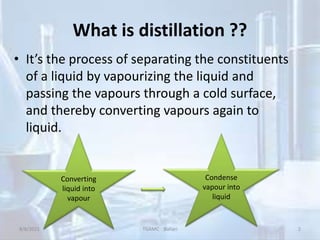
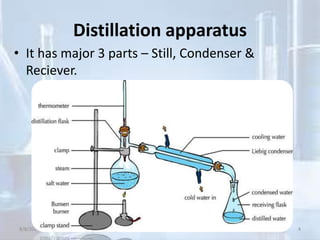
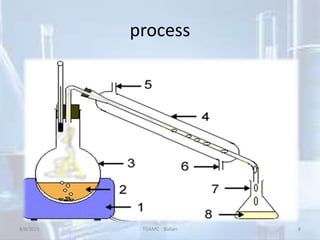
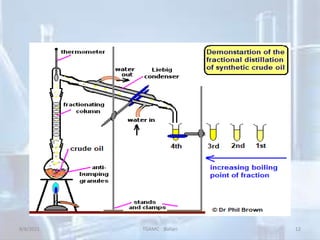
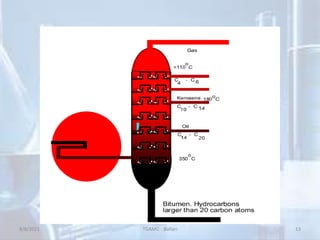
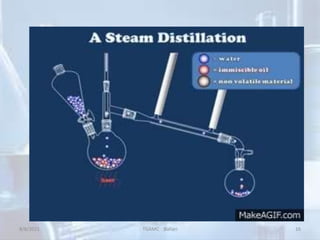
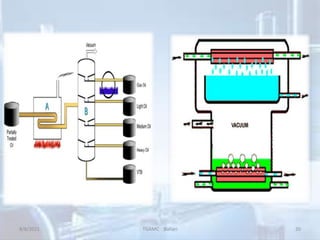
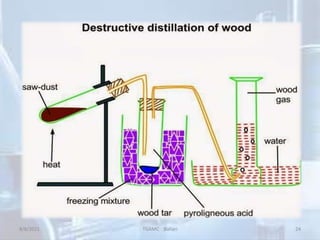
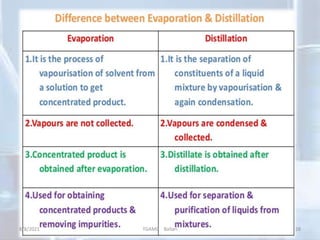
The document outlines distillation and evaporation processes, explaining distillation as the separation of liquid constituents through vaporization and condensation. It details various types of distillation, including simple, fractional, steam, vacuum, and destructive distillation, along with their applications in pharmaceuticals and industry. Additionally, evaporation is discussed as a vaporization process influenced by factors like temperature and surface area.