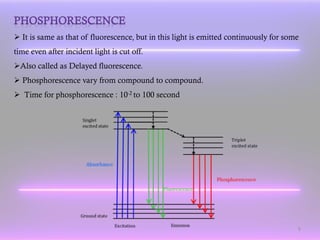
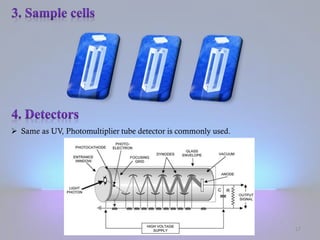
The document discusses the principles of fluorescence and phosphorescence, detailing the processes of singlet, doublet, and triplet electronic states, as well as factors affecting fluorescence intensity and quenching. It explains the instrumentation used for measuring fluorescence, highlighting the importance of concentration, structural and non-structural factors, and types of quenching. Applications of fluorescence measurement include the determination of various inorganic substances and organic compounds.