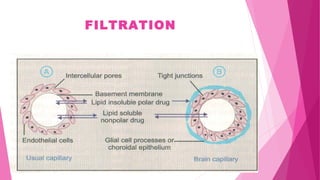
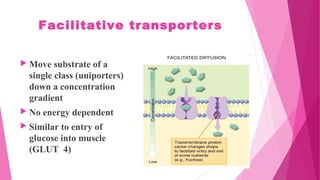
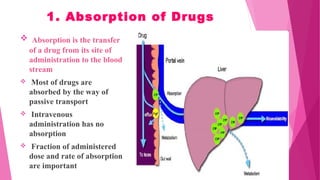
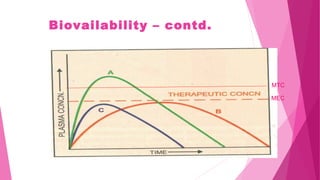
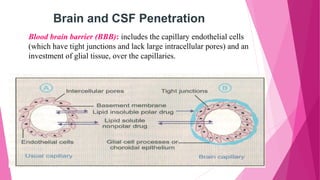
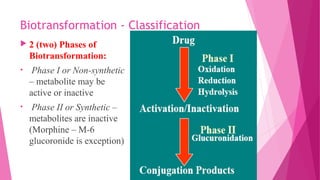
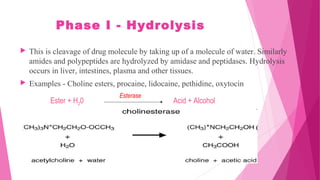
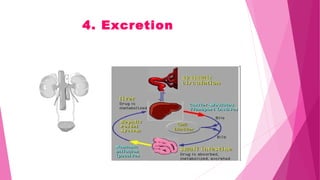
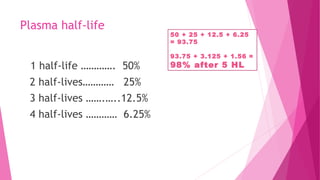
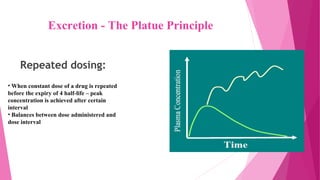
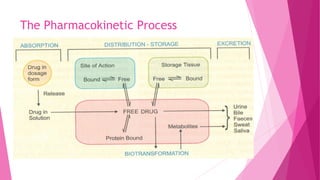
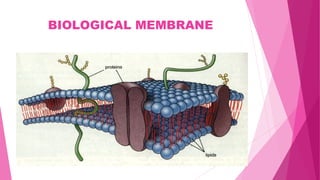
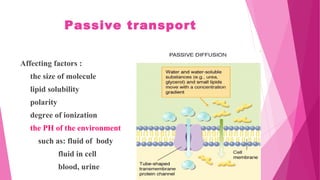
Pharmacokinetics is the study of how the body affects drugs. It involves absorption, distribution, metabolism, and excretion of drugs. Absorption is how drugs enter the bloodstream and distribution is how drugs spread to tissues. Metabolism converts drugs to inactive forms through phase I (oxidation) and phase II (conjugation) reactions. Excretion eliminates drugs from the body. Together, these processes determine the effects of drugs over time.







![Henderson–Hasselbalch
Equation
pKa = negative logarithm of acid dissociation constant
[A-] = ionized Drug
[HA] = unionized drug](https://image.slidesharecdn.com/pharmacokinetics-161223014142/85/General-Pharmacology-for-BPT-Students-11-320.jpg)