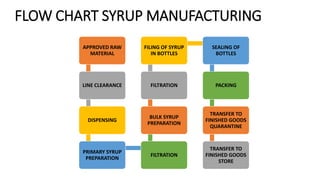
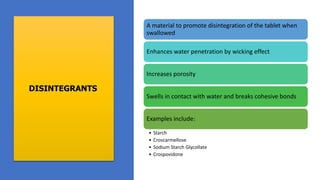
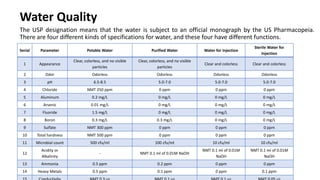
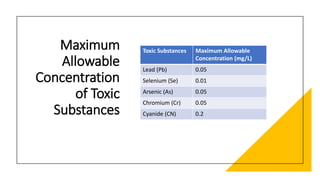
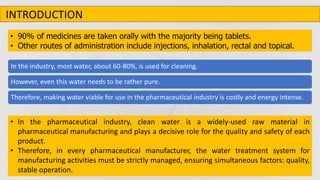
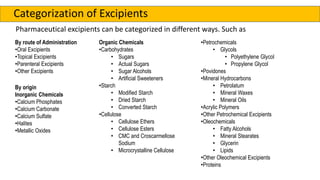
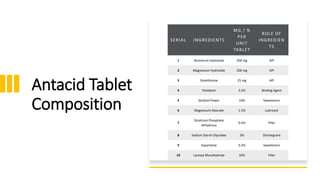
The document outlines various aspects of the pharmaceutical industry, emphasizing the importance of clean water and specialized raw materials such as active pharmaceutical ingredients (APIs) and excipients. It details manufacturing processes like direct compression and granulation, along with typical formulations and their key components, which influence tablet quality and performance. Additionally, it discusses common challenges encountered during tablet manufacturing, such as weight variation and capping, as well as potential remediation strategies.



![RECEIPT OF RAW
MATERIAL FROM STORE
VERIFICATION OF
MATERIAL & WEIGHTS AT
SHOP FLOOR
SIFTING OF INVIDUAL
MATERIAL
DRYING
MILLING/ GRANULE
SIZING
LUBRICATION/BLENDING[
YIELD CALCULATION]
COMPRESSION
[SIMULTANEOUS
OBSERVATIONS BY
PROD.&IPQA]
VISUAL INSPECTON
[YEILD ANALYSIS]
COATING
[SIMULTANEOUS
OBSERVATIONS ON EACH
LOT BY PROD.&IPQA]
VISUAL INSPECTION
[YEILD ANALYSIS]
PM VERIFICATION AND
BATCH CODING
PACKING
[SIMULTANEOUS
OBSERVATIONS BY
PROD.&IPQA]
FINISHED GOOD
ANALYSIS
TRANSFER TO F.G.STORE
[BPR TO QA]
FLOW CHART TABLET MANUFACTURING AND
PACKING](https://image.slidesharecdn.com/pharmaceuticalsindustry-231117181213-a9e0ca92/85/Pharmaceuticals-Industry-7-320.jpg)