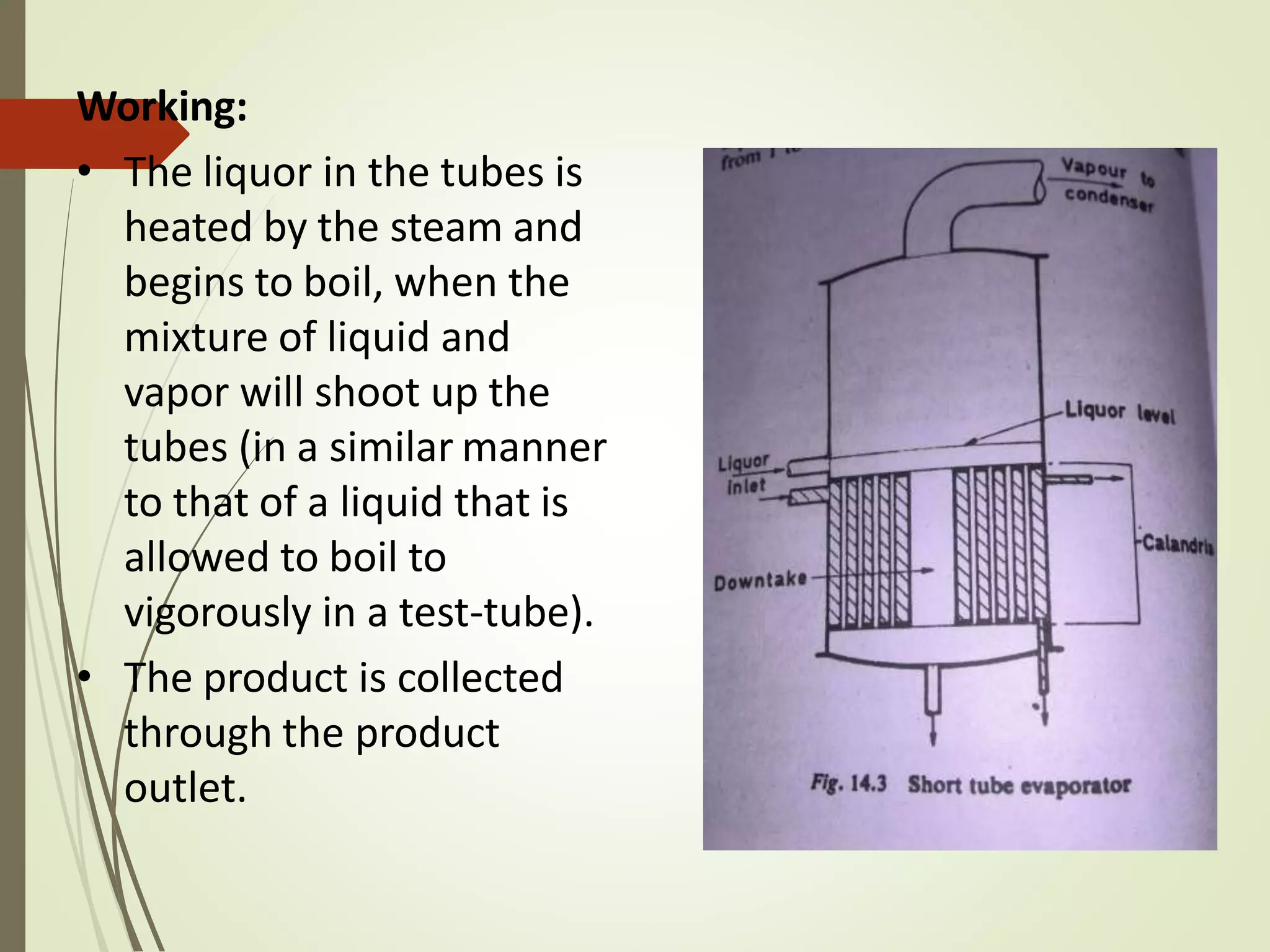
This document discusses evaporation and distillation. It defines evaporation and describes the mechanism and factors that affect the evaporation process. Various modes of evaporation and evaporators are described, including natural circulation evaporators like evaporating pans and short tube evaporators, and forced circulation evaporators. The advantages of evaporation under reduced pressure are highlighted. Applications of evaporation include concentrating solutions and producing products like salt.

























































































































![8. FRACTIONAL DISTILLATION
Fractionating columns Types
A] Packed columns
1
2
3](https://image.slidesharecdn.com/evaporationanddistillation-210214091128/75/Evaporation-and-Distillation-123-2048.jpg)

![8. FRACTIONAL DISTILLATION
Fractionating columns Types
B] Plate columns
1
2
5](https://image.slidesharecdn.com/evaporationanddistillation-210214091128/75/Evaporation-and-Distillation-125-2048.jpg)
![8. FRACTIONAL DISTILLATION
Fractionating columns Types
B] Plate columns (Bubble cap)](https://image.slidesharecdn.com/evaporationanddistillation-210214091128/75/Evaporation-and-Distillation-126-2048.jpg)



















