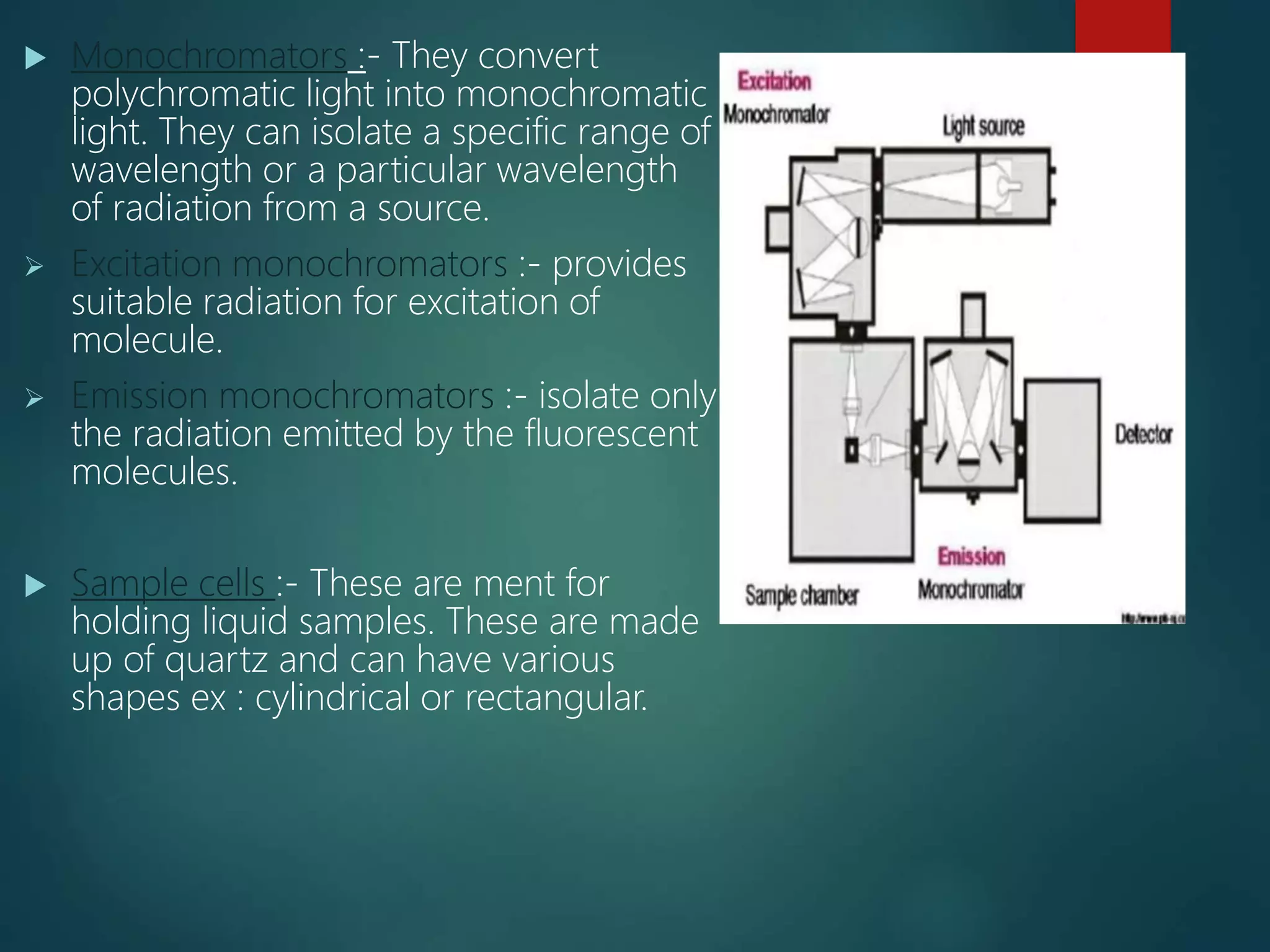
The document discusses spectrofluorimetry, a technique based on the absorption of UV/visible radiation, leading to the excitation of electrons in molecules. It outlines the principles, advantages, disadvantages, and factors affecting fluorescence intensity, as well as instrumentation and applications in various fields, including food and pharmaceutical analysis. Fluorescence is highlighted as a sensitive analytical method, primarily used for quantitative analysis rather than qualitative analysis.