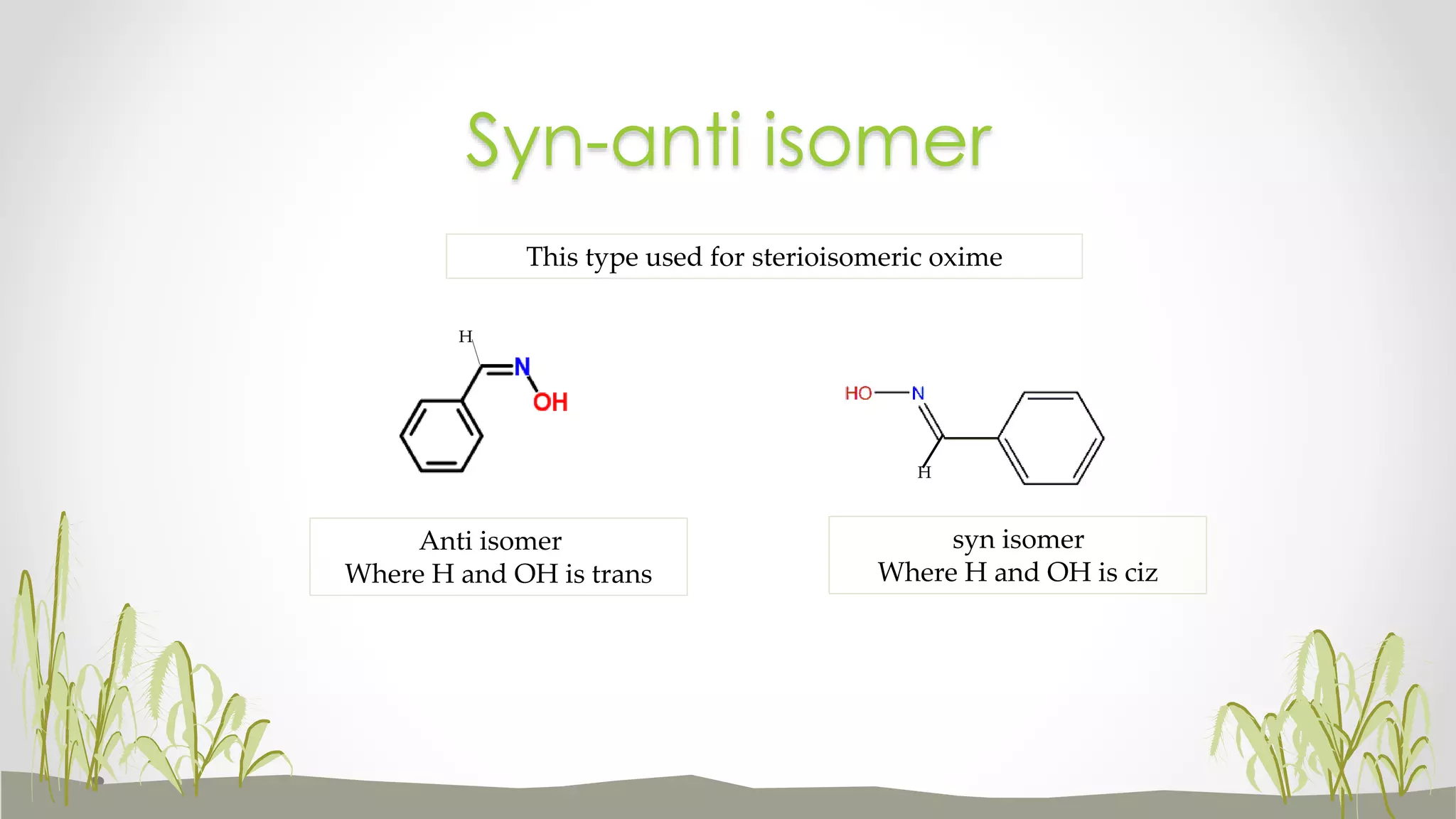
This document discusses geometrical isomers, which occur when rotation is restricted around a carbon-carbon double bond. Geometrical isomers can have different physical properties despite having the same molecular formula. The two types of geometrical isomers are cis-trans isomers and E-Z isomers. Cis-trans isomers have hydrogens on the same side (cis) or opposite sides (trans) of the double bond. E-Z isomers are assigned based on priority rules, with the Z isomer having higher priority groups on the same side and the E isomer having them on opposite sides. Examples are given of how geometrical isomers can impact drug action.