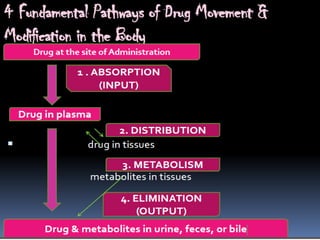
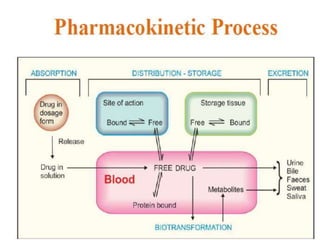
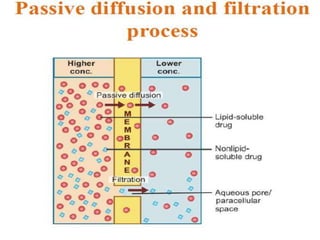
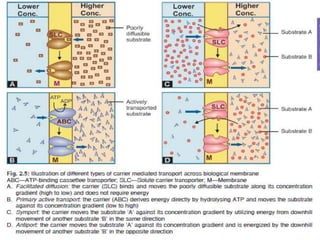
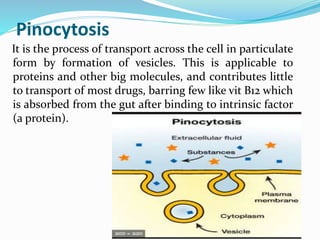
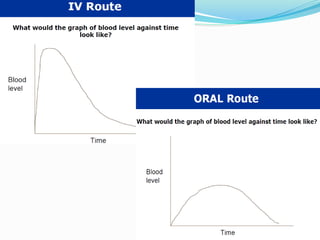
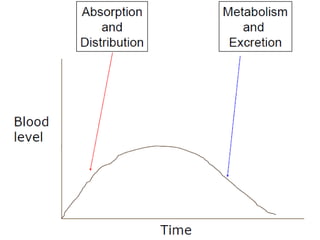
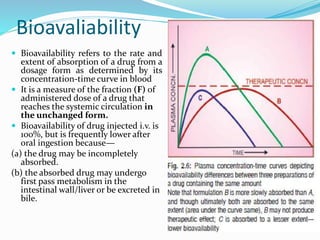
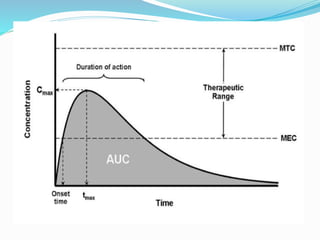
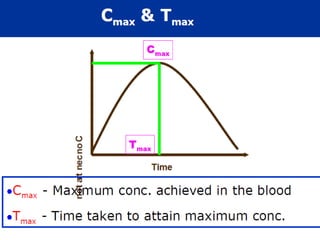
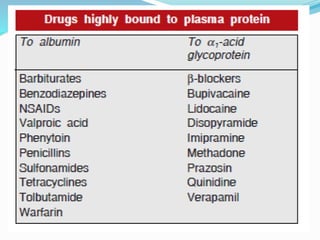
This document discusses the pharmacokinetics of drug absorption and distribution. It begins by defining pharmacokinetics as the quantitative study of how the body acts on drugs. It then discusses the different mechanisms of drug transportation across cell membranes, including passive diffusion, filtration, and carrier-mediated transport like facilitated diffusion and active transport. It describes factors that affect drug absorption like solubility, concentration, and route of administration. It also discusses concepts like bioavailability, bioequivalence, distribution, redistribution, barriers to drug movement, and plasma protein binding. In summary, it provides an overview of how drugs move into, through, and out of the body after administration.