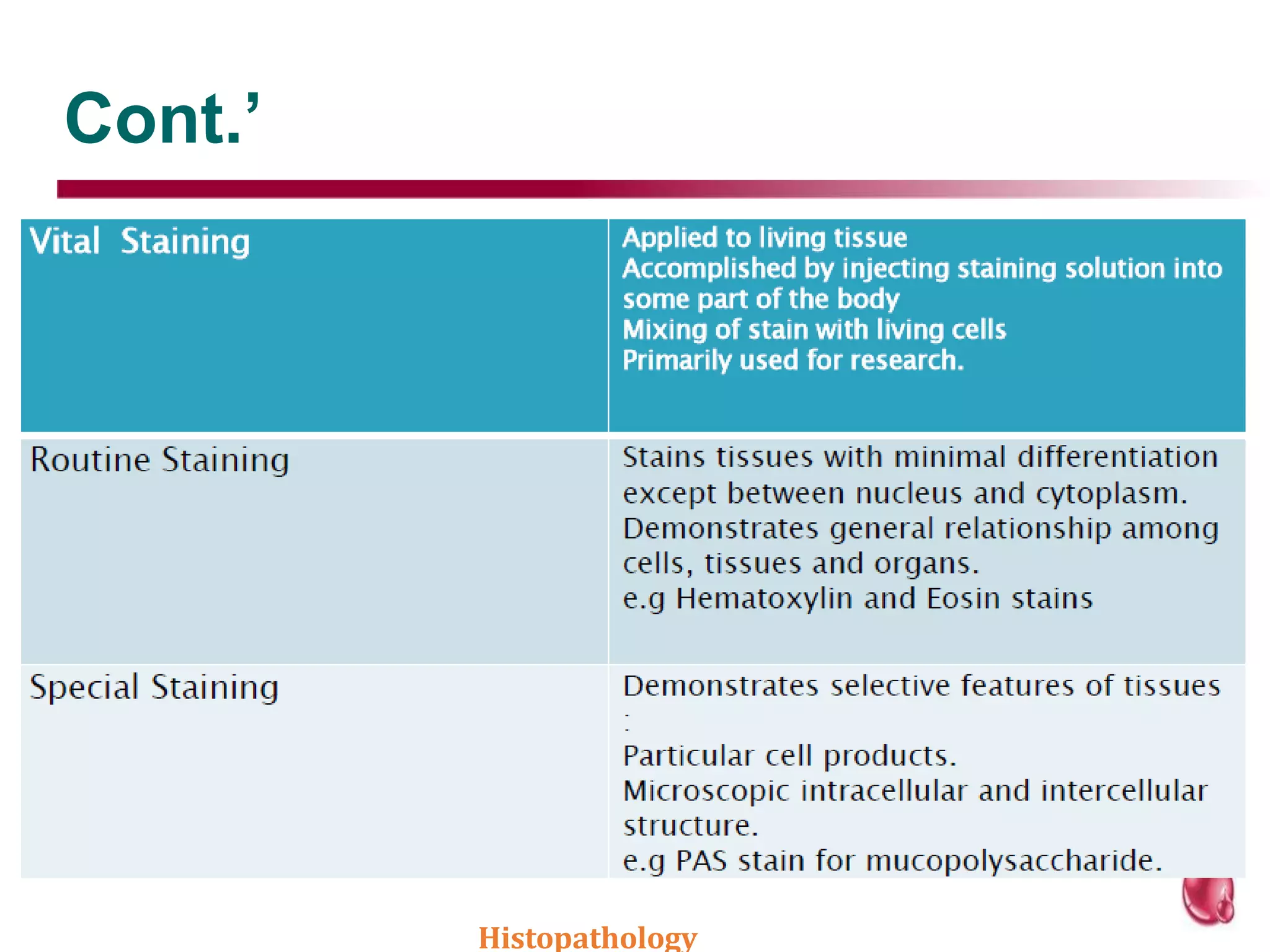
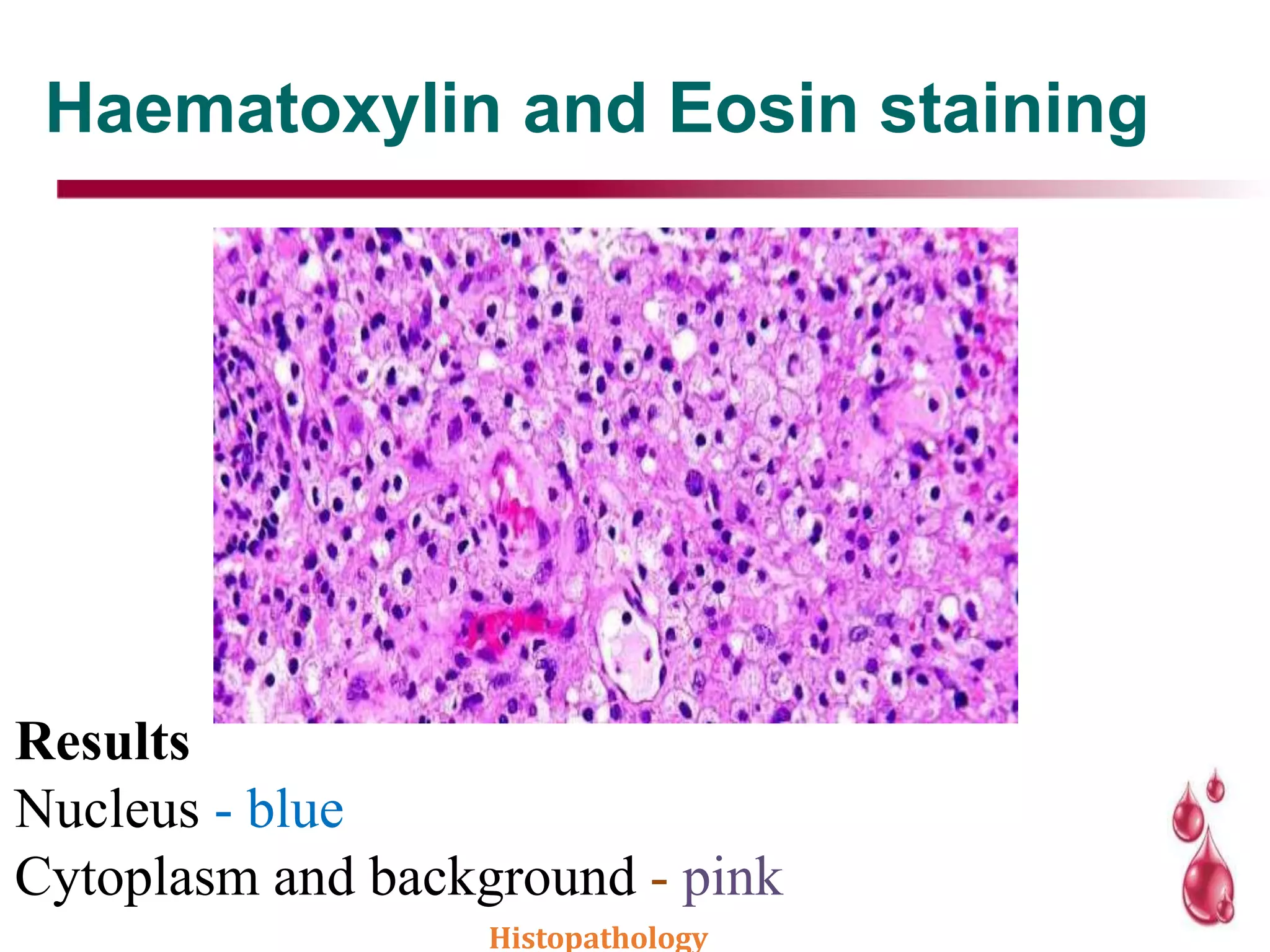
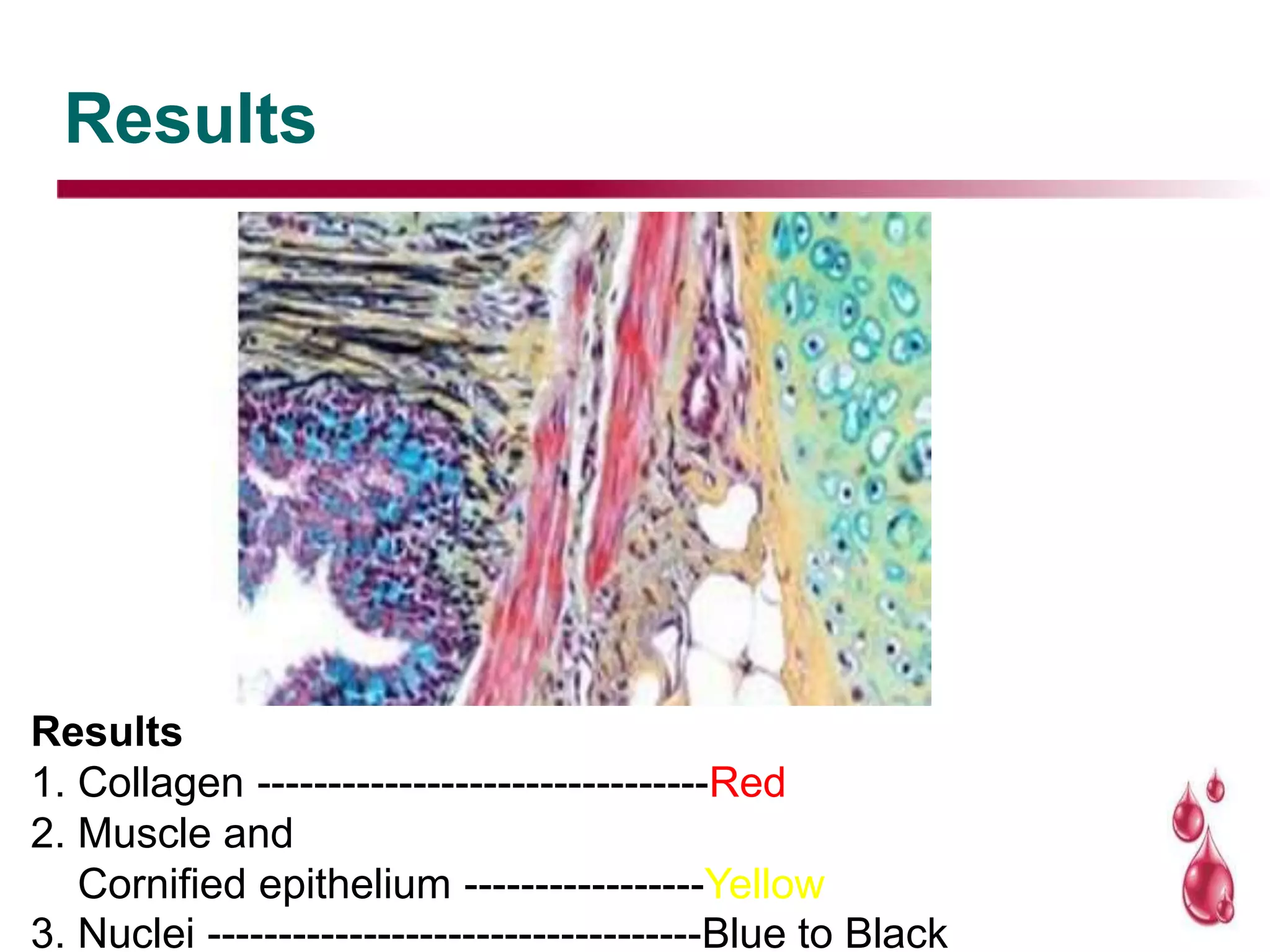
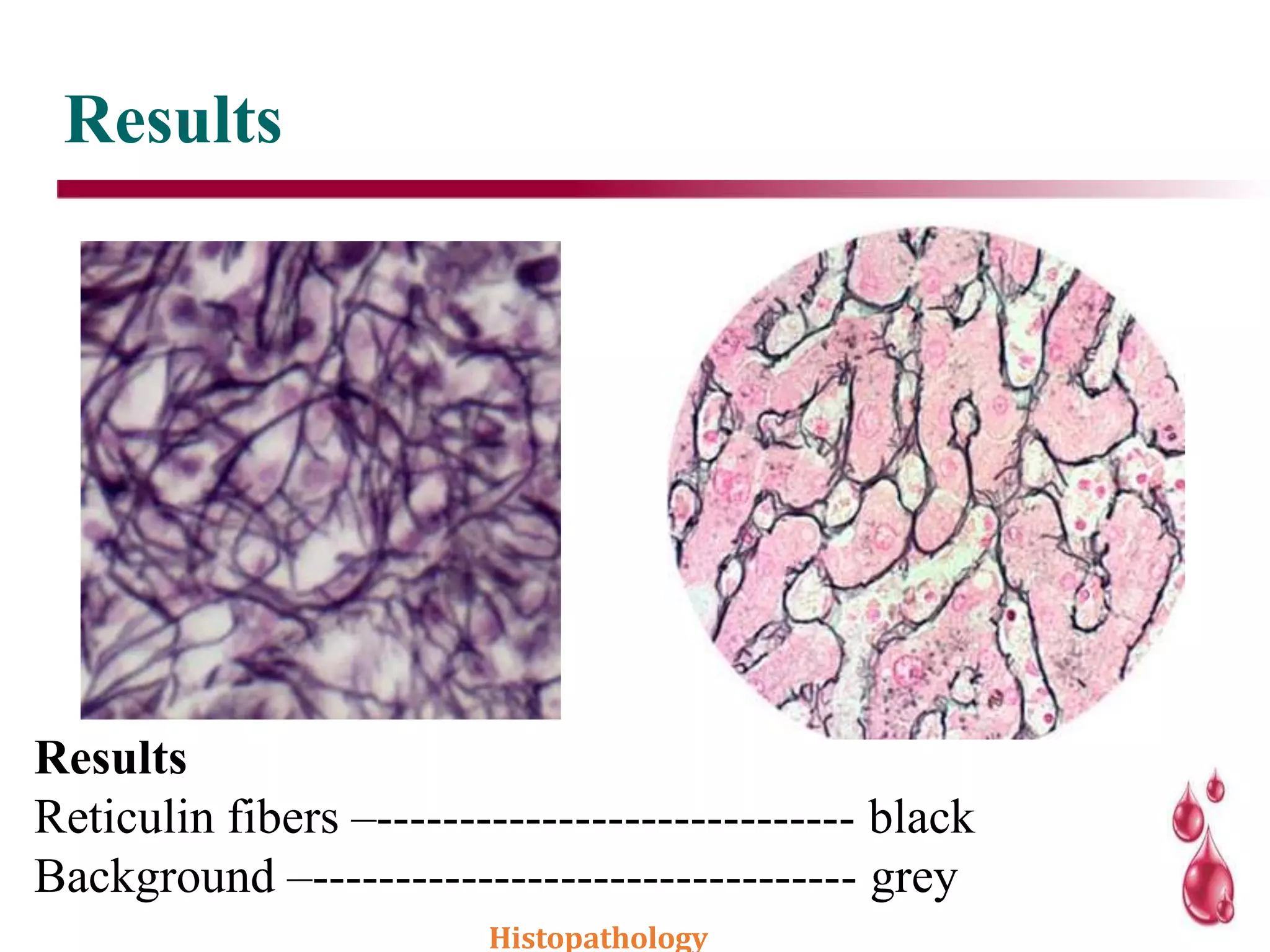
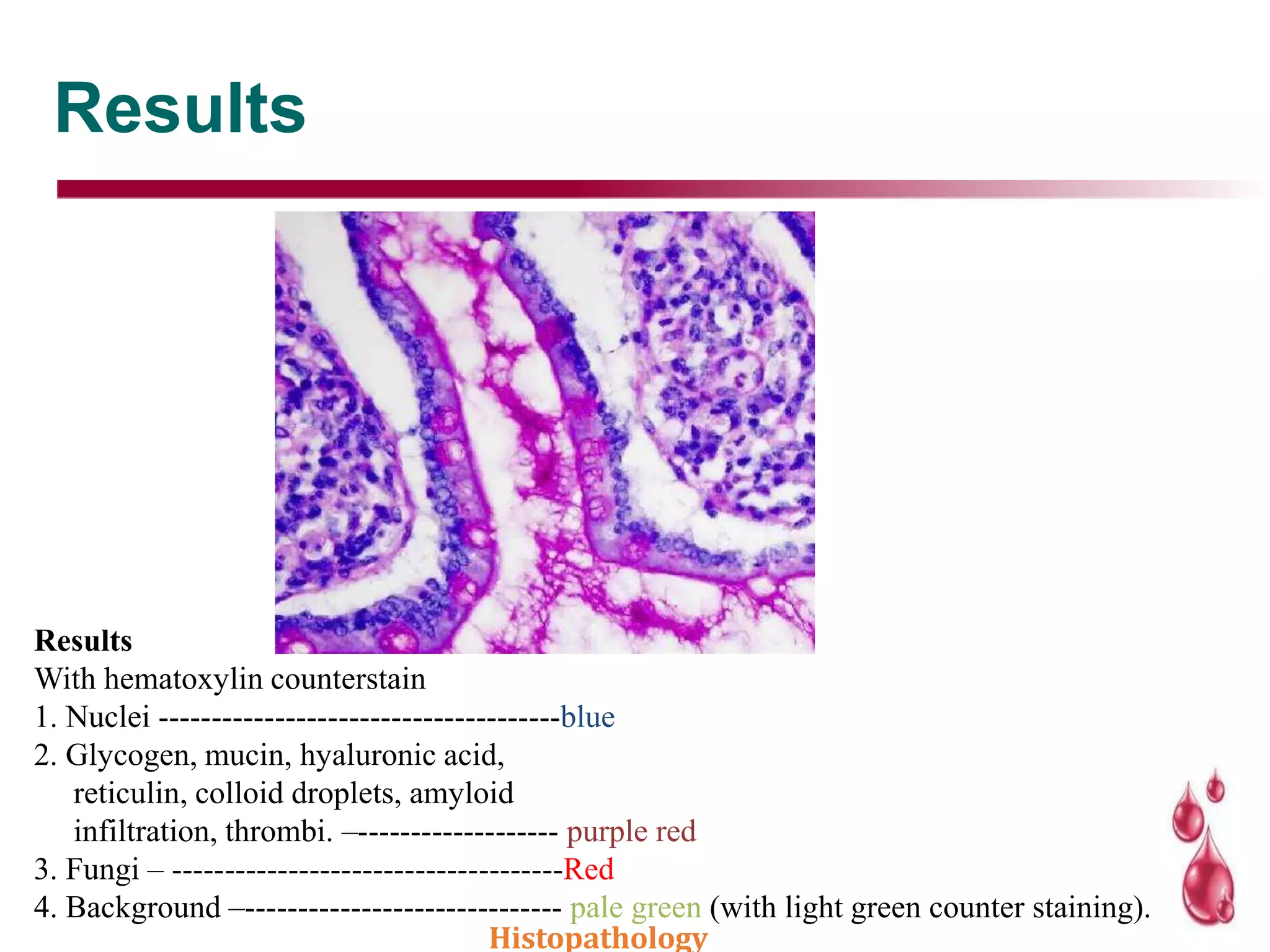
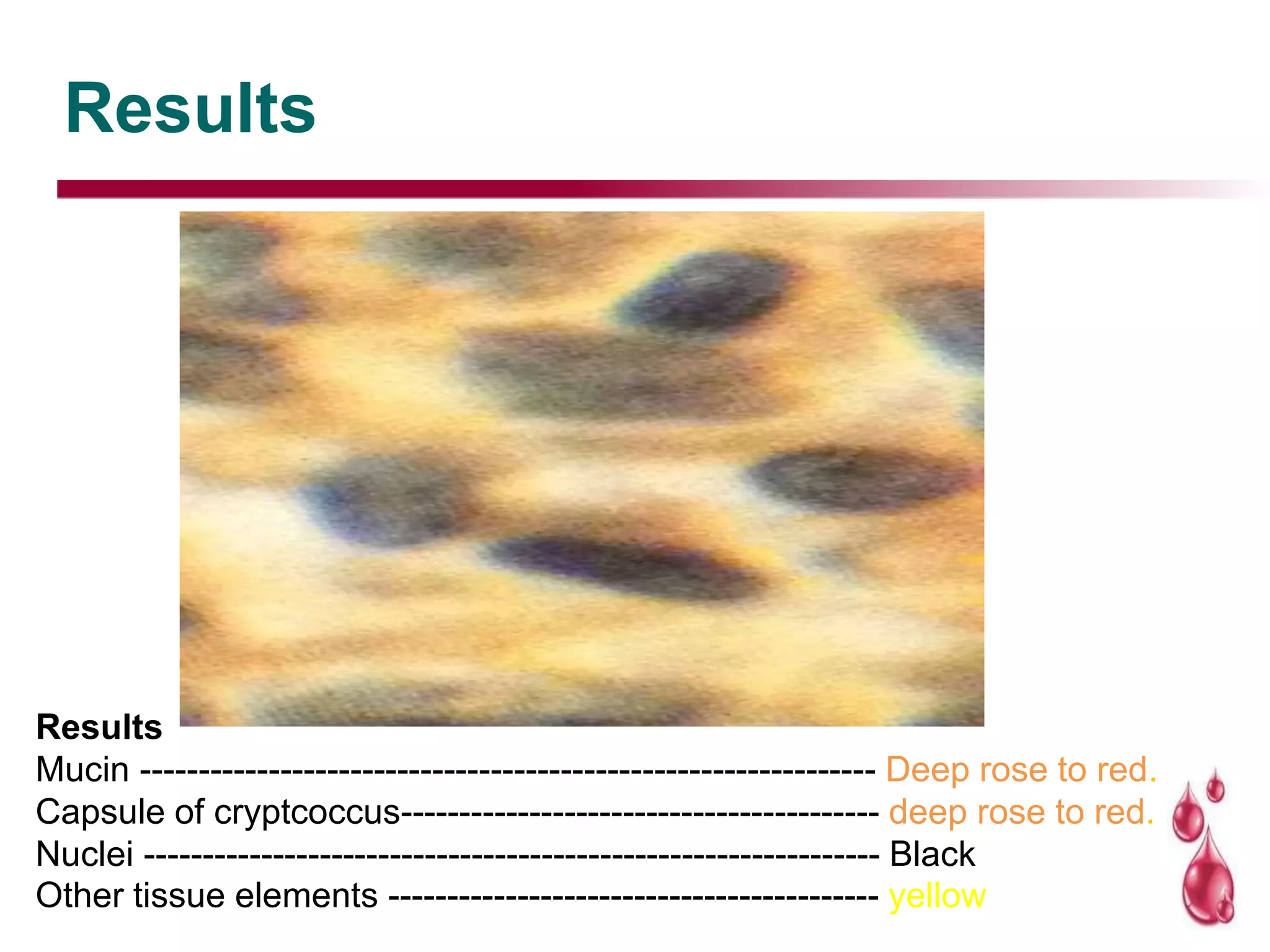
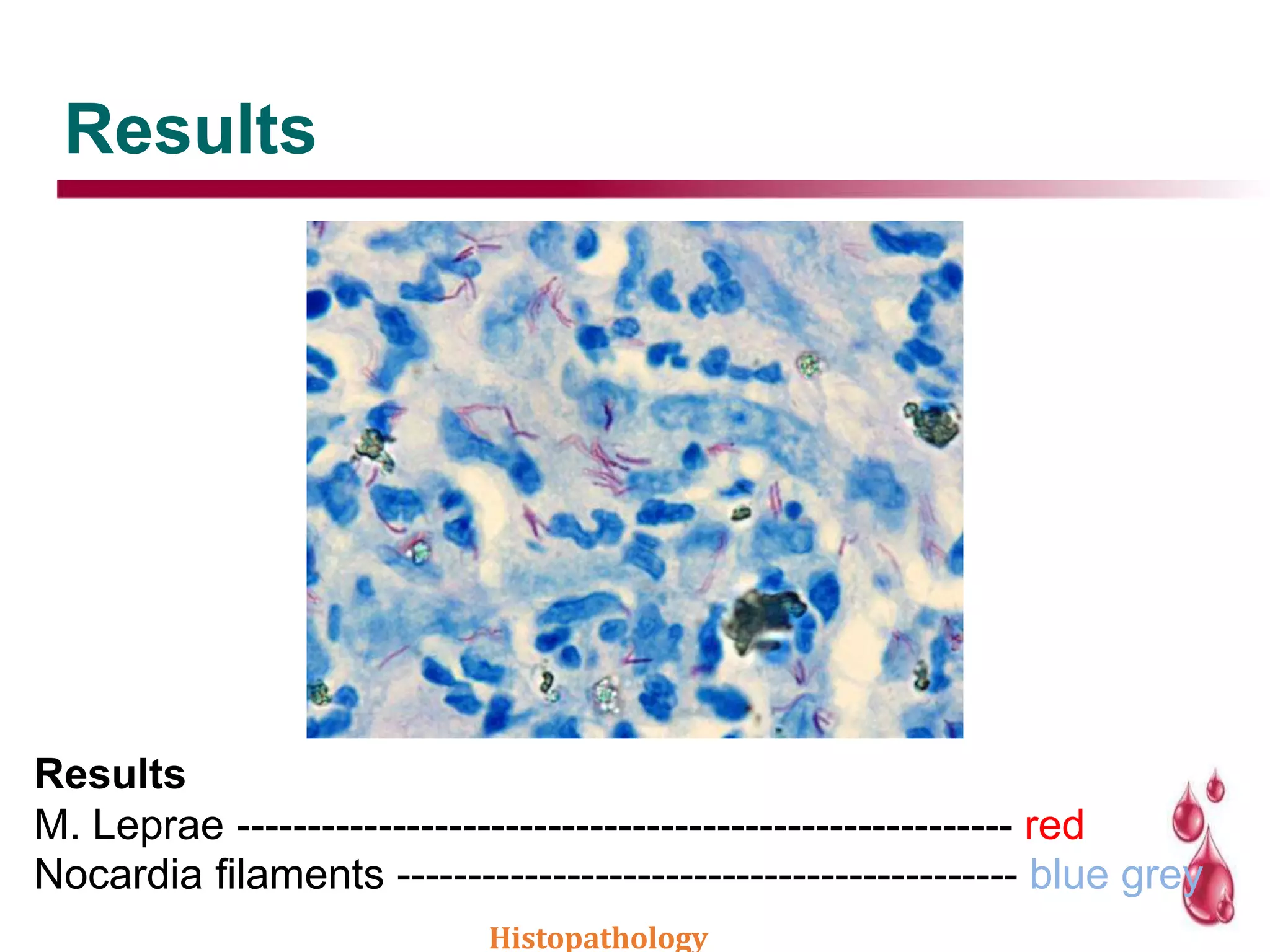
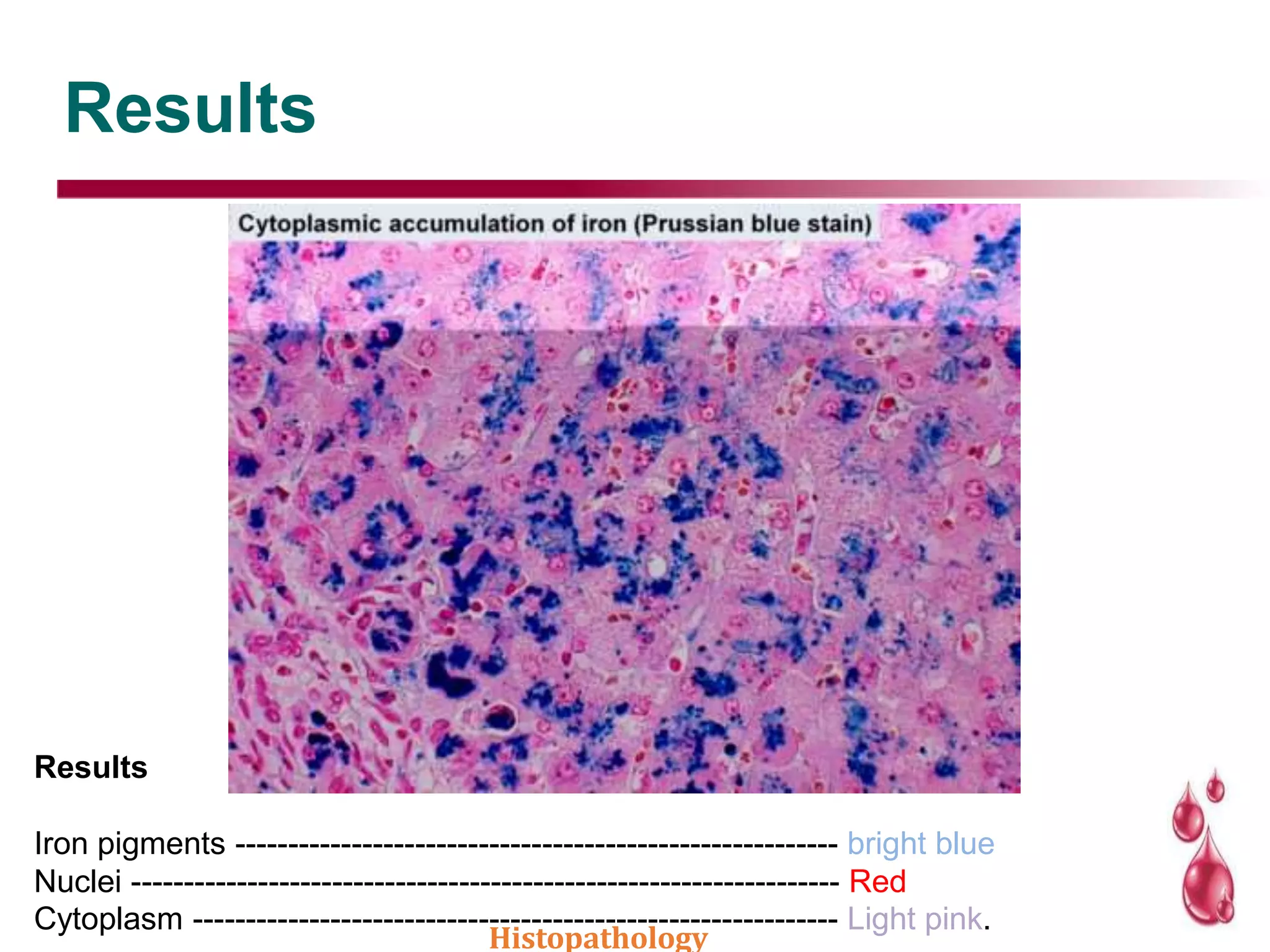
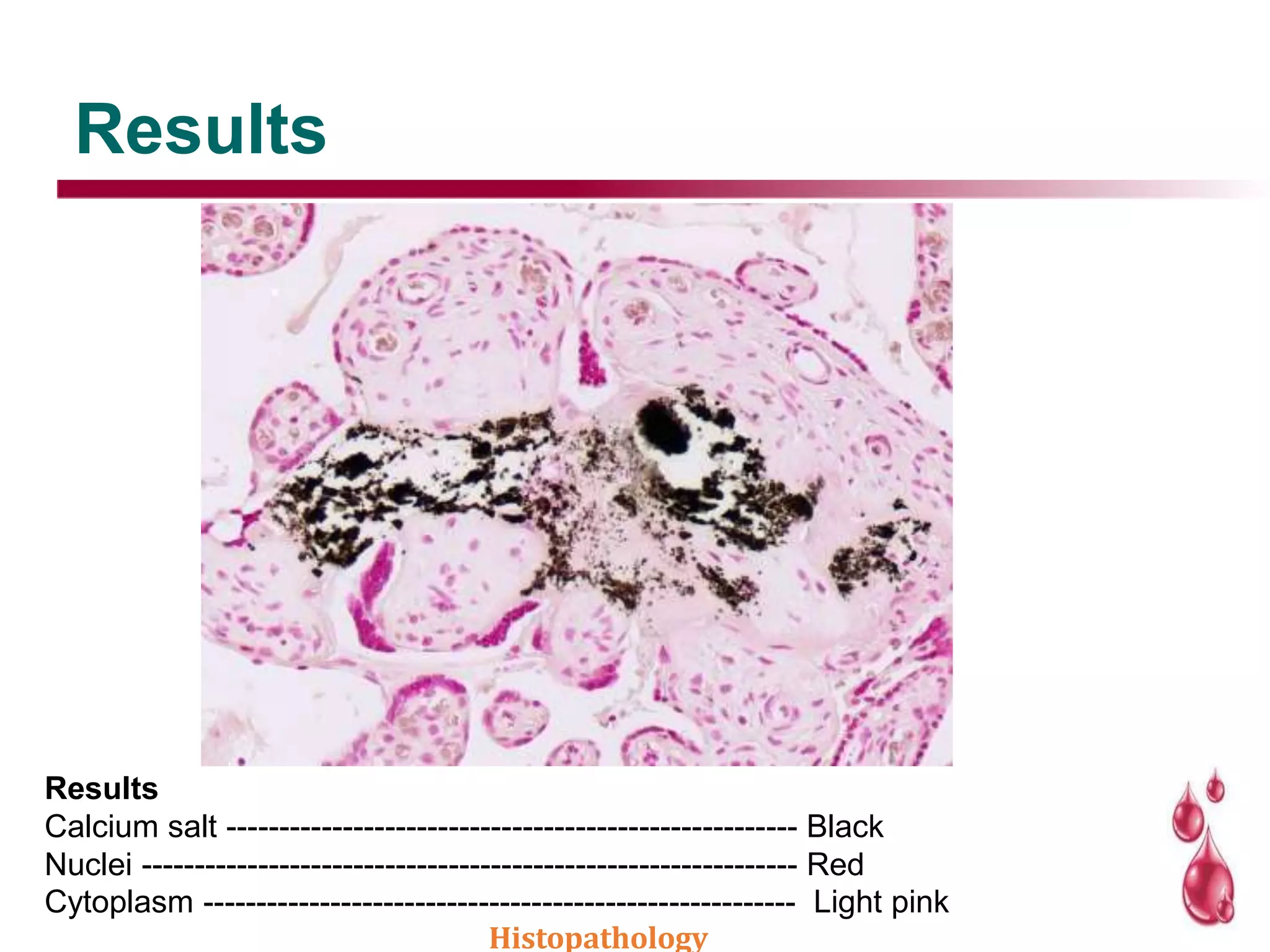
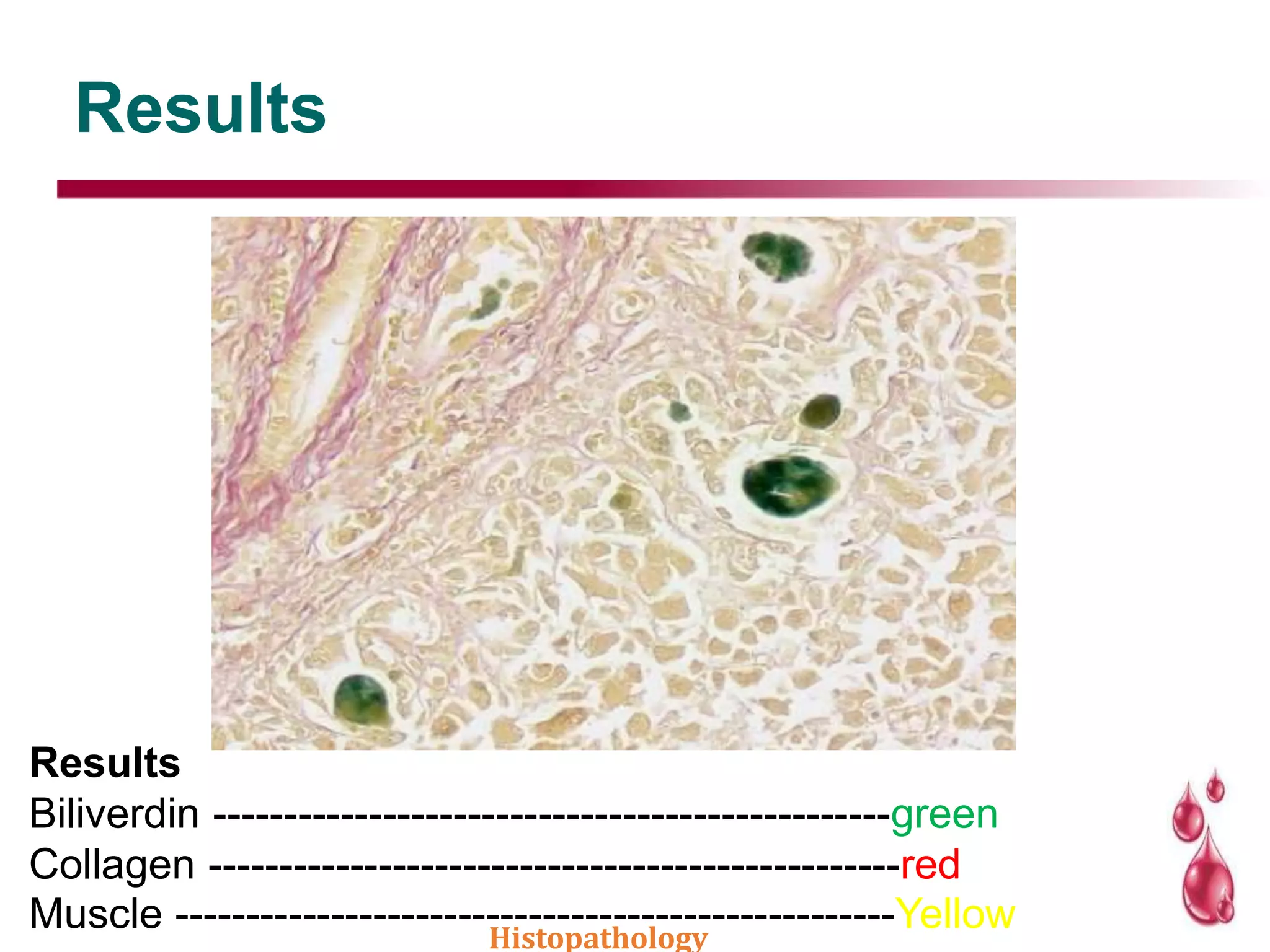
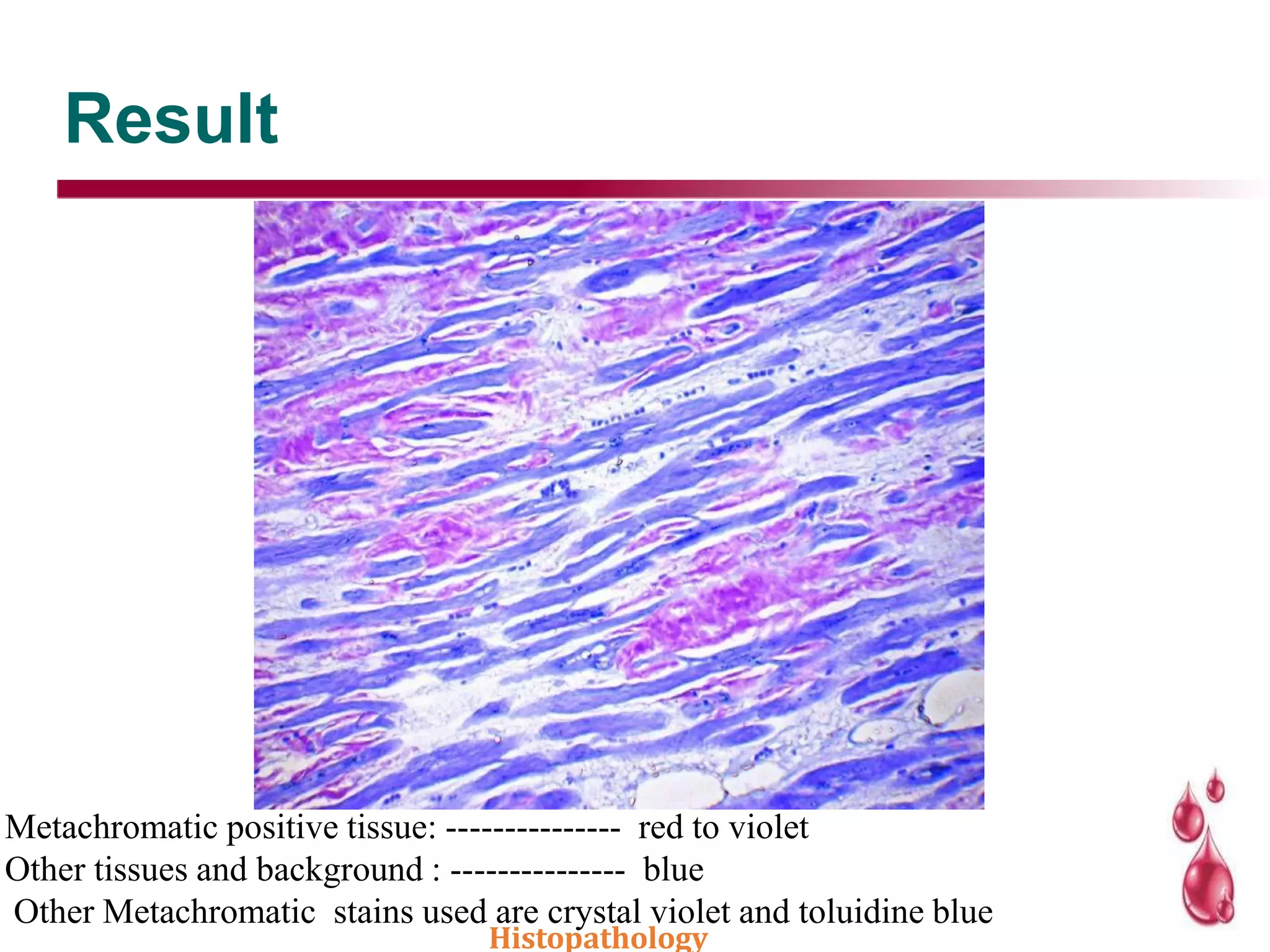
This document discusses histopathology staining techniques. It defines histopathology as staining tissue samples to examine cellular and intracellular structures microscopically. The two main categories of stains are natural dyes derived from natural resources, and artificial dyes produced through chemical reactions. Hematoxylin is a commonly used natural dye that stains nuclei blue, while eosin is an artificial dye that counterstains cytoplasm pink. The document outlines hematoxylin and eosin staining as well as other histological stains and their characteristics.