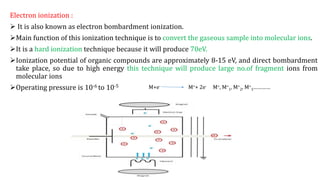
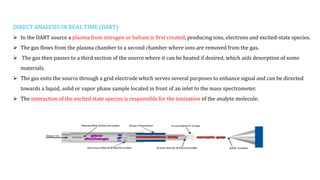
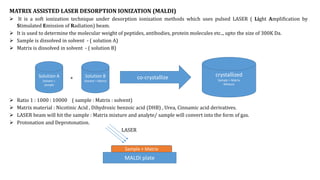
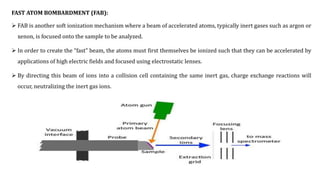
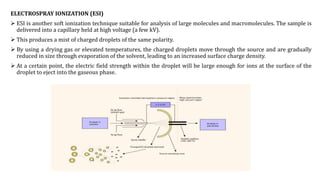
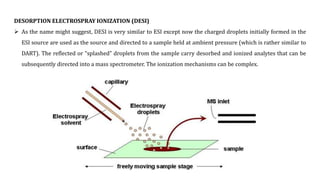
This document discusses various ionization techniques used in mass spectrometry. It describes gas phase methods like electron ionization and chemical ionization which use electron bombardment or carrier gases. Desorption methods like MALDI, FAB use lasers or atom beams. Inductively coupled plasma is used for liquid samples. Electrospray ionization and desorption electrospray ionization use charged droplets to ionize molecules. Each technique has different characteristics in terms of softness and applicable sample types.