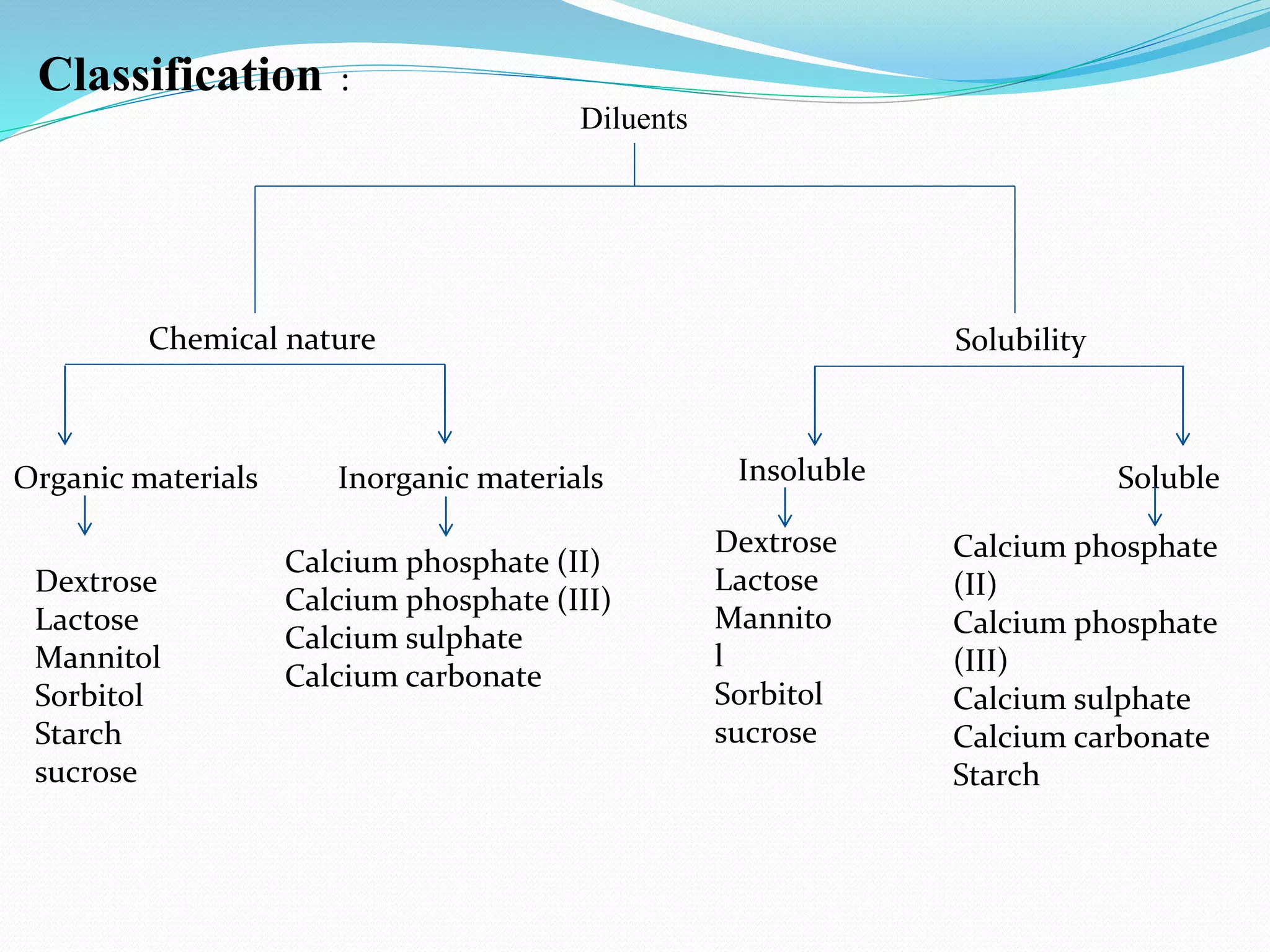
Excipients are inactive substances formulated with active pharmaceutical ingredients to create drug products. They serve important purposes like bulking up formulations, ensuring consistent drug release and stability, and determining properties of the final dosage form like tablet size and dissolution rate. Common excipients include diluents, binders, disintegrants, lubricants, and glidants. Diluents increase volume and include substances like lactose, starch and calcium phosphate. Binders promote adhesion while disintegrants facilitate breaking of tablets. Lubricants prevent adhesion during compression and glidants promote powder flow. Proper excipient selection is crucial for an efficacious and robust drug product.