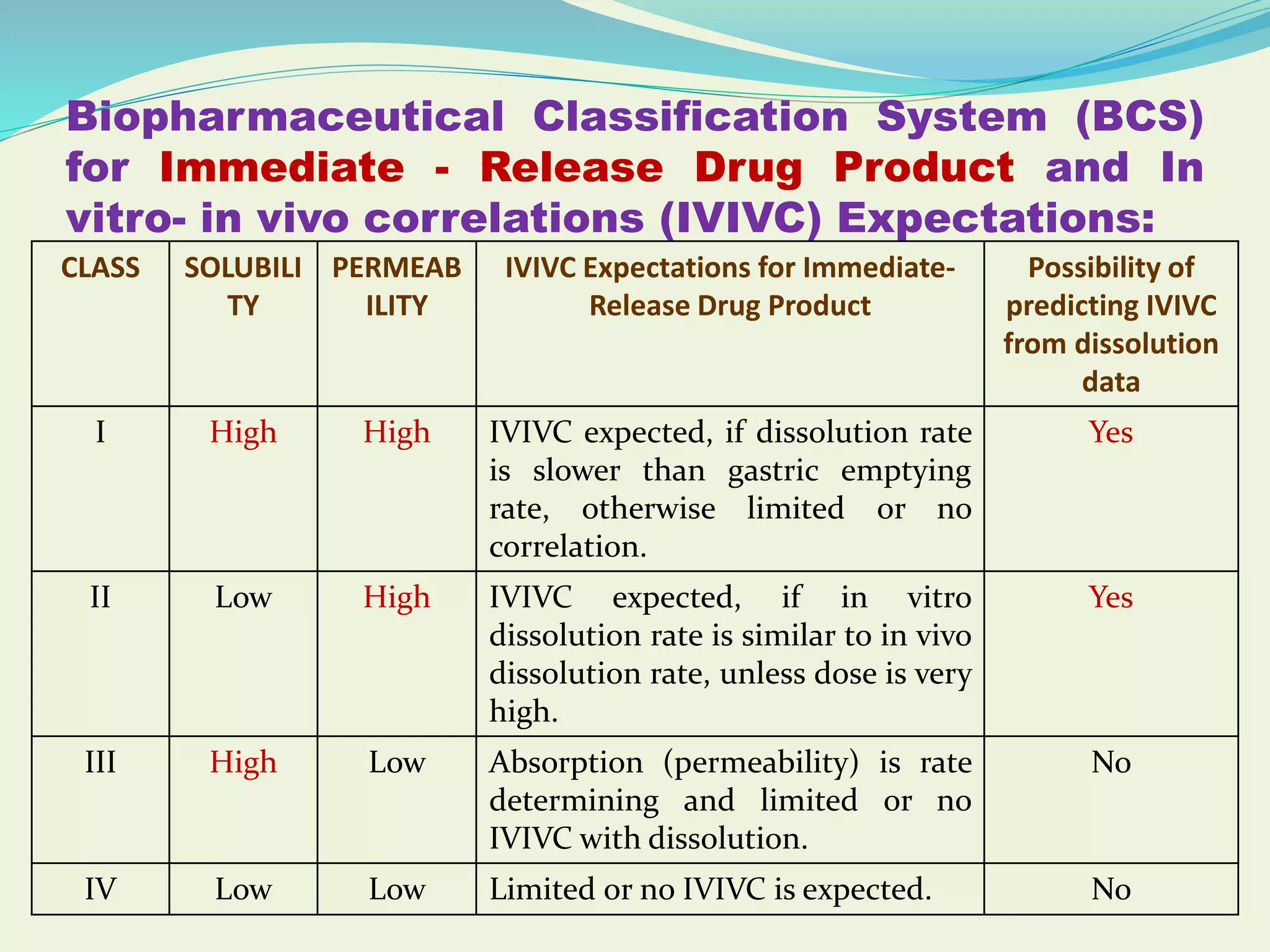
This document discusses in vitro-in vivo correlations (IVIVC), which describe the relationship between in vitro drug dissolution and in vivo absorption. It explains different levels of IVIVC, the criteria necessary for successful correlations, and their potential applications in drug formulation and development. The document also highlights the importance of IVIVC in minimizing human studies, supporting bioavailability, and improving product quality.