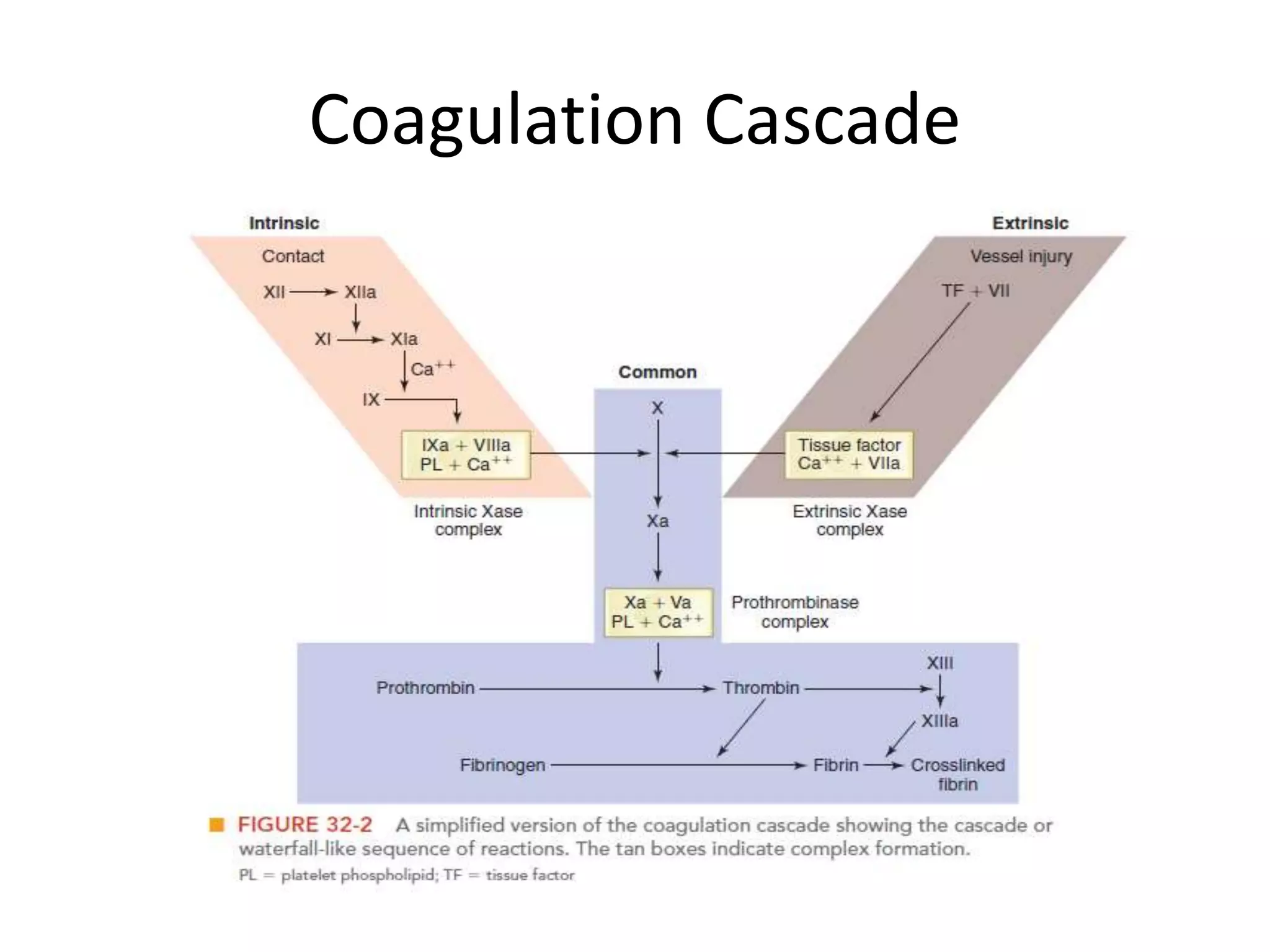
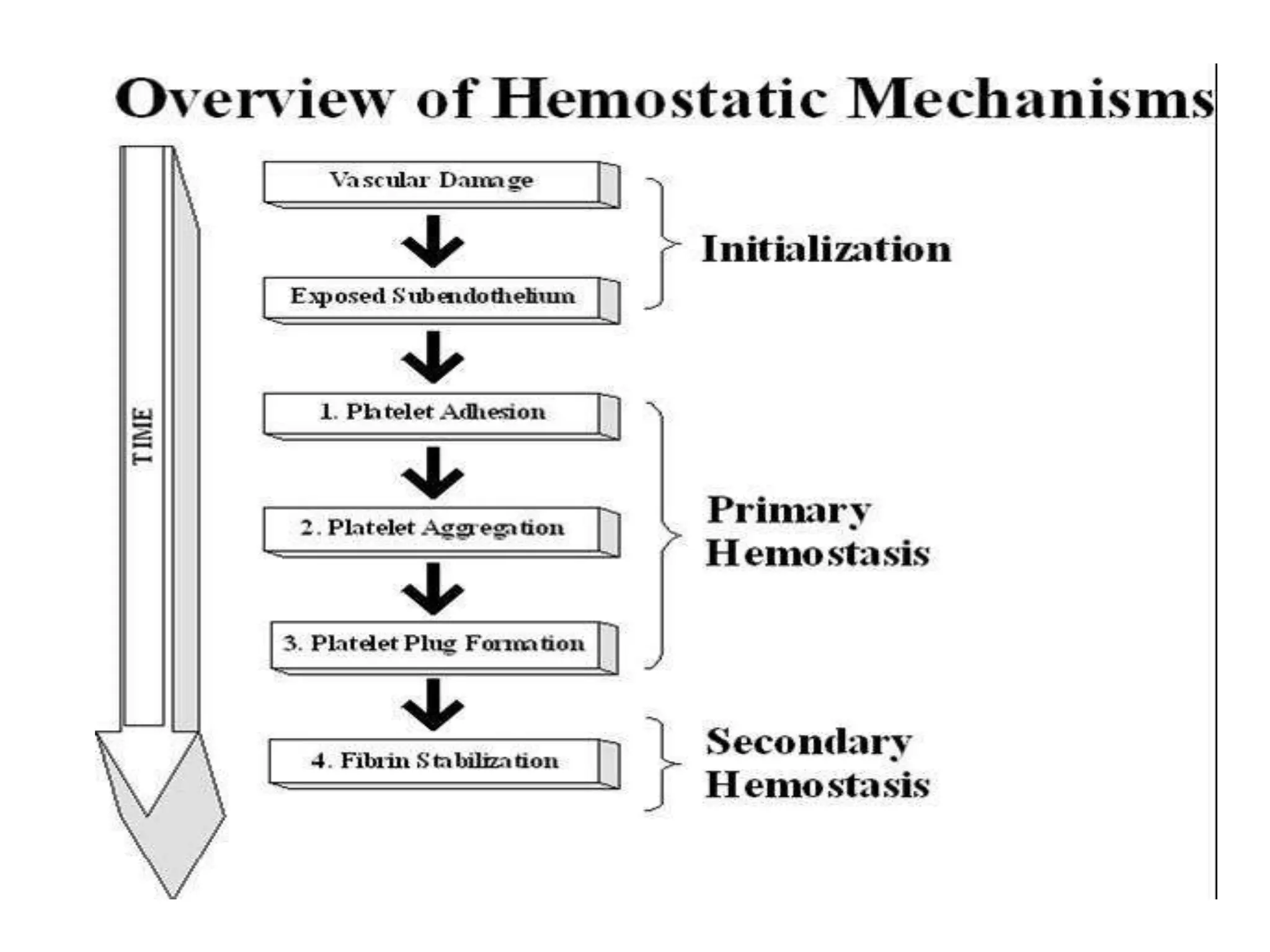
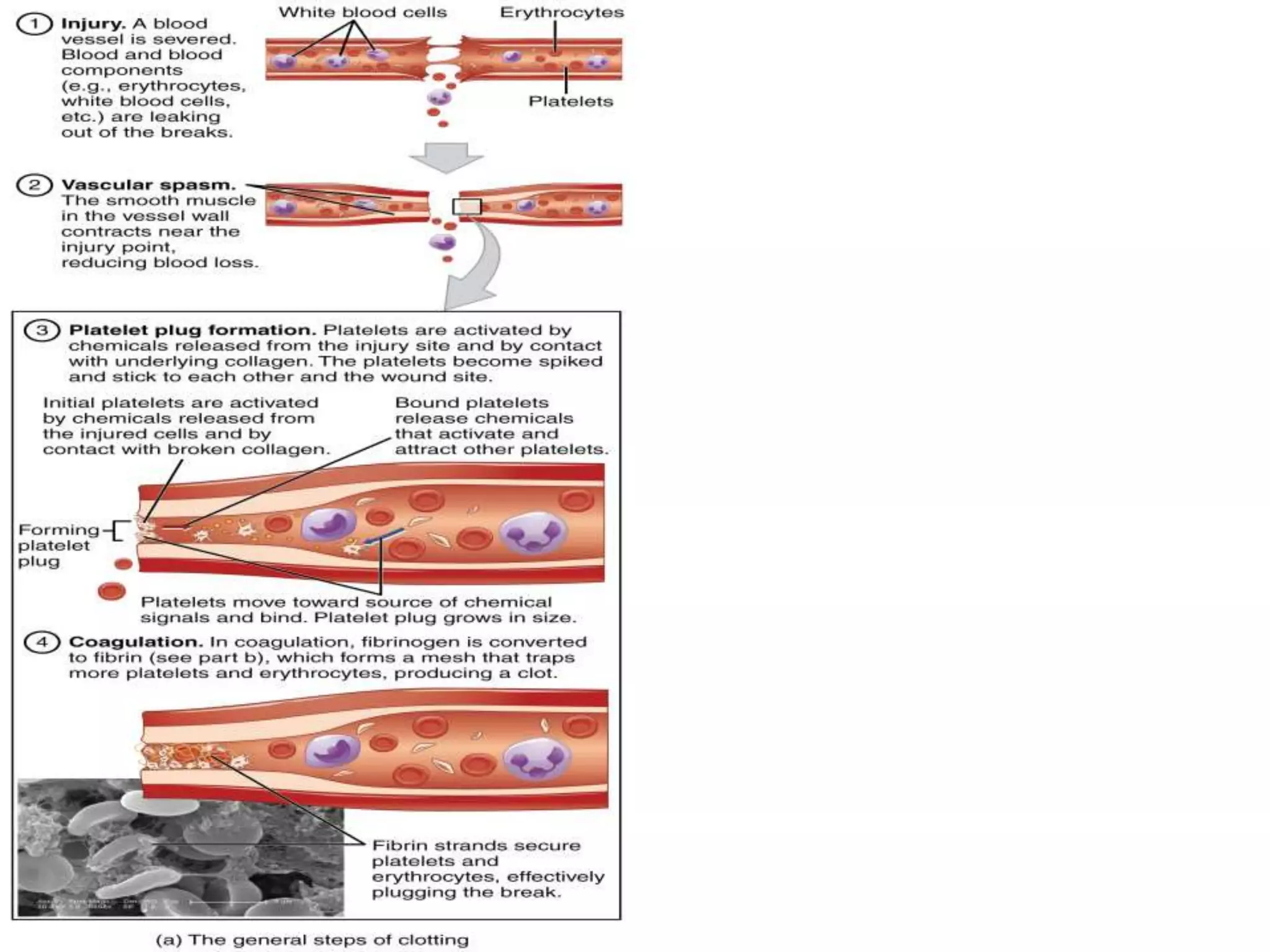
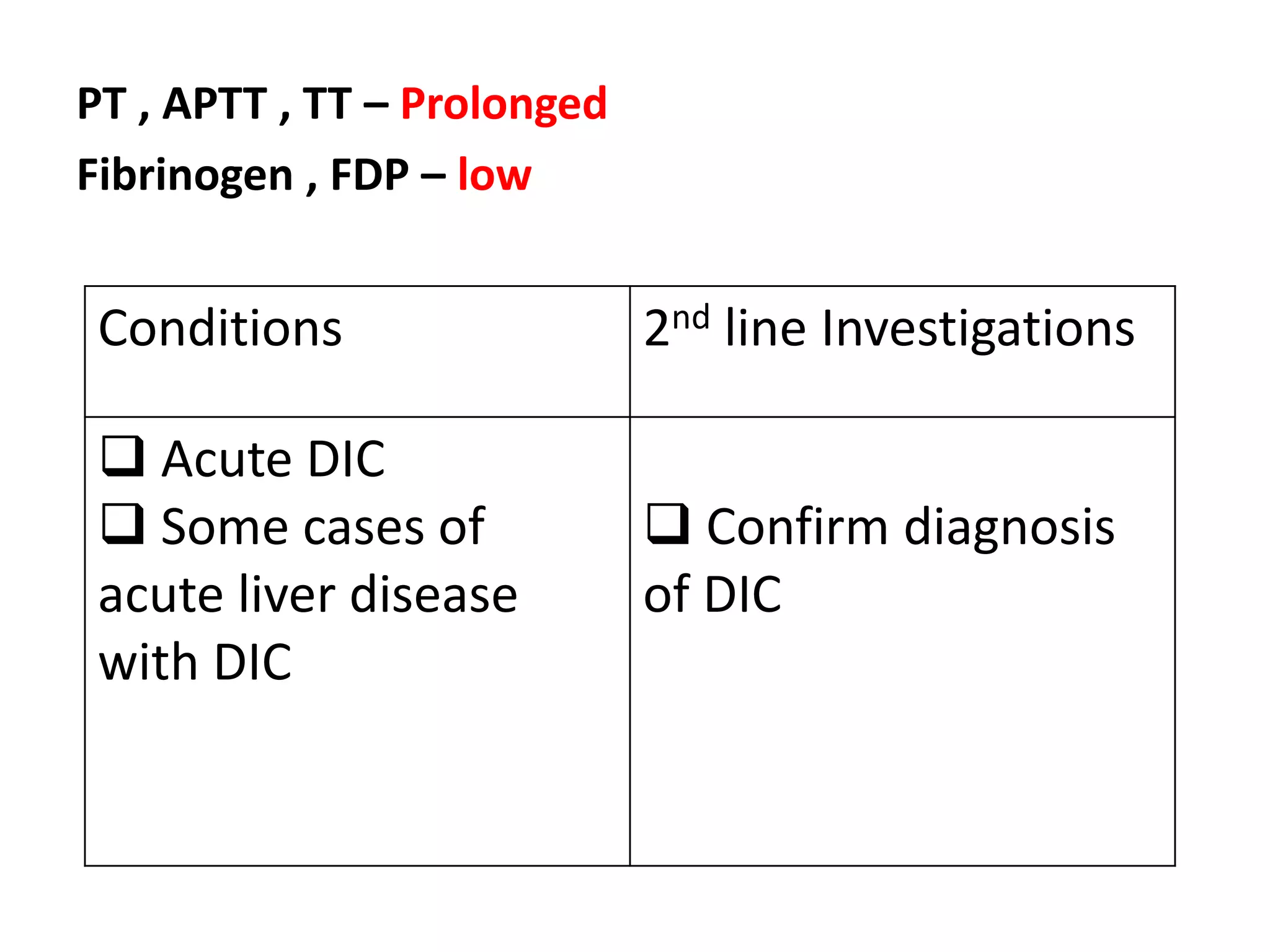
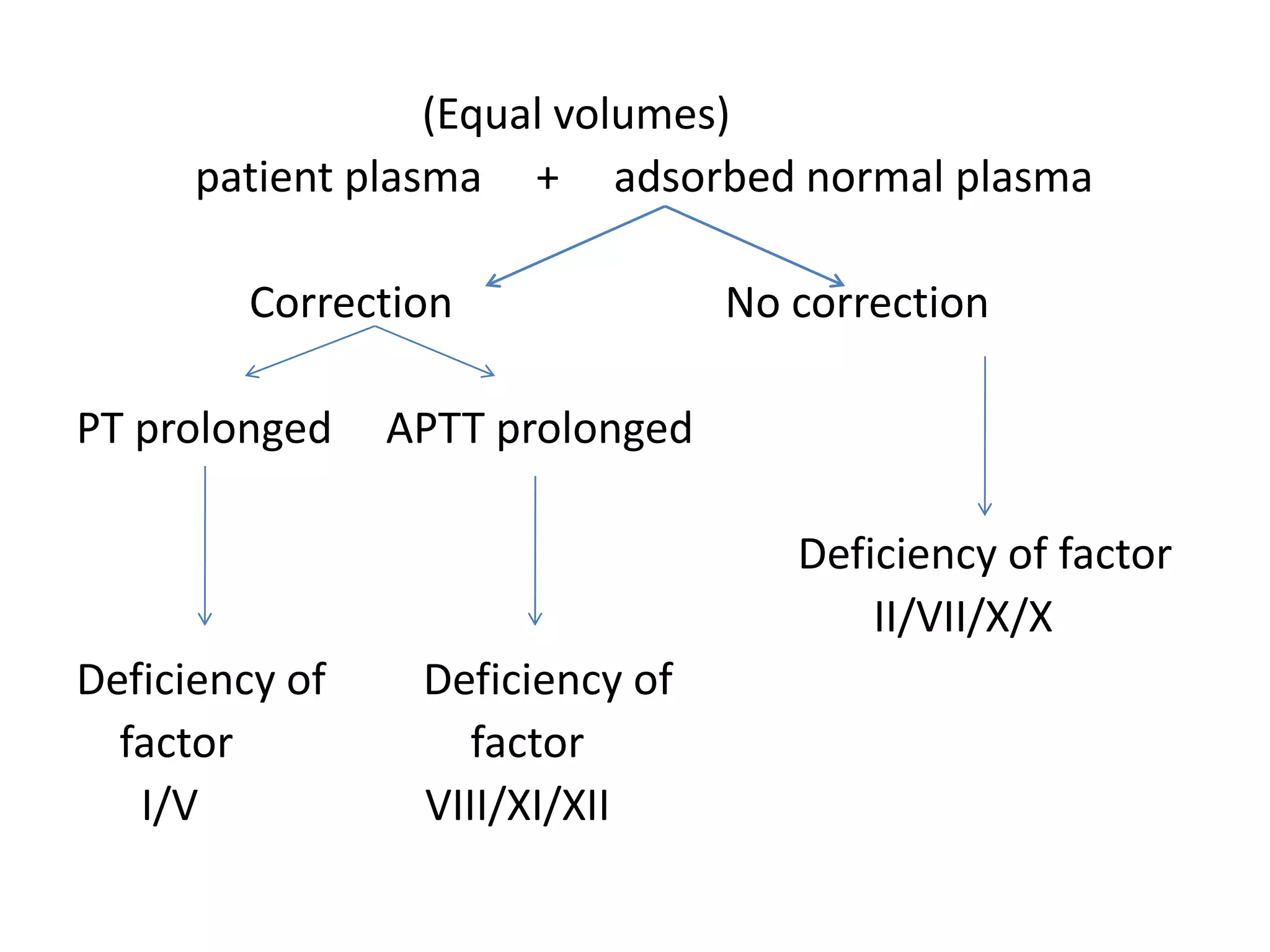
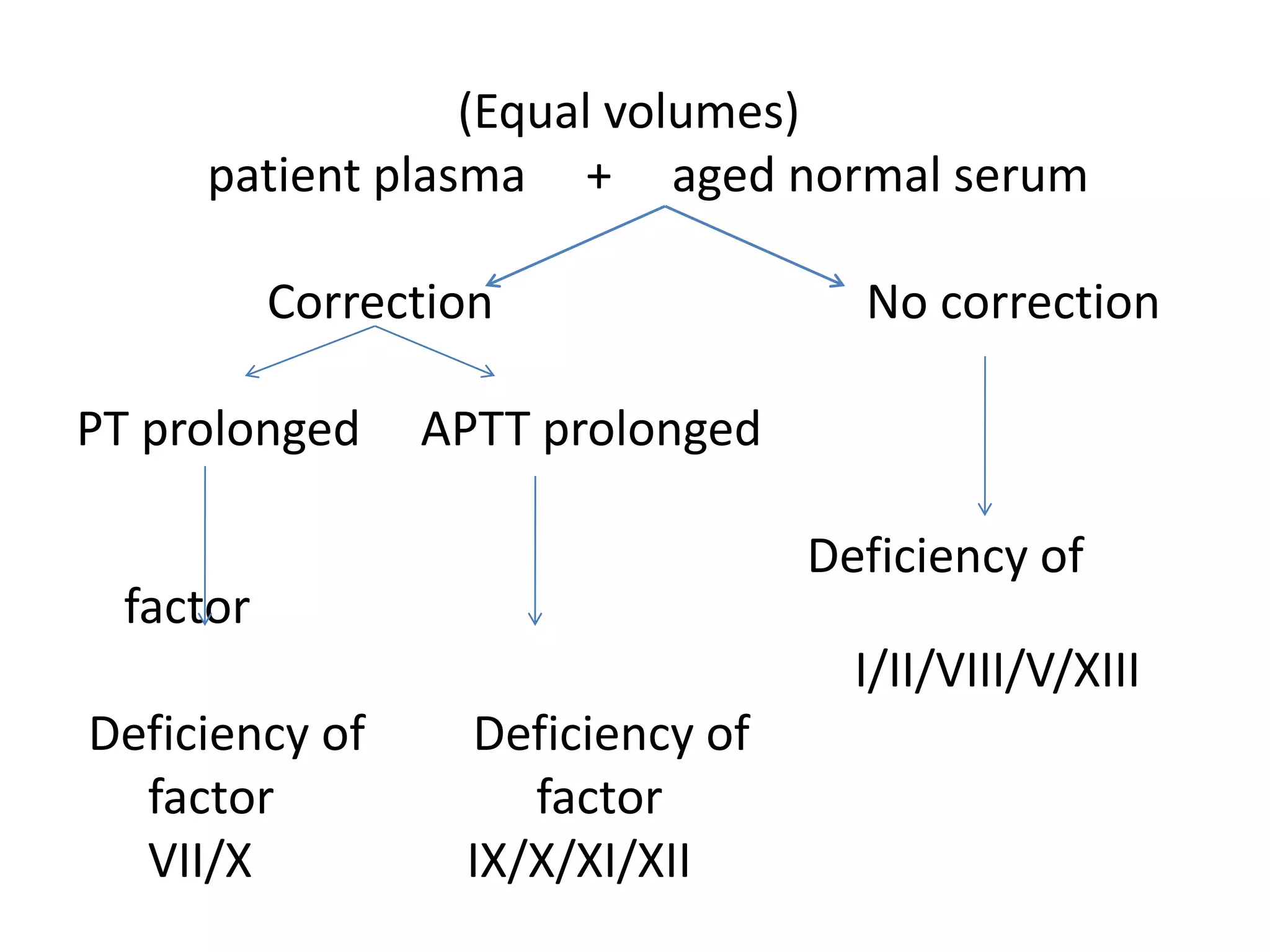
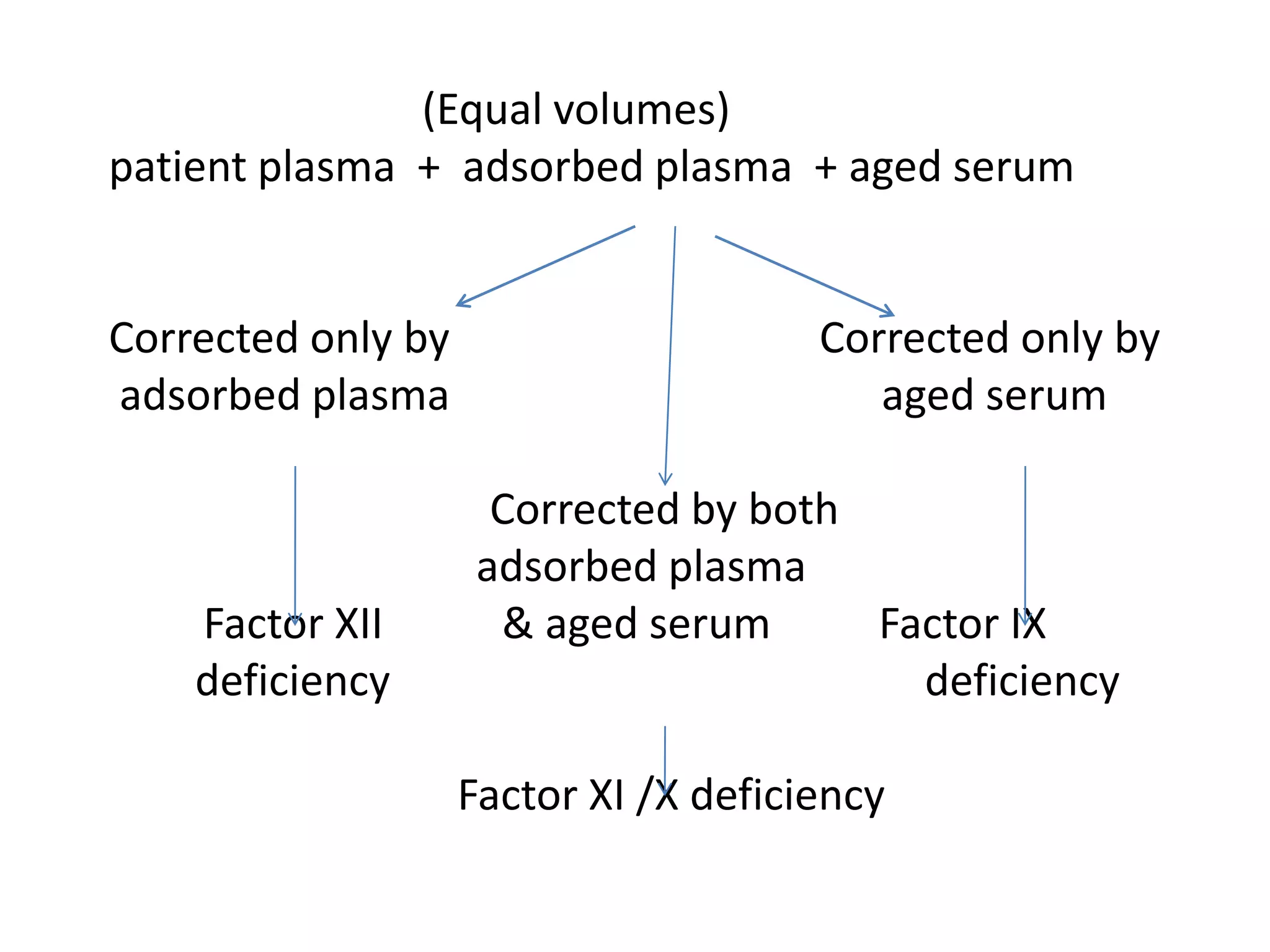
The document discusses interpretation of coagulation tests for coagulation disorders. It provides an overview of the physiology of coagulation, classification of coagulation disorders, description of specific disorders like hemophilia A and B, von Willebrand disease, fibrinogen disorders and disseminated intravascular coagulation. It also discusses interpretation of screening coagulation tests like prothrombin time, activated partial thromboplastin time and thrombin time as well as factor assays and interpretation for specific coagulation factor deficiencies.














![Lab Evaluation
• Laboratory studies for suspected hemophilia
include the following:
– Complete blood cell count
– Screening coagulation studies (prothrombin time
[PT], activated partial thromboplastin time [aPTT])
– FVIII assay (clot based or chromogenic)
– FVIII inhibitor assay (Bethesda assay, Nijmegen
modified Bethesda assay)](https://image.slidesharecdn.com/2modifiedinterpretationoftestsincoagulationdisorders-210901045435/75/Interpretation-of-tests-in-coagulation-disorders-21-2048.jpg)

![Prenatal Diagnosis
• Various methods are available for prenatal
diagnosis; the method of choice varies with the
type of mutation anticipated.
• A chorionic villus biopsy can be done at 11 weeks
of gestation and tested with DNA studies such as
restriction enzymes (restriction fragment length
polymorphism [RFLP]) or PCR methods.
• Direct sampling of fetal blood from the umbilical
vein is possible at many institutions, and a factor
assay can be performed on the blood sample.](https://image.slidesharecdn.com/2modifiedinterpretationoftestsincoagulationdisorders-210901045435/75/Interpretation-of-tests-in-coagulation-disorders-23-2048.jpg)



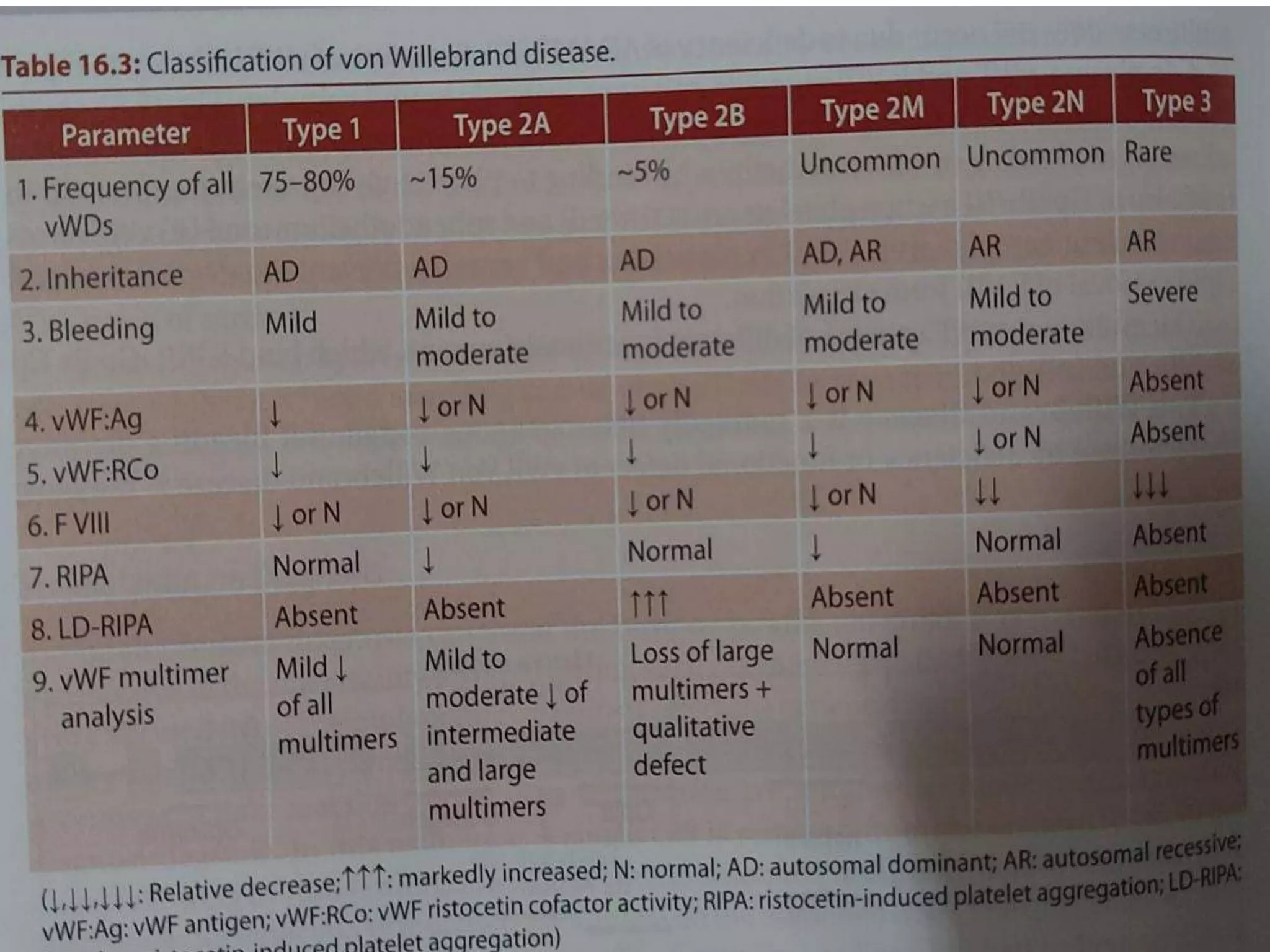















![Factor XI deficiency
• Factor XI deficiency is a congenital deficiency of blood
coagulation factor XI (known as plasma thromboplastin
antecedent [PTA] or antihemophilic factor C) resulting in a
systemic blood-clotting defect called hemophilia C or
Rosenthal syndrome, which may resemble classic hemophilia.](https://image.slidesharecdn.com/2modifiedinterpretationoftestsincoagulationdisorders-210901045435/75/Interpretation-of-tests-in-coagulation-disorders-43-2048.jpg)

![Factor XIII deficiency
• Factor XIII deficiency is a decrease or absence of factor XIII
(fibrin-stabilizing factor [FSF]) that prevents blood-clot
formation and results in a clinical hemorrhagic diathesis.
• Bleeding in a patient with both normal PT and aPTT should
raise the suspicion.
• Congenital factor XIII deficiency is a severe autosomal
recessive bleeding disorder associated with a characteristic
pattern of neonatal hemorrhage and lifelong bleeding
diathesis.](https://image.slidesharecdn.com/2modifiedinterpretationoftestsincoagulationdisorders-210901045435/75/Interpretation-of-tests-in-coagulation-disorders-45-2048.jpg)































![vWF Activity
• vWF activity (the binding of VWF to platelet
glycoprotein Ib [GPIb]) has traditionally been
assessed by ristocetin cofactor (RCoF) activity.
• In this test, ristocetin is added to a suspension
of washed formalin- or paraformaldehyde-
fixed platelets in the presence of the patient's
plasma (as a source of vWF).](https://image.slidesharecdn.com/2modifiedinterpretationoftestsincoagulationdisorders-210901045435/75/Interpretation-of-tests-in-coagulation-disorders-85-2048.jpg)