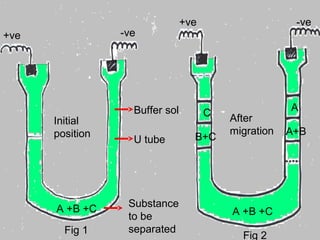
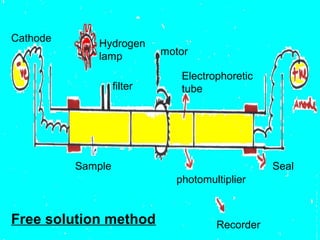
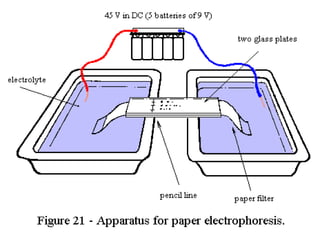
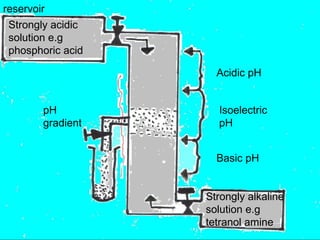
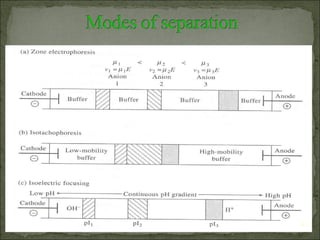
This document provides an overview of electrophoresis techniques. It discusses the basic principles of electrophoresis, including that the rate of migration depends on the charge-to-mass ratio of particles. It describes various electrophoretic techniques such as moving boundary electrophoresis, zone electrophoresis using free solution, gel, or paper methods, as well as isoelectric focusing, isotachophoresis, and capillary electrophoresis. It provides details on how each technique is performed and discusses their applications and advantages.