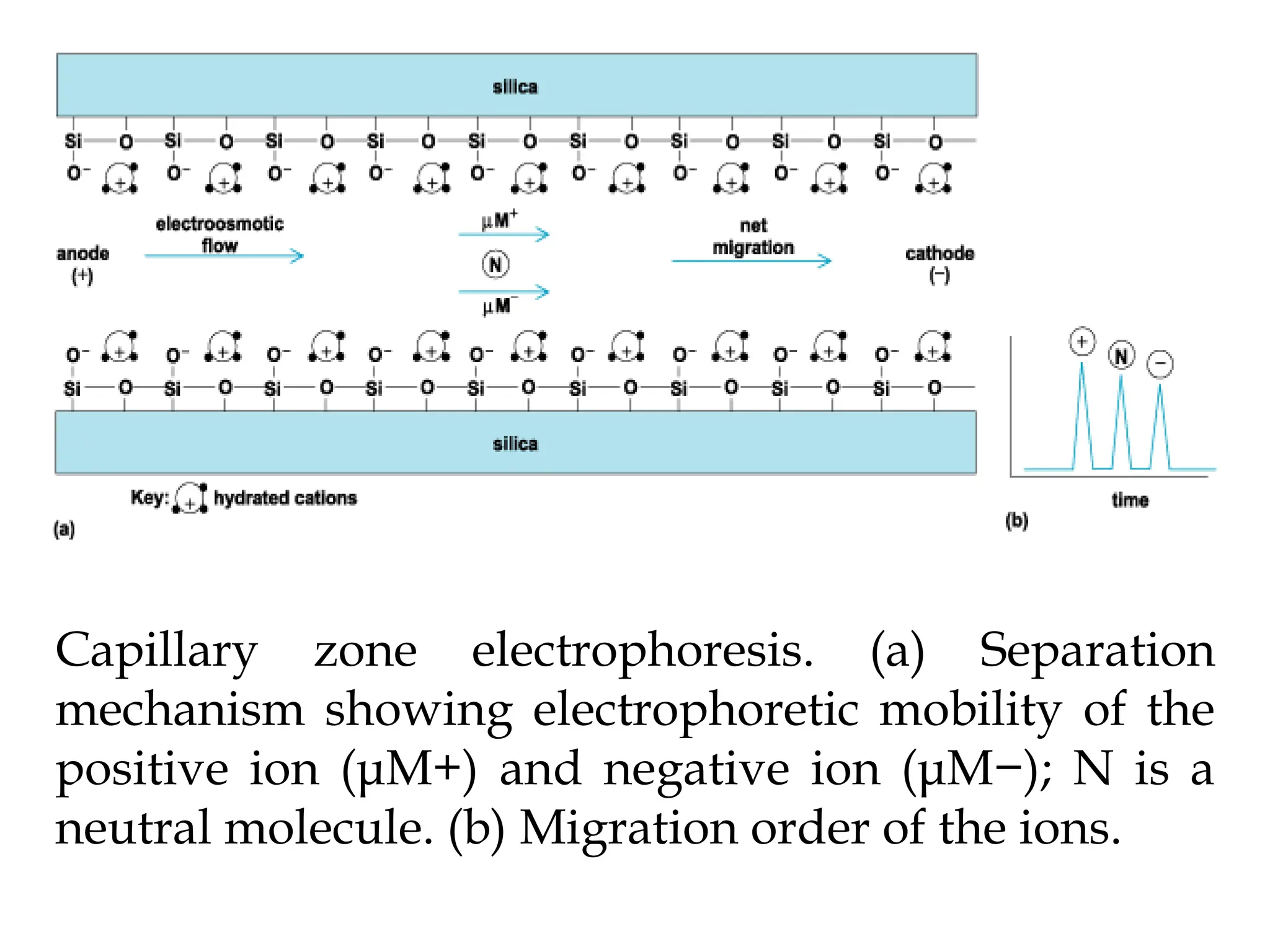
This document provides an overview of electrophoresis techniques. It defines electrophoresis as the migration of charged particles in an electric field, which allows for separation based on differences in charge-to-size ratios. Several electrophoresis methods are described, including capillary electrophoresis, paper electrophoresis, gel electrophoresis, and SDS-PAGE. Capillary electrophoresis provides high-speed separation of small sample volumes, while SDS-PAGE is used to determine protein molecular weights. Electrophoresis has applications in biochemistry, clinical analysis, environmental analysis and other fields.